International System of Units
The International System of Units, internationally known by the abbreviation SI (from French Système international d'unités), is the modern form of the

| Symbol | Name | Quantity |
|---|---|---|
| s | second | time |
| m | metre | length |
| kg | kilogram | mass |
| A | ampere | electric current |
| K | kelvin | thermodynamic temperature |
| mol | mole | amount of substance |
| cd | candela | luminous intensity |
The SI comprises a
The seven base units and the 22 coherent derived units with special names and symbols may be used in combination to express other coherent derived units. Since the sizes of coherent units will be convenient for only some applications and not for others, the SI provides twenty-four prefixes which, when added to the name and symbol of a coherent unit produce twenty-four additional (non-coherent) SI units for the same quantity; these non-coherent units are always decimal (i.e. power-of-ten) multiples and sub-multiples of the coherent unit.
The current way of defining the SI is a result of a decades-long move towards increasingly abstract and idealised formulation in which the realisations of the units are separated conceptually from the definitions. A consequence is that as science and technologies develop, new and superior realisations may be introduced without the need to redefine the unit. One problem with artefacts is that they can be lost, damaged, or changed; another is that they introduce uncertainties that cannot be reduced by advancements in science and technology.
The original motivation for the development of the SI was the diversity of units that had sprung up within the
Definition
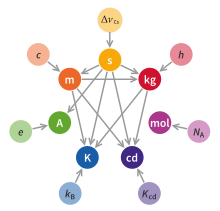
The International System of Units consists of a set of defining constants with corresponding base units, derived units, and a set of decimal-based multipliers that are used as prefixes.[1]: 125
SI defining constants
| Symbol | Defining constant | Exact value |
|---|---|---|
| ΔνCs | hyperfine transition frequency of Cs | 9192631770 Hz |
| c | speed of light | 299792458 m/s |
| h | Planck constant | 6.62607015×10−34 J⋅s |
| e | elementary charge | 1.602176634×10−19 C |
| k | Boltzmann constant | 1.380649×10−23 J/K |
| NA | Avogadro constant | 6.02214076×1023 mol−1 |
| Kcd | luminous efficacy of 540 THz radiation | 683 lm/W |
The seven defining constants are the most fundamental feature of the definition of the system of units.[1]: 125 The magnitudes of all SI units are defined by declaring that seven constants have certain exact numerical values when expressed in terms of their SI units. These defining constants are the speed of light in vacuum c, the hyperfine transition frequency of caesium ΔνCs, the Planck constant h, the elementary charge e, the Boltzmann constant k, the Avogadro constant NA, and the luminous efficacy Kcd. The nature of the defining constants ranges from fundamental constants of nature such as c to the purely technical constant Kcd. The values assigned to these constants were fixed to ensure continuity with previous definitions of the base units.[1]: 128
SI base units
The SI selects seven units to serve as base units, corresponding to seven base physical quantities. They are the second, with the symbol s, which is the SI unit of the physical quantity of time; the metre, symbol m, the SI unit of length; kilogram (kg, the unit of mass); ampere (A, electric current); kelvin (K, thermodynamic temperature); mole (mol, amount of substance); and candela (cd, luminous intensity).[1] The base units are defined in terms of the defining constants. For example, the kilogram is defined like this:[1]: 131
- 1 kg = (299792458)2/(6.62607015×10−34)(9192631770)h ΔνCs/c2.
All units in the SI can be expressed in terms of the base units, and the base units serve as a preferred set for expressing or analysing the relationships between units. The choice of which and even how many quantities to use as base quantities is not fundamental or even unique – it is a matter of convention.[1]: 126
| Unit name | Unit symbol | Dimension symbol | Quantity name | Typical symbols | Definition |
|---|---|---|---|---|---|
| second | s | time | The duration of 9192631770 periods of the radiation corresponding to the transition between the two caesium-133 atom.
