Iron(III) chloride
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Iron(III) chloride
Iron trichloride | |||
Other names
| |||
| Identifiers | |||
| |||
3D model (
JSmol ) |
|||
| ChEBI | |||
| ChemSpider | |||
ECHA InfoCard
|
100.028.846 | ||
| EC Number |
| ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII |
| ||
| UN number |
| ||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| FeCl3 | |||
| Molar mass |
| ||
| Appearance | Green-black by reflected light; purple-red by transmitted light; yellow solid as hexahydrate; brown as aqueous solution | ||
| Odor | Slight HCl | ||
| Density |
| ||
| Melting point | 307.6 °C (585.7 °F; 580.8 K) (anhydrous) 37 °C (99 °F; 310 K) (hexahydrate)[1] | ||
| Boiling point |
| ||
| 912 g/L (anhydrous or hexahydrate, 25 °C)[1] | |||
| Solubility in |
| ||
| +13,450·10−6 cm3/mol[2] | |||
| Viscosity | 12 cP (40% solution) | ||
| Hazards[4][5][Note 1] | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H290, H302, H314 | |||
| P234, P260, P264, P270, P273, P280, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P390, P405, P406, P501 | |||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| NIOSH (US health exposure limits): | |||
REL (Recommended)
|
TWA 1 mg/m3[3] | ||
| Safety data sheet (SDS) | ICSC
| ||
| Related compounds | |||
Other anions
|
|||
Other cations
|
|||
Related coagulants
|
|||
| Structure | |||
Hexagonal, hR24
| |||
| R3, No. 148[7] | |||
a = 0.6065 nm, b = 0.6065 nm, c = 1.742 nm α = 90°, β = 90°, γ = 120°
| |||
Formula units (Z)
|
6 | ||
Octahedral
| |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Iron(III) chloride describes the inorganic compounds with the formula
Electronic and optical properties

All forms of ferric chloride are
Structure
Iron(III) chloride can exist as an anhydrous material and a series of hydrates, which results in distinct structures.
Anhydrous
The
Iron(III) chloride has a relatively low melting point and boils at around 315 °C. The vapor consists of the
Hydrates
Ferric chloride form
Solution

Like the solid hydrates, aqueous solutions of ferric chloride also consist of the octahedral [FeCl2(H2O)4]+ of unspecified stereochemistry.
In contrast to the complicated behavior of its aqueous solutions, solutions of iron(III) chloride in diethyl ether and tetrahydrofuran are well-behaved. Both ethers form 1:2 adducts of the general formula FeCl3(ether)2. In these complexes, the iron is pentacoordinate.[14]
Preparation
Several hundred thousand kilograms of anhydrous iron(III) chloride are produced annually. The principal method, called direct chlorination, uses scrap iron as a precursor:[10]
- 2 Fe + 3 Cl2 → 2 FeCl3
The reaction is conducted at several hundred degrees such that the product is gaseous. Using excess chlorine guarantees that the intermediate ferrous chloride is converted to the ferric state.[10] A similar but laboratory-scale process also has been described.[15][16]
Aqueous solutions of iron(III) chloride are also produced industrially from a number of iron precursors, including iron oxides:
- Fe2O3 + 12 HCl + 9 H2O → FeCl3(H2O)6
In complementary route, iron metal can be oxidized by hydrochloric acid followed by chlorination:[10]
- Fe + 2 HCl → FeCl2 + H2
- FeCl2 + 0.5 Cl2 + 6 H2O → FeCl3(H2O)6
A number of variables apply to these processes, including the oxidation of iron by ferric chloride and the hydration of intermediates.[10] Hydrates of iron(III) chloride do not readily yield anhydrous ferric chloride. Attempted thermal dehydration yields hydrochloric acid and iron oxychloride. In the laboratory, hydrated iron(III) chloride can be converted to the anhydrous form by treatment with thionyl chloride[17] or trimethylsilyl chloride:[18]
- FeCl3·6H2O + 12 (CH3)3SiCl → FeCl3 + 6 ((CH3)3Si)2O + 12 HCl
Reactions
Being
In contrast to their kinetic lability, iron(III) chlorides are thermodynamically robust, as reflected by the vigorous methods applied to their synthesis, as described above.Anhydrous FeCl3
Aside from lability, which applies to anhydrous and hydrated forms, the reactivity of anhydrous ferric chloride reveals two trends: It is a
Reactions of anhydrous iron(III) chloride reflect its description as both
- FeCl3 + Fe2O3 → 3FeOCl
Alkali metal
- FeCl3 + 3 CH3CH2ONa → "Fe(OCH2CH3)3" + 3 NaCl
Iron(III) chloride forms a 1:2
Iron(III) chloride also reacts with tetraethylammonium chloride to give the yellow salt of the tetrachloroferrate ion ((Et4N)[FeCl4]). Similarly, combining FeCl3 with NaCl and KCl gives Na[FeCl4] and K[FeCl4], respectively.[24]
In addition to these simple
In terms of its being an oxidant, iron(III) chloride oxidizes iron powder to form iron(II) chloride via a comproportionation reaction:[10]
- 2 FeCl3 + Fe → 3 FeCl2
A traditional synthesis of anhydrous
- 2 FeCl3 + C6H5Cl → 2 FeCl2 + C6H4Cl2 + HCl
iron(III) chloride releases chlorine gas when heated above 160 °C, generating
- 2FeCl3 → 2FeCl2 + Cl2
To suppress this reaction, the preparation of iron(III) chloride requires an excess of chlorinating agent, as discussed above.[16][10]
Hydrated FeCl3
Unlike the anhydrous material, hydrated ferric chloride is not a particularly strong Lewis acid since water ligands have quenched the Lewis acidity by binding to Fe(III).
