Iridium
 | |||||||||||||||||||||||||||||||||
| Iridium | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /ɪˈrɪdiəm/ | ||||||||||||||||||||||||||||||||
| Appearance | Silvery white | ||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Ir) | |||||||||||||||||||||||||||||||||
| Iridium in the periodic table | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
kJ/mol | |||||||||||||||||||||||||||||||||
| Heat of vaporization | 564 kJ/mol | ||||||||||||||||||||||||||||||||
| Molar heat capacity | 25.10 J/(mol·K) | ||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||
Discovery and first isolation | Smithson Tennant (1803) | ||||||||||||||||||||||||||||||||
| Isotopes of iridium | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
Iridium is a
Iridium was discovered in 1803 among insoluble impurities in natural platinum. Smithson Tennant, the primary discoverer, named it after the Greek goddess Iris, personification of the rainbow, because of the striking and diverse colors of its salts. Iridium is one of the rarest elements in Earth's crust, with estimated annual production and consumption of only 7.3 tonnes (16 thousand pounds) in 2018.[12]
The dominant uses of iridium are the metal itself and its alloys, as in high-performance spark plugs, crucibles for recrystallization of semiconductors at high temperatures, and electrodes for the production of chlorine in the chloralkali process. Important compounds of iridium are chlorides and iodides in industrial catalysis. Iridium is a component of some OLEDs.
Iridium is found in meteorites in much higher abundance than in the Earth's crust.[13] For this reason, the unusually high abundance of iridium in the clay layer at the Cretaceous–Paleogene boundary gave rise to the Alvarez hypothesis that the impact of a massive extraterrestrial object caused the extinction of dinosaurs and many other species 66 million years ago, now known to be produced by the impact that formed the Chicxulub crater. Similarly, an iridium anomaly in core samples from the Pacific Ocean suggested the Eltanin impact of about 2.5 million years ago.[14]
It is thought that the total amount of iridium in the planet is much higher than that observed in crustal rocks, but as with other platinum-group metals, the high density and
Characteristics
Physical properties
A member of the
Iridium's
The measured
Iridium is extremely brittle, to the point of being hard to weld because the heat-affected zone cracks, but it can be made more ductile by addition of small quantities of titanium and zirconium (0.2% of each apparently works well).[22]
The
Chemical properties
Iridium is the most
Isotopes
Iridium has two naturally occurring stable
At least 32
Chemistry
| Oxidation states[b] | |
|---|---|
| −3 | [Ir(CO) 3]3− |
| −1 | [Ir(CO)3(PPh3)]1− |
| 0 | Ir4(CO)12 |
| +1 | [IrCl(CO)(PPh3)2] |
| +2 | Ir(C5H5)2 |
| +3 | IrCl3 |
| +4 | IrO2 |
| +5 | Ir4F20 |
| +6 | IrF 6 |
| +7 | [Ir(O2)O2]+ |
| +8 | IrO4 |
| +9 | [IrO4]+[4] |
Oxidation states
Iridium forms compounds in
Binary compounds
Iridium does not form
2. It is a blue black solid that adopts the fluorite structure.[15] A sesquioxide, Ir
2O
3, has been described as a blue-black powder, which is oxidized to IrO
2 by HNO
3.[27] The corresponding disulfides, diselenides, sesquisulfides, and sesquiselenides are known, as well as IrS
3.[15]
Binary trihalides, IrX
3, are known for all of the halogens.