| ||
| metre | m | length | , , , etc. | The distance travelled by light in vacuum in 1/299792458 second. | |
| kilogram [n 1] |
kg | mass | The kilogram is defined by setting the Planck constant h to 6.62607015×10−34 J⋅s (J = kg⋅m2⋅s−2), given the definitions of the metre and the second.[2] | ||
| ampere | A | electric current | The flow of 1/1.602176634×10−19 times the elementary charge e per second, which is approximately 6.2415090744×1018 elementary charges per second. | ||
| kelvin | K | thermodynamic temperature |
The kelvin is defined by setting the fixed numerical value of the Boltzmann constant k to 1.380649×10−23 J⋅K−1, (J = kg⋅m2⋅s−2), given the definition of the kilogram, the metre, and the second. | ||
| mole | mol | amount of substance | The amount of substance of 6.02214076×1023 elementary entities.[n 2] This number is the fixed numerical value of the Avogadro constant, NA, when expressed in the unit mol−1. | ||
| candela | cd | luminous intensity | The luminous intensity, in a given direction, of a source that emits monochromatic radiation of frequency 5.4×1014 hertz and that has a radiant intensity in that direction of 1/683 watt per steradian. | ||
| |||||
Derived units
The system allows for an unlimited number of additional units, called derived units, which can always be represented as products of powers of the base units, possibly with a nontrivial numeric multiplier. When that multiplier is one, the unit is called a coherent derived unit. For example, the coherent derived SI unit unit of velocity is the metre per second, with the symbol m/s.[1]: 139 The base and coherent derived units of the SI together form a coherent system of units (the set of coherent SI units). A useful property of a coherent system is that when the numerical values of physical quantities are expressed in terms of the units of the system, then the equations between the numerical values have exactly the same form, including numerical factors, as the corresponding equations between the physical quantities.[3]: 6
Twenty-two coherent derived units have been provided with special names and symbols as shown in the table below. The radian and steradian have no base units but are treated as derived units for historical reasons.[1]: 137
| Name | Symbol | Quantity | In SI base units | In other SI units |
|---|---|---|---|---|
| radian[N 1] | rad | plane angle | m/m | 1 |
| steradian[N 1] | sr | solid angle | m2/m2 | 1 |
| hertz | Hz | frequency | s−1 | |
| newton | N | force, weight | kg⋅m⋅s−2 | |
| pascal | Pa | stress
|
kg⋅m−1⋅s−2 | N/m2 = J/m3 |
| joule | J | work, heat
|
kg⋅m2⋅s−2 | N⋅m = Pa⋅m3 |
| watt | W | power, radiant flux | kg⋅m2⋅s−3 | J/s |
| coulomb | C | electric charge | s⋅A | |
| volt | V | electric potential, voltage, emf | kg⋅m2⋅s−3⋅A−1 | W/A = J/C |
| farad | F | capacitance | kg−1⋅m−2⋅s4⋅A2 | C/V = C2/J |
ohm
|
Ω | reactance
|
kg⋅m2⋅s−3⋅A−2 | V/A = J⋅s/C2 |
| siemens | S | electrical conductance
|
kg−1⋅m−2⋅s3⋅A2 | Ω−1 |
| weber | Wb | magnetic flux | kg⋅m2⋅s−2⋅A−1 | V⋅s |
| tesla | T | magnetic flux density
|
kg⋅s−2⋅A−1 | Wb/m2 |
| henry | H | inductance | kg⋅m2⋅s−2⋅A−2 | Wb/A |
degree Celsius
|
°C | temperature relative to 273.15 K | K | |
| lumen | lm | luminous flux | cd⋅m2/m2 | cd⋅sr |
| lux | lx | illuminance | cd⋅m2/m4 | lm/m2 = cd⋅sr⋅m−2 |
| becquerel | Bq | activity referred to a radionuclide (decays per unit time) | s−1 | |
| gray | Gy | ionising radiation )
|
m2⋅s−2 | J/kg |
| sievert | Sv | ionising radiation )
|
m2⋅s−2 | J/kg |
| katal | kat | catalytic activity
|
mol⋅s−1 | |
| Notes | ||||
The derived units in the SI are formed by powers, products, or quotients of the base units and are potentially unlimited in number.[5]: 103 [4]: 14, 16

A combination of base and derived units may be used to express a derived unit. For example, the SI unit of force is the newton (N), the SI unit of pressure is the pascal (Pa) – and the pascal can be defined as one newton per square metre (N/m2).[6]
Prefixes
Like all metric systems, the SI uses metric prefixes to systematically construct, for the same physical quantity, a set of units that are decimal multiples of each other over a wide range. For example, driving distances are normally given in kilometres (symbol km) rather than in metres. Here the metric prefix 'kilo-' (symbol 'k') stands for a factor of 1000; thus, 1 km = 1000 m.