Like the anhydrous material, hydrated ferric chloride is oxophilic. For example,
Aqueous iron(III) chloride serves as a one-electron oxidant illustrated by its reaction with copper(I) chloride to give copper(II) chloride and iron(II) chloride.
- FeCl3 + CuCl → FeCl2 + CuCl2
This fundamental reaction is relevant to the use of ferric chloride solutions in etching copper.
Organometallic chemistry
The interaction of anhydrous iron(III) chloride with
- 2 FeCl3 + LiCH3 → FeCl2 + Li[FeCl4] + 0.5 CH3CH3
- Li[FeCl4] + LiCH3 → Li2[FeCl4] + 0.5 CH3CH3
To a significant extent,
- 3 C5H5MgBr + FeCl3 → Fe(C5H5)2 + 1/n (C5H5)n + 3 MgBrCl
This conversion, although not of practical value, was important in the history of organometallic chemistry where ferrocene is emblematic of the field.[32]
Uses
Water treatment
In the largest application iron(III) chloride is in
Iron(III) chloride is also used to remove soluble
Etching and metal cleaning
It is also used as a leaching agent in chloride hydrometallurgy,[36] for example in the production of Si from FeSi (Silgrain process by Elkem).[37]
In another commercial application, a solution of iron(III) chloride is useful for etching copper according to the following equation:
- 2 FeCl3 + Cu → 2 FeCl2 + CuCl2
The soluble
Iron(III) chloride is used in many other hobbies involving metallic objects.[38][39][40][41][42]
Organic chemistry
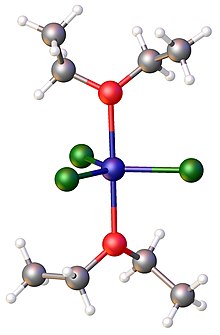
In industry, iron(III) chloride is used as a catalyst for the reaction of
- H2C=CH2 + Cl2 → ClCH2CH2Cl
Ethylene dichloride is a commodity chemical, which is mainly used for the industrial production of vinyl chloride, the monomer for making PVC.[44]
Illustrating it use as a
Organic synthesis research
Although iron(III) chlorides are seldom used in practical organic synthesis, they have received considerable attention as reagents because they are inexpensive, earth abundant, and relatively nontoxic. Many experiments probe both its redox activity and its Lewis acidity.[20] For example, iron(III) chloride oxidizes naphthols to naphthoquinones:[20][46] 3-Alkylthiophenes are polymerized to polythiophenes upon treatment with ferric chloride.[47] Iron(III) chloride has been shown to promote C-C coupling reaction.[48]
Several reagents have been developed based on
When pretreated with sodium hydride, iron(III) chloride gives a hydride reducing agent that convert alkenes and ketones into alkanes and alcohols, respectively.[51]

Histology
Iron(III) chloride is a component of useful stains, such as Carnoy's solution, a histological fixative with many applications. Also, it is used to prepare Verhoeff's stain.[52]
Natural occurrence
Like many metal halides, FeCl3 naturally occurs as a trace mineral. The rare mineral molysite is usually associated with volcanoes and fumaroles.[53][54]
FeCl3-based aerosol are produced by a reaction between iron-rich dust and hydrochloric acid from sea salt. This iron salt aerosol causes about 1-5% of naturally-occurring oxidization of methane and is thought to have a range of cooling effects; thus, it has been proposed as a catalyst for Atmospheric Methane Removal.[55]
The clouds of Venus are hypothesized to contain approximately 1% FeCl3 dissolved in sulfuric acid.[56][57]
Safety
Iron(III) chlorides are widely used in
Notes
References
- ^ ISBN 1-4398-5511-0.