6, is a volatile yellow solid, composed of octahedral molecules. It decomposes in water and is reduced to IrF
4.[15] Iridium pentafluoride is also a strong oxidant, but it is a tetramer, Ir
4F
20, formed by four corner-sharing octahedra.[15]
Complexes
Iridium has extensive
Iridium in its complexes is always
6Ir
2H
11 is believed to contain both the IrH4−
5 and the 18-electron IrH5−
4 anion.[38]
Iridium also forms oxyanions with oxidation states +4 and +5. K
2IrO
3 and KIrO
3 can be prepared from the reaction of potassium oxide or potassium superoxide with iridium at high temperatures. Such solids are not soluble in conventional solvents.[39]
Just like many elements, iridium forms important chloride complexes. Hexachloroiridic (IV) acid, H
2IrCl
6, and its ammonium salt are the most common iridium compounds from both industrial and preparative perspectives.[40] They are intermediates in the purification of iridium and used as precursors for most other iridium compounds, as well as in the preparation of anode coatings. The IrCl2−
6 ion has an intense dark brown color, and can be readily reduced to the lighter-colored IrCl3−
6 and vice versa.[40] Iridium trichloride, IrCl
3, which can be obtained in anhydrous form from direct oxidation of iridium powder by chlorine at 650 °C,[40] or in hydrated form by dissolving Ir
2O
3 in hydrochloric acid, is often used as a starting material for the synthesis of other Ir(III) compounds.[15] Another compound used as a starting material is ammonium hexachloroiridate(III), (NH
4)
3IrCl
6.[citation needed]
In the presence of air, iridium metal dissolves in molten alkali-metal cyanides to produce the Ir(CN)3−
6 (hexacyanoiridate) ion and upon oxidation produces the most stable oxide.[citation needed]
Organoiridium chemistry


Iridium complexes played a pivotal role in the development of
History
Platinum group

The discovery of iridium is intertwined with that of platinum and the other metals of the


In 1735, Antonio de Ulloa and Jorge Juan y Santacilia saw Native Americans mining platinum while the Spaniards were travelling through Colombia and Peru for eight years. Ulloa and Juan found mines with the whitish metal nuggets and took them home to Spain. Ulloa returned to Spain and established the first mineralogy lab in Spain and was the first to systematically study platinum, which was in 1748. His historical account of the expedition included a description of platinum as being neither separable nor calcinable. Ulloa also anticipated the discovery of platinum mines. After publishing the report in 1748, Ulloa did not continue to investigate the new metal. In 1758, he was sent to superintend mercury mining operations in Huancavelica.[46]
In 1741, Charles Wood,[48] a British metallurgist, found various samples of Colombian platinum in Jamaica, which he sent to William Brownrigg for further investigation.
In 1750, after studying the platinum sent to him by Wood, Brownrigg presented a detailed account of the metal to the
Discovery
In 1803 British scientist Smithson Tennant (1761–1815) analyzed the insoluble residue and concluded that it must contain a new metal. Vauquelin treated the powder alternately with alkali and acids[25] and obtained a volatile new oxide, which he believed to be of this new metal—which he named ptene, from the Greek word πτηνός ptēnós, "winged".[51][52] Tennant, who had the advantage of a much greater amount of residue, continued his research and identified the two previously undiscovered elements in the black residue, iridium and osmium.[16][25] He obtained dark red crystals (probably of Na
2[IrCl
6]·nH
2O) by a sequence of reactions with sodium hydroxide and hydrochloric acid.[52] He named iridium after Iris (Ἶρις), the Greek winged goddess of the rainbow and the messenger of the Olympian gods, because many of the salts he obtained were strongly colored.[c][53] Discovery of the new elements was documented in a letter to the Royal Society on June 21, 1804.[16][54]
Metalworking and applications
British scientist
2 and H
2 gas for each 1 kilogram (2.2 lb) of iridium.[16]