The current version of the SI provides twenty-four metric prefixes that signify decimal powers ranging from 10−30 to 1030, the most recent being adopted in 2022.[1]: 143–144 [7][8][9] Most prefixes correspond to integer powers of 1000; the only ones that do not are those for 10, 1/10, 100, and 1/100. The conversion between different SI units for one and the same physical quantity is always through a power of ten. This is why the SI (and metric systems more generally) are called decimal systems of measurement units.[10]
The grouping formed by a prefix symbol attached to a unit symbol (e.g. 'km', 'cm') constitutes a new inseparable unit symbol. This new symbol can be raised to a positive or negative power. It can also be combined with other unit symbols to form compound unit symbols.[1]: 143 For example, g/cm3 is an SI unit of density, where cm3 is to be interpreted as (cm)3.
Prefixes are added to unit names to produce multiples and
The BIPM specifies 24 prefixes for the International System of Units (SI):
| Prefix | Base 10 | Decimal | Adoption [nb 1] | |
|---|---|---|---|---|
| Name | Symbol | |||
| quetta | Q | 1030 | 1000000000000000000000000000000 | 2022[12] |
| ronna | R | 1027 | 1000000000000000000000000000 | |
| yotta | Y | 1024 | 1000000000000000000000000 | 1991 |
| zetta | Z | 1021 | 1000000000000000000000 | |
| exa | E | 1018 | 1000000000000000000 | 1975[13] |
| peta | P | 1015 | 1000000000000000 | |
| tera | T | 1012 | 1000000000000 | 1960 |
| giga | G | 109 | 1000000000 | |
| mega | M | 106 | 1000000 | 1873 |
| kilo | k | 103 | 1000 | 1795 |
| hecto | h | 102 | 100 | |
| deca | da | 101 | 10 | |
| — | — | 100 | 1 | — |
| deci | d | 10−1 | 0.1 | 1795 |
| centi | c | 10−2 | 0.01 | |
| milli | m | 10−3 | 0.001 | |
| micro | μ | 10−6 | 0.000001 | 1873 |
| nano | n | 10−9 | 0.000000001 | 1960 |
| pico | p | 10−12 | 0.000000000001 | |
| femto | f | 10−15 | 0.000000000000001 | 1964 |
| atto | a | 10−18 | 0.000000000000000001 | |
| zepto | z | 10−21 | 0.000000000000000000001 | 1991 |
| yocto | y | 10−24 | 0.000000000000000000000001 | |
| ronto | r | 10−27 | 0.000000000000000000000000001 | 2022[12] |
| quecto | q | 10−30 | 0.000000000000000000000000000001 | |
| ||||
Coherent and non-coherent SI units
The base units and the derived units formed as the product of powers of the base units with a numerical factor of one form a
The kilogram is the only coherent SI unit whose name and symbol include a prefix. For historical reasons, the names and symbols for multiples and sub-multiples of the unit of mass are formed as if the gram were the base unit. Prefix names and symbols are attached to the unit name gram and the unit symbol g respectively. For example, 10−6 kg is written milligram and mg, not microkilogram and μkg.[1]: 144
Several different quantities may share the same coherent SI unit. For example, the joule per kelvin (symbol J/K) is the coherent SI unit for two distinct quantities: heat capacity and entropy; another example is the ampere, which is the coherent SI unit for both electric current and magnetomotive force. This illustrates why it is important not to use the unit alone to specify the quantity. As the SI Brochure states,[1]: 140 "this applies not only to technical texts, but also, for example, to measuring instruments (i.e. the instrument read-out needs to indicate both the unit and the quantity measured)".
