Iron(II) chloride
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Iron(II) chloride
Iron dichloride | |||
| Other names
Ferrous chloride
Rokühnite | |||
| Identifiers | |||
| |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.028.949 | ||
| EC Number |
| ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| FeCl2 | |||
| Molar mass | 126.751 g/mol (anhydrous) 198.8102 g/mol (tetrahydrate) | ||
| Appearance | Tan solid (anhydrous) Pale green solid (di-tetrahydrate) | ||
| Density | 3.16 g/cm3 (anhydrous) 2.39 g/cm3 (dihydrate) 1.93 g/cm3 (tetrahydrate) | ||
| Melting point | 677 °C (1,251 °F; 950 K) (anhydrous) 120 °C (dihydrate) 105 °C (tetrahydrate) | ||
| Boiling point | 1,023 °C (1,873 °F; 1,296 K) (anhydrous) | ||
| 64.4 g/100 mL (10 °C), 68.5 g/100 mL (20 °C), 105.7 g/100 mL (100 °C) | |||
THF
|
Soluble | ||
| log P | −0.15 | ||
| +14750·10−6 cm3/mol | |||
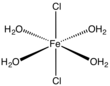
| Structure | |||
Monoclinic
| |||
| Octahedral at Fe | |||
| Pharmacology | |||
| B03AA05 (WHO) | |||
| Hazards | |||
| NFPA 704 (fire diamond) | |||
| NIOSH (US health exposure limits): | |||
REL (Recommended)
|
TWA 1 mg/m3[1] | ||
| Safety data sheet (SDS) | Iron (II) chloride MSDS | ||
| Related compounds | |||
Other anions
|
Iron(II) fluoride Iron(II) bromide Iron(II) iodide | ||
Other cations
|
Cobalt(II) chloride Manganese(II) chloride Copper(II) chloride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Iron(II) chloride, also known as ferrous chloride, is the
Production

Hydrated forms of ferrous chloride are generated by treatment of wastes from
- Fe + 2 HCl → FeCl2 + H2
The production of ferric chloride involves the use of ferrous chloride. Ferrous chloride is also a byproduct from the production of titanium, since some
Anhydrous FeCl2
Ferrous chloride is prepared by addition of iron powder to a solution of hydrochloric acid in methanol. This reaction gives the methanol solvate of the dichloride, which upon heating in a vacuum at about 160 °C converts to anhydrous FeCl2.[4] The net reaction is shown:
- Fe + 2 HCl → FeCl2 + H2
FeBr2 and FeI2 can be prepared analogously.
An alternative synthesis of anhydrous ferrous chloride is the reduction of
- 2 FeCl3 + C6H5Cl → 2 FeCl2 + C6H4Cl2 + HCl
For the preparation of ferrocene ferrous chloride is generated in situ by comproportionation of FeCl3 with iron powder in tetrahydrofuran (THF).[6] Ferric chloride decomposes to ferrous chloride at high temperatures.
Hydrates
The dihydrate, FeCl2(H2O)2, crystallizes from concentrated hydrochloric acid.

Reactions
FeCl2 and its hydrates form complexes with many ligands. For example, solutions of the hydrates react with two molar equivalents of [(C2H5)4N]Cl to give the salt [(C2H5)4N]2[FeCl4].[10]
The anhydrous FeCl2, which is soluble in THF,
Applications
Unlike the related
Natural occurrence
Lawrencite, (Fe,Ni)Cl2, is the natural counterpart, and a typically (though rarely occurring) meteoritic mineral.[14] The natural form of the dihydrate is rokühnite - a very rare mineral.[15] Related, but more complex (in particular, basic or hydrated) minerals are hibbingite, droninoite and kuliginite.
References
- ^ NIOSH Pocket Guide to Chemical Hazards. "#0346". National Institute for Occupational Safety and Health (NIOSH).
- ^ .
- ^ a b Egon Wildermuth, Hans Stark, Gabriele Friedrich, Franz Ludwig Ebenhöch, Brigitte Kühborth, Jack Silver, Rafael Rituper "Iron Compounds" in Ullmann's Encyclopedia of Industrial Chemistry. Wiley-VCH, Wienheim, 2005.
- )
- )
- .
- )
- .
- ISBN 978-0-470-13232-6.
- )
- .
- JSTOR 25046917.
- ISBN 978-0-471-93623-7.
- ^ "Lawrencite".
- ^ "Rokühnite".