These extreme difficulties in melting the metal limited the possibilities for handling iridium. John Isaac Hawkins was looking to obtain a fine and hard point for fountain pen nibs, and in 1834 managed to create an iridium-pointed gold pen. In 1880, John Holland and William Lofland Dudley were able to melt iridium by adding phosphorus and patented the process in the United States; British company Johnson Matthey later stated they had been using a similar process since 1837 and had already presented fused iridium at a number of World Fairs.[16] The first use of an alloy of iridium with ruthenium in thermocouples was made by Otto Feussner in 1933. These allowed for the measurement of high temperatures in air up to 2,000 °C (3,630 °F).[16]
In
Occurrence
Along with all elements having
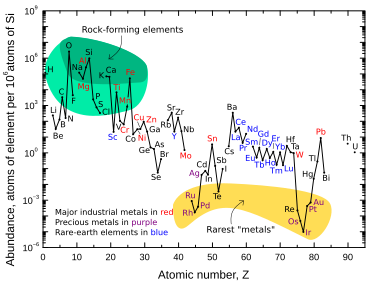

Iridium is one of the nine least abundant stable
Iridium is found in nature as an uncombined element or in natural
Marine oceanography
Iridium is found within marine organisms,
Iridium in sediments can come from cosmic dust, volcanoes, precipitation from seawater, microbial processes, or hydrothermal vents,[69] and its abundance can be strongly indicative of the source.[70][69] It tends to associate with other ferrous metals in manganese nodules.[67] Iridium is one of the characteristic elements of extraterrestrial rocks, and, along with osmium, can be used as a tracer element for meteoritic material in sediment.[71][72] For example, core samples from the Pacific Ocean with elevated iridium levels suggested the Eltanin impact of about 2.5 million years ago.[14]
Some of the
Cretaceous–Paleogene boundary presence

The Cretaceous–Paleogene boundary of 66 million years ago, marking the temporal border between the Cretaceous and Paleogene periods of geological time, was identified by a thin stratum of iridium-rich clay.[74] A team led by Luis Alvarez proposed in 1980 an extraterrestrial origin for this iridium, attributing it to an asteroid or comet impact.[74] Their theory, known as the Alvarez hypothesis, is now widely accepted to explain the extinction of the non-avian dinosaurs. A large buried impact crater structure with an estimated age of about 66 million years was later identified under what is now the Yucatán Peninsula (the Chicxulub crater).[75][76] Dewey M. McLean and others argue that the iridium may have been of volcanic origin instead, because Earth's core is rich in iridium, and active volcanoes such as Piton de la Fournaise, in the island of Réunion, are still releasing iridium.[77][78]
Production
| Year | Consumption (tonnes) |
Price (US$)[79] |
|---|---|---|
| 2001 | 2.6 | $415.25/ ozt ($13.351/g)
|
| 2002 | 2.5 | $294.62/ ozt ($9.472/g)
|
| 2003 | 3.3 | $93.02/ ozt ($2.991/g)
|
| 2004 | 3.60 | $185.33/ ozt ($5.958/g)
|
| 2005 | 3.86 | $169.51/ ozt ($5.450/g)
|
| 2006 | 4.08 | $349.45/ ozt ($11.235/g)
|
| 2007 | 3.70 | $444.43/ ozt ($14.289/g)
|
| 2008 | 3.10 | $448.34/ ozt ($14.414/g)
|
| 2009 | 2.52 | $420.4/ ozt ($13.52/g)
|
| 2010 | 10.40 | $642.15/ ozt ($20.646/g)
|
| 2011 | 9.36 | $1,035.87/ ozt ($33.304/g)
|
| 2012 | 5.54 | $1,066.23/ ozt ($34.280/g)
|
| 2013 | 6.16 | $826.45/ ozt ($26.571/g)
|
| 2014 | 6.1 | $556.19/ ozt ($17.882/g)
|
| 2015 | 7.81 | $544/ ozt ($17.5/g)
|
| 2016 | 7.71 | $586.90/ ozt ($18.869/g)
|
| 2017 | n.d. | $908.35/ ozt ($29.204/g)
|
| 2018 | n.d. | $1,293.27/ ozt ($41.580/g)
|
| 2019 | n.d. | $1,485.80/ ozt ($47.770/g)
|
| 2020 | n.d. | $1,633.51/ ozt ($52.519/g)
|
| 2021 | n.d. | $5,400.00/ ozt ($173.614/g)
|
| 2022 | n.d. | $3,980.00/ ozt ($127.960/g)
|
| 2023 | n.d. | $4,652.38/ ozt ($149.577/g)
|
| 2024 | n.d. | $5,000.00/ ozt ($160.754/g)
|
Worldwide production of iridium was about 7,300 kilograms (16,100 lb) in 2018.[12] The price is high and varying (see table). Illustrative factors that affect the price include oversupply of Ir crucibles[79][80] and changes in
Platinum metals occur together as dilute ores. Iridium is one of the rarer platinum metals: for every 190 tonnes of platinum obtained from ores, only 7.5 tonnes of iridium is isolated.
