Isocyanide
An isocyanide (also called isonitrile or carbylamine) is an organic compound with the functional group –N+≡C−. It is the isomer of the related nitrile (–C≡N), hence the prefix is isocyano.[1] The organic fragment is connected to the isocyanide group through the nitrogen atom, not via the carbon. They are used as building blocks for the synthesis of other compounds.[2]
Properties
Structure and bonding
The C-N distance in isocyanides is 115.8 pm in methyl isocyanide. The C-N-C angles are near 180°.[3]
Akin to carbon monoxide, isocyanides are described by two resonance structures, one with a triple bond between the nitrogen and the carbon and one with a double bond between. The π lone pair of the nitrogen stabilizes the structure and is responsible of the linearity of isocyanides, although the reactivity of isocyanides reflects some carbene character, at least in a formal sense. Thus, both resonance structures are useful representations.[4] They are susceptible to polymerization.[4]
Spectroscopy
Isocyanides exhibit a strong absorption in their IR spectra in the range of 2165–2110 cm−1.[5]
The electronic symmetry about the isocyanide 14N nucleus results in a slow quadrupolar
Odour
Isocyanides have a very disagreeable odour. To quote from Lieke, "Es besitzt einen penetranten, höchst unangenehmen Geruch; das Oeffnen eines Gefässes mit Cyanallyl reicht hin, die Luft eines Zimmers mehrere Tage lang zu verpesten, ..." (It has a penetrating, extremely unpleasant odour; the opening of a flask of allyl [iso]cyanide is enough to foul up the air in a room for several days). Note that in Lieke's day, the difference between isocyanide and nitrile was not fully appreciated.[This quote needs a citation]
Ivar Karl Ugi states that "The development of the chemistry of isocyanides has probably suffered only little delay through the characteristic odor of volatile isonitriles, which has been described by Hofmann and Gautier as 'highly specific, almost overpowering', 'horrible', and 'extremely distressing'. It is true that many potential workers in this field have been turned away by the odour, but this is heavily outweighed by the fact that isonitriles can be detected even in traces, and that most of the routes leading to the formation of isonitriles were discovered through the odor of these compounds."[6] Isocyanides have been investigated as potential non-lethal weapons.[7]
Some isocyanides convey less offensive odours such as malt, natural rubber, creosote, cherry or old wood.
Toxicity
While some isocyanides (e.g., cyclohexyl isocyanide) are toxic, others "exhibit no appreciable toxicity for mammals". Referring to ethyl isocyanide, toxicological studies in the 1960s at Bayer showed that "oral and subcutaneous doses of 500-5000 mg/kg can be tolerated by mice".[6]
Synthesis
Many routes to isocyanides have been developed.[2]
From formamides
Commonly, isocyanides are synthesized by
- RNHC(O)H + ArSO2Cl + 2 C5H5N → RNC + [C5H5NH]+[ArSO3]− + [C5H5NH]+Cl−
The formamide precursors are, in turn, prepared from amines by formylation with formic acid or formyl acetyl anhydride[14].or from the Ritter reaction of alkenes (and other sources of carbocations) and hydrogen cyanide.[15]
From dichlorocarbene
In the
- Me3CNH2 + CHCl3 + 3 NaOH → Me3CNC + 3 NaCl + 3 H2O
As it is only effective for primary amines this reaction can be used as a chemical test for their presence.
Silver cyanide route
Of historical interest but not often of practical value, the first isocyanide,
- RI + AgCN → RNC + AgI
Other methods
Another route to isocyanides entails deprotonation of
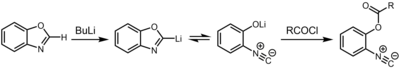
Reactions
Isocyanides have diverse reactivity.[2]
Isocyanides are stable to strong base (they are often made under strongly basic conditions), but they are sensitive to acid. In the presence of aqueous acid, isocyanides hydrolyse to the corresponding
- RNC + H2O → RNHC(O)H
This reaction is used to destroy odorous isocyanide mixtures. Some isocyanides can polymerize in the presence of Lewis and Bronsted acids.[18]
Isocyanides participate in many
Isocyanides also participate in
Isocyanides have also been shown to be a useful reagent in palladium catalysed reactions with a wide variety of compounds being formed using this method.[21]
The α position of isocyanides have substantial acidity. For example, benzyl isocyanide has a pKa of 27.4. In comparison, benzyl cyanide has a pKa of 21.9.[22] In the gas phase, CH3NC is 1.8 kcal/mol less acidic than CH3CN.[23]
Chlorination of isocyanides gives isocyanide dichlorides.
Ligands in coordination chemistry
Isocyanides form
Naturally occurring isocyanides
Only few naturally occurring compounds exhibit the isocyanide functionality. The first was discovered in 1957 in an extract of the mold
- Xanthocillin is a rare natural productthat contains two isocyanide groups.
Nomenclature
IUPAC uses the
The sometimes used old term "carbylamine" conflicts with systematic nomenclature. An amine always has three single bonds,[28] whereas an isocyanide has only one single and one multiple bond.
The isocyanamide functional group consists of a amino group attached to an isocyano moiety. for nomenclature as suffix of isonitrile or prefix of isocyano is used depending upon priority table.
References
- ^ IUPAC Goldbook isocyanides
- ^ ISSN 1463-9270.
- .
- ^ .
- ^ .
- ^ .
- PMID 19408909.
- ^ PMID 16953613.
- ^ B. E. Hoogenboom, O. H. Oldenziel, and A. M. van Leusen "Toluenesulfonylmethyl isocyanide" Organic Syntheses, Coll. Vol. 6, p.987 (1988).
- .
- .
- doi:10.1039/a708081f.
- PMID 35821692.
- .
- .
- .
- .
- .
- .
- PMID 21915395.
- PMID 23443313.
- ^ "Bordwell pKa Table (Acidity in DMSO)". www.chem.wisc.edu. Retrieved 2018-12-20.
- ISSN 0002-7863.
- ISBN 9780120311224.
- ^ Bassett, J.M.; Barker, G.K.; Green, M.; Howard, J.A.; Stone, G.A.; Wolsey, W.C. "Chemistry of low-valent metal isocyanide complexes". Journal of the Chemical Society, Dalton Transactions. 1981: 219–227.
- .
- .
- ^ IUPAC Nomenclature of Organic Compounds (Recommendations 1993)

![{\displaystyle \left[{\ce {R}}-{\overset {\oplus }{\ce {N}}}{\ce {#}}{\overset {\ominus }{\ce {C}}}{\ce {:\,<->R-{\ddot {N}}=C{:}}}\right]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/31596ad44ed69b7ad7230333e4059f48ef0d3b65)