- ISBN 1-4398-5511-0.
- ^ NIOSH Pocket Guide to Chemical Hazards. "#0346". National Institute for Occupational Safety and Health (NIOSH).
- ^ HSNO Chemical Classification Information Database, New Zealand Environmental Risk Management Authority, retrieved 19 Sep 2010
- ^ Various suppliers, collated by the Baylor College of Dentistry, Texas A&M University. (accessed 2010-09-19)
- ^ GHS classification – ID 831, Japanese GHS Inter-ministerial Committee, 2006, retrieved 19 Sep 2010
- ^ .
- ISBN 978-0-273-74275-3.
- ^ S2CID 105925459.
- ^ ISBN 3527306730.
- ISBN 978-0-12-352651-9.
- .
- .
- ^ .
- ISBN 9780470131626.
- ^ a b c H. Lux (1963). "Iron (III) Chloride". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 2. NY, NY: Academic Press. p. 1492.
- ISBN 9780470132593.
- ISBN 9780470132609.
- ^ ISBN 9780750633659.
- ^ ISBN 0471936235.
- ^
Kikkawa S, Kanamaru F, Koizumi M, et al. (1984). "Layered Intercalation Compounds". In Holt SL Jr (ed.). ISBN 9780470132531.
- ISBN 0306476576.
- OCLC 162129468.
- .
- ISBN 9780470132371.
- ISBN 9780582462366.
- .
- ^ .
- PMID 31031511. and references therein.
- .
- S2CID 4181383.
- .
- Akzo Nobel Base Chemicals. 2007. Archived from the original(PDF) on 13 August 2010. Retrieved 26 Oct 2007.
- ^ "Phosphorus Treatment and Removal Technologies" (PDF). Minnesota Pollution Control Agency. June 2006.
- S2CID 229396639.
- .
- .
- ^ John David Graham. "Safer Printmaking—Intaglio". University of Saskatchewan. Retrieved 5 February 2024.
- ^ Harris P, Hartman R, Hartman J (November 1, 2002). "Etching Iron Meteorites". Meteorite Times. Retrieved October 14, 2016.
- ^ Mike Lockwood, Carl Zambuto. "A message about mirror coating and recoating". Lockwood Custom Optics, Inc. Lockwood Custom Optics. Retrieved 5 February 2024.
- ^ CoinValueLookup. "Buffalo Nickel No Date Value: How Much Is It Worth Today?". CoinValueLookup. CoinValueLookup. Retrieved 5 February 2024.
- S2CID 201676001.
- ISBN 9783527306732.
- ^ "Toxic Substances – 1,2-Dichloroethane". ATSDR. Retrieved 2023-08-30.
- .
- .
- S2CID 206542971.
- PMID 34133136.
- ISBN 0471936235.
- ISBN 0471936235.
- ISBN 0471936235.
- ^ Mallory, Sheehan, Hrapchak (1990). "Verhoeff's Elastic Stain". In Carson F, Cappellano CH (eds.). Histotechnology – A Self-Instructional Text. Chicago: ASCP Press. Retrieved 2 January 2013 – via The Visible Mouse Project, U.C. Davis.
- ^ "Molysite". mindat.org. Mindat. Retrieved 5 February 2024.
- ^ "IMA list of Minerals". International Mineralogical Association. International Mineralogical Association. Retrieved 5 February 2024.
- – via esd.copernicus.org.
- S2CID 4369293.
- .
Further reading
- Lide DR, ed. (1990). ISBN 9780849304712.
- Stecher PG, Finkel MJ, Siegmund OH, eds. (1960). The Merck Index of Chemicals and Drugs (7th ed.). Rahway, New Jersey, US: Merck & Co.
- Nicholls D (1974). Complexes and First-Row Transition Elements, Macmillan Press, London, 1973. A Macmillan chemistry text. London: Macmillan Press. ISBN 9780333170885.
- Wells AF (1984). Structural Inorganic Chemistry. Oxford science publications (5th ed.). Oxford, UK: Oxford University Press. ISBN 9780198553700.
- Reich HJ, Rigby HJ, eds. (1999). Acidic and Basic Reagents. Handbook of Reagents for Organic Synthesis. New York: John Wiley & Sons, Inc. ISBN 9780471979258.