| Country | 2016 | 2017 | 2018 | 2019 | 2020 |
|---|---|---|---|---|---|
| World | 7,720 | 7,180 | 7,540 | 7,910 | 8,170 |
South Africa * |
6,624 | 6,057 | 6,357 | 6,464 | 6,786 |
| 598 | 619 | 586 | 845 | 836 | |
Canada * |
300 | 200 | 400 | 300 | 300 |
Russia * |
200 | 300 | 200 | 300 | 250 |
Applications
Due to iridium's resistance to corrosion it has industrial applications. The main areas of use are electrodes for producing chlorine and other corrosive products, OLEDs, crucibles, catalysts (e.g. acetic acid), and ignition tips for spark plugs.[82]
Ir metal and alloys
Resistance to heat and corrosion are the bases for several uses of iridium and its alloys.
Owing to its high melting point, hardness, and
Certain long-life aircraft engine parts are made of an iridium alloy, and an iridium–titanium alloy is used for deep-water pipes because of its corrosion resistance.[25] Iridium is used for multi-pored spinnerets, through which a plastic polymer melt is extruded to form fibers, such as rayon.[89] Osmium–iridium is used for compass bearings and for balances.[16]
Because of their resistance to arc erosion, iridium alloys are used by some manufacturers for the centre electrodes of spark plugs,[87][90] and iridium-based spark plugs are particularly used in aviation.
Catalysis
Iridium compounds are used as catalysts in the Cativa process for carbonylation of methanol to produce acetic acid.[91][92]
Iridium complexes are often active for
Medical imaging
The radioisotope
Photocatalysis and OLEDs
Iridium complexes are key components of white OLEDs. Similar complexes are used in photocatalysis.[99]
Scientific
An alloy of 90% platinum and 10% iridium was used in 1889 to construct the
Historical

Iridium–osmium alloys were used in fountain pen nib tips. The first major use of iridium was in 1834 in nibs mounted on gold.[16] Since 1944, the famous Parker 51 fountain pen was fitted with a nib tipped by a ruthenium and iridium alloy (with 3.8% iridium). The tip material in modern fountain pens is still conventionally called "iridium", although there is seldom any iridium in it; other metals such as ruthenium, osmium, and tungsten have taken its place.[102]
An iridium–platinum alloy was used for the
The pigment iridium black, which consists of very finely divided iridium, is used for painting porcelain an intense black; it was said that "all other porcelain black colors appear grey by the side of it".[104]
Precautions
This section needs to be updated. (January 2023) |
Iridium in bulk metallic form is not biologically important or hazardous to health due to its lack of reactivity with tissues; there are only about 20
A radioisotope of iridium, 192
Ir, is dangerous, like other radioactive isotopes. The only reported injuries related to iridium concern accidental exposure to radiation from 192
Ir used in
Notes
- ^ At room temperature and standard atmospheric pressure, iridium has been calculated to have a density of 22.65 g/cm3 (0.818 lb/cu in), 0.04 g/cm3 (0.0014 lb/cu in) higher than osmium measured the same way.[9] Still, the experimental X-ray crystallography value is considered to be the most accurate, and as such iridium is considered to be the second densest element.[10]
- ^ Most common oxidation states of iridium are in bold. The right column lists one representative compound for each oxidation state.