Furthermore, the same coherent SI unit may be a base unit in one context, but a coherent derived unit in another. For example, the ampere is a base unit when it is a unit of electric current, but a coherent derived unit when it is a unit of magnetomotive force.[1]: 140
| Name | Symbol | Derived quantity | Typical symbol |
|---|---|---|---|
| square metre | m2 | area | A |
| cubic metre | m3 | volume | V |
| metre per second | m/s | speed, velocity | v |
| metre per second squared | m/s2 | acceleration | a |
reciprocal metre
|
m−1 | wavenumber | σ, ṽ |
| vergence (optics) | V, 1/f | ||
| kilogram per cubic metre | kg/m3 | density | ρ |
| kilogram per square metre | kg/m2 | surface density
|
ρA |
| cubic metre per kilogram | m3/kg | specific volume | v |
| ampere per square metre | A/m2 | current density | j |
ampere per metre
|
A/m | magnetic field strength
|
H |
| mole per cubic metre | mol/m3 | concentration | c |
| kilogram per cubic metre | kg/m3 | mass concentration | ρ, γ |
| candela per square metre | cd/m2 | luminance | Lv |
| Name | Symbol | Quantity | In SI base units |
|---|---|---|---|
pascal-second
|
Pa⋅s | dynamic viscosity
|
m−1⋅kg⋅s−1 |
| newton-metre | N⋅m | moment of force
|
m2⋅kg⋅s−2 |
| newton per metre | N/m | surface tension | kg⋅s−2 |
| radian per second | rad/s | angular velocity, angular frequency | s−1 |
radian per second squared
|
rad/s2 | angular acceleration | s−2 |
watt per square metre
|
W/m2 | heat flux density, irradiance | kg⋅s−3 |
joule per kelvin
|
J/K | entropy, heat capacity | m2⋅kg⋅s−2⋅K−1 |
| joule per kilogram-kelvin | J/(kg⋅K) | specific entropy
|
m2⋅s−2⋅K−1 |
| joule per kilogram | J/kg | specific energy | m2⋅s−2 |
| watt per metre-kelvin | W/(m⋅K) | thermal conductivity
|
m⋅kg⋅s−3⋅K−1 |
| joule per cubic metre | J/m3 | energy density | m−1⋅kg⋅s−2 |
| volt per metre | V/m | electric field strength
|
m⋅kg⋅s−3⋅A−1 |
| coulomb per cubic metre | C/m3 | electric charge density
|
m−3⋅s⋅A |
| coulomb per square metre | C/m2 | electric displacement
|
m−2⋅s⋅A |
| farad per metre | F/m | permittivity | m−3⋅kg−1⋅s4⋅A2 |
| henry per metre | H/m | permeability | m⋅kg⋅s−2⋅A−2 |
| joule per mole | J/mol | molar energy | m2⋅kg⋅s−2⋅mol−1 |
| joule per mole-kelvin | J/(mol⋅K) | molar entropy, molar heat capacity
|
m2⋅kg⋅s−2⋅K−1⋅mol−1 |
| coulomb per kilogram | C/kg | exposure (x- and γ-rays) | kg−1⋅s⋅A |
| gray per second | Gy/s | absorbed dose rate
|
m2⋅s−3 |
| watt per steradian | W/sr | radiant intensity | m2⋅kg⋅s−3 |
| watt per square metre-steradian | W/(m2⋅sr) | radiance | kg⋅s−3 |
| katal per cubic metre | kat/m3 | catalytic activity concentration
|
m−3⋅s−1⋅mol |
Lexicographic conventions
Unit names
According to the SI Brochure,
The English spelling and even names for certain SI units and metric prefixes depend on the variety of English used.
Unit symbols and the values of quantities
Symbols of SI units are intended to be unique and universal, independent of the context language.[5]: 130–35 The SI Brochure has specific rules for writing them.[5]: 130–35
In addition, the SI Brochure provides style conventions for among other aspects of displaying quantities units: the quantity symbols, formatting of numbers and the decimal marker, expressing measurement uncertainty, multiplication and division of quantity symbols, and the use of pure numbers and various angles.[1]: 147
In the United States, the guideline produced by the National Institute of Standards and Technology (NIST)[11]: 37 clarifies language-specific details for American English that were left unclear by the SI Brochure, but is otherwise identical to the SI Brochure.[14] For example, since 1979, the litre may exceptionally be written using either an uppercase "L" or a lowercase "l", a decision prompted by the similarity of the lowercase letter "l" to the numeral "1", especially with certain typefaces or English-style handwriting. The American NIST recommends that within the United States "L" be used rather than "l".[11]
Realisation of units
Metrologists carefully distinguish between the definition of a unit and its realisation. The SI units are defined by declaring that seven defining constants[1]: 125–129 have certain exact numerical values when expressed in terms of their SI units. The realisation of the definition of a unit is the procedure by which the definition may be used to establish the value and associated uncertainty of a quantity of the same kind as the unit.[1]: 135
For each base unit the BIPM publishes a mises en pratique, (French for 'putting into practice; implementation',[16]) describing the current best practical realisations of the unit.[17] The separation of the defining constants from the definitions of units means that improved measurements can be developed leading to changes in the mises en pratique as science and technology develop, without having to revise the definitions.
The published mise en pratique is not the only way in which a base unit can be determined: the SI Brochure states that "any method consistent with the laws of physics could be used to realise any SI unit".[5]: 111 Various consultative committees of the CIPM decided in 2016 that more than one mise en pratique would be developed for determining the value of each unit.[18] These methods include the following:
- At least three separate experiments be carried out yielding values having a relative
- The definition of the thermometry and dielectric constant gas thermometry be better than one part in 10−6 and that these values be corroborated by other measurements.[21]
Organizational status
The International System of Units, or SI,
The only other types of measurement system that still have widespread use across the world are the imperial and US customary measurement systems. The international yard and pound are defined in terms of the SI.[22]
International System of Quantities
The quantities and equations that provide the context in which the SI units are defined are now referred to as the International System of Quantities (ISQ). The ISQ is based on the
Controlling authority
The SI is regulated and continually developed by three international organisations that were established in 1875 under the terms of the Metre Convention. They are the General Conference on Weights and Measures (CGPM[b]),[26] the International Committee for Weights and Measures (CIPM[c]), and the International Bureau of Weights and Measures (BIPM[d]). All the decisions and recommendations concerning units are collected in a brochure called
History
CGS and MKS systems

The concept of a system of units emerged a hundred years before the SI. In the 1860s,
Metre Convention
A French-inspired initiative for international cooperation in metrology led to the signing in 1875 of the Metre Convention, also called Treaty of the Metre, by 17 nations.[e][30]: 353–354 The General Conference on Weights and Measures (French: Conférence générale des poids et mesures – CGPM), which was established by the Metre Convention,[29] brought together many international organisations to establish the definitions and standards of a new system and to standardise the rules for writing and presenting measurements.[31]: 37 [32] Initially the convention only covered standards for the metre and the kilogram. This became the foundation of the MKS system of units.[4]: 2
Giovanni Giorgi and the problem of electrical units
At the close of the 19th century three different systems of units of measure existed for electrical measurements: a
Electric current with named unit 'ampere' was chosen as the base unit, and the other electrical quantities derived from it according to the laws of physics. When combined with the MKS the new system, known as MKSA was approved in 1946.[4]
9th CGPM, the precursor to SI
In 1948, the 9th CGPM commissioned a study to assess the measurement needs of the scientific, technical, and educational communities and "to make recommendations for a single practical system of units of measurement, suitable for adoption by all countries adhering to the Metre Convention".[36] This working document was Practical system of units of measurement. Based on this study, the 10th CGPM in 1954 defined an international system derived six base units: the metre, kilogram, second, ampere, degree Kelvin, and candela.