- ^ Iridium literally means "of rainbows".
- ^ The definition of the meter was changed again in 1983. The meter is currently defined as the distance traveled by light in a vacuum during a time interval of 1⁄299,792,458 of a second.
References
- ^ "Standard Atomic Weights: Iridium". CIAAW. 2017.
- ISSN 1365-3075.
- ^ ISBN 978-1-62708-155-9.
- ^ S2CID 4463905.
- ISBN 0-8493-0486-5.
- ISBN 0-8493-0464-4.
- .
- ^ "Iridium – Element information, properties and uses | Periodic Table". www.rsc.org. Retrieved 2023-10-26.
- ^ Helmenstine, Anne Marie (May 6, 2022). "What is the Densest Element on the Periodic Table?". Thoughtco.com. Retrieved October 9, 2022.
- S2CID 267570193.
- ^ "Iridium (Ir) | AMERICAN ELEMENTS". American Elements: The Materials Science Company. Retrieved 2023-11-22.
- ^ a b Singerling, Sheryl A.; Schulte, Ruth F. (August 2021). "Platinum-Group Metals". 2018 Minerals Yearbook (PDF). USGS. p. 57.11.
- PMID 11857903. Retrieved January 19, 2016.
- ^ S2CID 4362591.
- ^ OCLC 213025882.
- ^ S2CID 267552692. Archived from the originalon 2022-09-29. Retrieved 2022-09-29.
- ISBN 978-81-265-1045-0.
- S2CID 267393021. Archived from the originalon 2011-09-27. Retrieved 2008-10-02.
- ISBN 978-0-7514-0413-5.
- ISBN 9780849304712.
- S2CID 267570193. Archived from the originalon 2012-02-07. Retrieved 2008-09-17.
- ^ US 3293031A, Cresswell, Peter & Rhys, David, published 20/12/1966
- S2CID 4211238.
- .
- ^ ISBN 978-0-19-850340-8.
- ISBN 978-0-19-960563-7.
- ^ ISBN 978-1439814611.
- ISBN 978-0028657233.
- doi:10.1021/ic50060a047. Archived from the original(PDF) on 2019-04-12. Retrieved 2019-01-19.
- ^
- ^ OCLC 35279504.
- S2CID 53992437.
- ISBN 978-0-8247-8006-7.
- .
- PMID 19593837.
- OCLC 47901436.
- PMID 27268136.
- .
- .
- ^ ISBN 978-3527306732.
- .
- OCLC 224478241. Archived from the original(PDF) on 2012-11-19.
- .
- .
- PMID 21336364.
- ^ OCLC 23991202.
- ISBN 978-0-905118-83-3.
- ^ Dixon, Joshua; Brownrigg, William (1801). The literary life of William Brownrigg. To which are added an account of the coal mines near Whitehaven: And Observations on the means of preventing epidemic fevers. p. 52. Archived from the original on 24 March 2017.
- S2CID 186213277.
- ^ Marggraf, Andreas Sigismund (1760). Versuche mit dem neuen mineralischen Körper Platina del pinto genannt. Archived from the original on 24 March 2017.
- ^ Thomson, T. (1831). A System of Chemistry of Inorganic Bodies. Vol. 1. Baldwin & Cradock, London; and William Blackwood, Edinburgh. p. 693.
- ^ .
- OCLC 23991202.
- JSTOR 107152.
- OCLC 31409781.
- S2CID 121129342.
- ^ Waller, I. (1964). "The Nobel Prize in Physics 1961: presentation speech". Nobel Lectures, Physics 1942–1962. Elsevier.
- ^ "History/Origin of Chemicals". NASA. Retrieved 1 January 2013.
- S2CID 238198587.
- .
- ^ a b "Iridium" (PDF). Human Health Fact Sheet. Argonne National Laboratory. 2005. Archived from the original (PDF) on March 4, 2012. Retrieved 2008-09-20.