The 9th CGPM also approved the first formal recommendation for the writing of symbols in the metric system when the basis of the rules as they are now known was laid down.[37] These rules were subsequently extended and now cover unit symbols and names, prefix symbols and names, how quantity symbols should be written and used, and how the values of quantities should be expressed.[5]: 104, 130
Birth of the SI
The 10th CGPM in 1954 resolved to create an international system of units[31]: 41 and in 1960, the 11th CGPM published a long detailed report, including the decision to name system the International System of Units, abbreviated SI from the French name, Le Système international d'unités.[5]: 110
The International Bureau of Weights and Measures (BIPM) has described SI as "the modern form of metric system".[5]: 95 In 1971 the mole became the seventh base unit of the SI.[4]: 2
2019 redefinition
After the metre was redefined in 1960, the International Prototype of the Kilogram (IPK) was the only physical artefact upon which base units (directly the kilogram and indirectly the ampere, mole and candela) depended for their definition, making these units subject to periodic comparisons of national standard kilograms with the IPK.[38] During the 2nd and 3rd Periodic Verification of National Prototypes of the Kilogram, a significant divergence had occurred between the mass of the IPK and all of its official copies stored around the world: the copies had all noticeably increased in mass with respect to the IPK. During extraordinary verifications carried out in 2014 preparatory to redefinition of metric standards, continuing divergence was not confirmed. Nonetheless, the residual and irreducible instability of a physical IPK undermined the reliability of the entire metric system to precision measurement from small (atomic) to large (astrophysical) scales.[39] By avoiding the use of an artifact to define units, all issues with the loss, damage, and change of the artifact are avoided.[1]: 125
A proposal was made that:[40]
- In addition to the speed of light, four constants of nature – the Planck constant, an elementary charge, the Boltzmann constant, and the Avogadro constant – be defined to have exact values
- The International Prototype of the Kilogram be retired
- The current definitions of the kilogram, ampere, kelvin, and mole be revised
- The wording of base unit definitions should change emphasis from explicit unit to explicit constant definitions.
The new definitions were adopted at the 26th CGPM on 16 November 2018, and came into effect on 20 May 2019.[41] The change was adopted by the European Union through Directive (EU) 2019/1258.[42]
Prior to its redefinition in 2019, the SI was defined through the seven base units from which the derived units were constructed as products of powers of the base units. After the redefinition, the SI is defined by fixing the numerical values of seven defining constants. This has the effect that the distinction between the base units and derived units is, in principle, not needed, since all units, base as well as derived, may be constructed directly from the defining constants. Nevertheless, the distinction is retained because "it is useful and historically well established", and also because the ISO/IEC 80000 series of standards, which define the International System of Quantities (ISQ), specifies base and derived quantities that necessarily have the corresponding SI units.[1]: 129
Related units
Non-SI units accepted for use with SI
Many non-SI units continue to be used in the scientific, technical, and commercial literature. Some units are deeply embedded in history and culture, and their use has not been entirely replaced by their SI alternatives. The CIPM recognised and acknowledged such traditions by compiling a list of
Metric units not recognised by SI
Although the term metric system is often used as an informal alternative name for the International System of Units, that exist outside of any system of units. Most of the units of the other metric systems are not recognised by the SI.
Unacceptable uses
Sometimes, SI unit name variations are introduced, mixing information about the corresponding physical quantity or the conditions of its measurement; however, this practice is unacceptable with the SI. "Unacceptability of mixing information with units: When one gives the value of a quantity, any information concerning the quantity or its conditions of measurement must be presented in such a way as not to be associated with the unit."[5] Instances include: "
See also
- Conversion of units – Comparison of various scales
- List of international common standards
- Metrication
- Outline of the metric system – Overview of and topical guide to the metric system
Organisations
- International Bureau of Weights and Measures – Intergovernmental measurement science and measurement standards setting organisation
- Institute for Reference Materials and Measurements (EU)
- National Institute of Standards and Technology – Measurement standards laboratory in the United States (US)
Standards and conventions
- Conventional electrical unit – Historical high-precision units of measurement
- Coordinated Universal Time (UTC) – Primary time standard
- Unified Code for Units of Measure – System of codes for unambiguously representing measurement units
Notes
- ^ Ohm's law: 1 Ω = 1 V/A from the relationship E = I × R, where E is electromotive force or voltage (unit: volt), I is current (unit: ampere), and R is resistance (unit: ohm).
- ^ From French: Conférence générale des poids et mesures.
- ^ from French: Comité international des poids et mesures
- ^ from French: Bureau international des poids et mesures
- ^ Argentina, Austria-Hungary, Belgium, Brazil, Denmark, France, German Empire, Italy, Peru, Portugal, Russia, Spain, Sweden and Norway, Switzerland, Ottoman Empire, United States, and Venezuela.
- Attribution
[1]![]() This article incorporates text from this source, which is available under the CC BY 3.0 license.
This article incorporates text from this source, which is available under the CC BY 3.0 license.
References
- ^ ISBN 978-92-822-2272-0, archivedfrom the original on 18 October 2021
- ^ Materese, Robin (16 November 2018). "Historic Vote Ties Kilogram and Other Units to Natural Constants". NIST. Retrieved 16 November 2018.
- ^ ISO 80000-1:2009 Quantities and units – Part 1: General.
- ^ a b c d e f g h i j David B. Newell; Eite Tiesinga, eds. (2019). The International System of Units (SI) (PDF) (NIST Special publication 330, 2019 ed.). Gaithersburg, MD: NIST. Retrieved 30 November 2019.
- ^ ISBN 92-822-2213-6, archived(PDF) from the original on 4 June 2021, retrieved 16 December 2021
- ^ "Units & Symbols for Electrical & Electronic Engineers". Institution of Engineering and Technology. 1996. pp. 8–11. Archived from the original on 28 June 2013. Retrieved 19 August 2013.
- ^ "Earth now weighs six ronnagrams: New metric prefixes voted in". phys.org. 18 November 2022.
- ^ "List of Resolutions for the 27th meeting of the General Conference on Weights and Measures" (PDF). BIPM. 18 November 2022. Archived from the original (PDF) on 18 November 2022. Retrieved 18 November 2022.
- ^ "New prefixes for the SI adopted by the General Conference on Weights and Measures". BIPM. Retrieved 11 January 2023.
- ^ "Decimal Nature of the Metric System". US Metric Association. 2015. Archived from the original on 15 April 2020. Retrieved 15 April 2020.
- ^ a b c Thompson, Ambler; Taylor, Barry N. (March 2008). Guide for the Use of the International System of Units (SI) (Report). National Institute of Standards and Technology. §10.5.3. Retrieved 21 January 2022.
- ^ a b "On the extension of the range of SI prefixes". 18 November 2022. Retrieved 5 February 2023.
- ^ "Metric (SI) Prefixes". NIST.
- ^ "Interpretation of the International System of Units (the Metric System of Measurement) for the United States" (PDF). Federal Register. 73 (96): 28432–28433. 9 May 2008. FR Doc number E8-11058. Retrieved 28 October 2009.
- ^ "Avogadro Project". National Physical Laboratory. Retrieved 19 August 2010.
- NIST. 2013. Archivedfrom the original on 14 July 2017. Retrieved 9 May 2020.
- BIPM. 2019. Archivedfrom the original on 9 April 2020. Retrieved 11 April 2020.
- ^ "International Committee for Weights and Measures – Proceedings of the 106th meeting" (PDF).
- ^ "Recommendations of the Consultative Committee for Mass and Related Quantities to the International Committee for Weights and Measures" (PDF). 12th Meeting of the CCM. Sèvres: Bureau International des Poids et Mesures. 26 March 2010. Archived from the original (PDF) on 14 May 2013. Retrieved 27 June 2012.
- ^ "Recommendations of the Consultative Committee for Amount of Substance – Metrology in Chemistry to the International Committee for Weights and Measures" (PDF). 16th Meeting of the CCQM. Sèvres: Bureau International des Poids et Mesures. 15–16 April 2010. Archived from the original (PDF) on 14 May 2013. Retrieved 27 June 2012.