- .
- ^ "Cuproiridsite CuIr2S4" (PDF). Handbook of mineralogy.org. Retrieved 3 March 2022.
- ^ Vitaly A. Stepanov; Valentina E. Kungurova; Vitaly I. Gvozdev (2010). "Irasite Discovery in Copper-Nickel Ores of Shanuch Deposit (KAMCHATKA)" (PDF). New Data on Minerals. 45: 23. Retrieved 3 March 2022.
- ^ Garuti, Giorgio; Gazzotti, Moreno; Torres-Ruiz, Jose (1995). "Iridium, Rhodium, and Platinum Sulfides in Chromitites from the Ultramafic Massifs of Finero, Italy, and Ojen, Spain" (PDF). The Canadian Mineralogist. 33: 509–520. Retrieved 2 November 2022.
- ^ ISBN 978-0471238966.
- ^ .
- ^ .
- ^ .
- .
- ISBN 978-1-4613-4668-5.
- .
- S2CID 37386975.
- ^ S2CID 16017767.
- .
- OCLC 40298401.
- ISBN 978-0-8137-2307-5.
- .
- ^ a b c Platinum-Group Metals. U.S. Geological Survey Mineral Commodity Summaries
- ^ Hagelüken, C. (2006). "Markets for the catalysts metals platinum, palladium, and rhodium" (PDF). Metall. 60 (1–2): 31–42. Archived from the original (PDF) on March 4, 2009.
- ^ "Platinum 2013 Interim Review" (PDF). Platinum Today. Johnson Matthey. Retrieved 2014-01-10.
- ^ a b Ryan, Marge (2022-11-16). "Recycling and thrifting: the answer to the iridium question in electrolyser growth".
- S2CID 96640406.
- .
- S2CID 267561907. Archived from the original(PDF) on 2008-10-29. Retrieved 2008-10-01.
- ^ "Mineral Yearbook 2020 tables-only release". USGS.
- ^ .
- JSTOR 93031.
- S2CID 135705244.
- ISBN 9783527301249.
- ISBN 978-3527306732.
- .
- PMID 17672517.
- PMID 17960897.
- ISBN 978-3-642-32832-9.
- .
- ISBN 978-0-07-028121-9.
- ISBN 9781416058977.
- S2CID 96268110.
- ^ Penzes, W. B. (2001). "Time Line for the Definition of the Meter". National Institute for Standards and Technology. Retrieved 2008-09-16.
- ^ General section citations: Recalibration of the U.S. National Prototype Kilogram, R. S. Davis, Journal of Research of the National Bureau of Standards, 90, No. 4, July–August 1985 (5.5 MB PDF Archived 2017-02-01 at the Wayback Machine); and The Kilogram and Measurements of Mass and Force, Z. J. Jabbour et al., J. Res. Natl. Inst. Stand. Technol. 106, 2001, 25–46 (3.5 MB PDF)
- ^ Mottishaw, J. (1999). "Notes from the Nib Works—Where's the Iridium?". The PENnant. XIII (2).
- ^ Crookes, W., ed. (1867). "The Paris Exhibition". The Chemical News and Journal of Physical Science. XV: 182.
- ^ Pepper, J. H. (1861). The Playbook of Metals: Including Personal Narratives of Visits to Coal, Lead, Copper, and Tin Mines, with a Large Number of Interesting Experiments Relating to Alchemy and the Chemistry of the Fifty Metallic Elements. Routledge, Warne, and Routledge. p. 455.
- ISBN 9780444594532.
- ^ "Radioisotope Brief: Iridium-192 (Ir-192)" (PDF). Radiation Emergencies. Centers for Disease Control and Prevention. 2004-08-18. Retrieved 2008-09-20.
External links
- Iridium at The Periodic Table of Videos(University of Nottingham)
- Iridium in Encyclopædia Britannica