- ^ "Recommendations of the Consultative Committee for Thermometry to the International Committee for Weights and Measures" (PDF). 25th Meeting of the CCT. Sèvres: Bureau International des Poids et Mesures. 6–7 May 2010. Archived from the original (PDF) on 14 May 2013. Retrieved 27 June 2012.
- ^ United States. National Bureau of Standards (1959). Research Highlights of the National Bureau of Standards. U.S. Department of Commerce, National Bureau of Standards. p. 13. Retrieved 31 July 2019.
- ^ "1.16" (PDF). International vocabulary of metrology – Basic and general concepts and associated terms (VIM) (3rd ed.). International Bureau of Weights and Measures (BIPM): Joint Committee for Guides in Metrology. 2012. Retrieved 28 March 2015.
- ISBN 3642007384.
- ^ "ISO 80000-1:2022 Quantities and units Part 1: General".
- ^ "Interpretation of the International System of Units (the Metric System of Measurement) for the United States". Federal Register. 73. National Institute of Standards and Technology: 28432. 16 May 2008. Archived from the original on 16 August 2017. Retrieved 6 December 2022.
- ^ "VIM3: International Vocabulary of Metrology". BIPM. Archived from the original on 31 October 2020.
- ^ "Amtliche Maßeinheiten in Europa 1842" [Official units of measure in Europe 1842]. spasslernen (in German). 1 May 2009. Archived from the original on 25 September 2012. Retrieved 26 March 2011. Text version of Malaisé's book: Malaisé, Ferdinand von (1842). Theoretisch-practischer Unterricht im Rechnen [Theoretical and practical instruction in arithmetic] (in German). München: Verlag des Verf. pp. 307–322. Retrieved 7 January 2013.
- ^ National Bureau of Standards. p. 12.
- ISBN 978-0-349-11507-8.
- ^ S2CID 258172637.
- ISBN 978-0-19-530786-3.
- ISBN 978-0-19-860522-5.
- ^ Maxwell, J. C. (1873). A treatise on electricity and magnetism. Vol. 2. Oxford: Clarendon Press. pp. 242–245. Retrieved 12 May 2011.
- ^ "Historical figures: Giovanni Giorgi". International Electrotechnical Commission. 2011. Archived from the original on 15 May 2011. Retrieved 5 April 2011.
- ^ "BIPM – Resolution 6 of the 9th CGPM". Bipm.org. 1948. Retrieved 22 August 2017.
- ^ "Resolution 7 of the 9th meeting of the CGPM (1948): Writing and printing of unit symbols and of numbers". International Bureau of Weights and Measures. Retrieved 6 November 2012.
- ^ "Redefining the kilogram". UK National Physical Laboratory. Retrieved 30 November 2014.
- ^ "A Turning Point for Humanity: Redefining the World's Measurement System". NIST. 12 May 2018. Retrieved 16 January 2024.
- BIPM. p. 188. Retrieved 27 April 2021.
- BIPM. p. 7.
[BIPM director Martin] Milton responded to a question about what would happen if ... the CIPM or the CGPM voted not to move forward with the redefinition of the SI. He responded that he felt that by that time the decision to move forward should be seen as a foregone conclusion.
- Eur-Lex. 23 July 2019. Retrieved 28 August 2019.
- NIST. Archivedfrom the original on 16 March 2020. Retrieved 14 April 2020.
... the International System of Units (SI), popularly known as the metric system.
Further reading
- Unit Systems in Electromagnetism
- MW Keller et al. (PDF) Metrology Triangle Using a Watt Balance, a Calculable Capacitor, and a Single-Electron Tunneling Device
- "The Current SI Seen From the Perspective of the Proposed New SI" (PDF). Barry N. Taylor. Journal of Research of the National Institute of Standards and Technology, Vol. 116, No. 6, Pgs. 797–807, Nov–Dec 2011.
- B. N. Taylor, Ambler Thompson, International System of Units (SI), ISBN 1437915582.