Isomerase
In biochemistry, isomerases are a general class of enzymes that convert a molecule from one isomer to another. Isomerases facilitate intramolecular rearrangements in which bonds are broken and formed. The general form of such a reaction is as follows:
There is only one
Isomerization
Isomerases
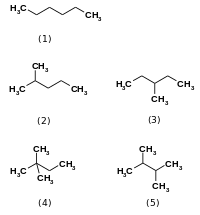
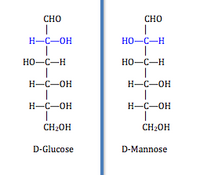
Stereoisomers have the same ordering of individual bonds and the same connectivity but the three-dimensional arrangement of bonded atoms differ. For example,
The prevalence of each isomer in nature depends in part on the
Calculating isomerase kinetics from experimental data can be more difficult than for other enzymes because the use of product inhibition experiments is impractical.[4] That is, isomerization is not an irreversible reaction since a reaction vessel will contain one substrate and one product so the typical simplified model for calculating reaction kinetics does not hold. There are also practical difficulties in determining the rate-determining step at high concentrations in a single isomerization. Instead, tracer perturbation can overcome these technical difficulties if there are two forms of the unbound enzyme. This technique uses isotope exchange to measure indirectly the interconversion of the free enzyme between its two forms. The radiolabeled substrate and product diffuse in a time-dependent manner. When the system reaches equilibrium the addition of unlabeled substrate perturbs or unbalances it. As equilibrium is established again, the radiolabeled substrate and product are tracked to determine energetic information.[5]
The earliest use of this technique elucidated the kinetics and mechanism underlying the action of phosphoglucomutase, favoring the model of indirect transfer of phosphate with one intermediate and the direct transfer of glucose.[6] This technique was then adopted to study the profile of proline racemase and its two states: the form which isomerizes L-proline and the other for D-proline. At high concentrations it was shown that the transition state in this interconversion is rate-limiting and that these enzyme forms may differ just in the protonation at the acidic and basic groups of the active site.[5]
Nomenclature
Generally, "the names of isomerases are formed as "substrate isomerase" (for example, enoyl CoA isomerase), or as "substrate type of isomerase" (for example, phosphoglucomutase)."[7]
Classification
Enzyme-catalyzed reactions each have a uniquely assigned classification number. Isomerase-catalyzed reactions have their own EC category: EC 5.[8] Isomerases are further classified into six subclasses:
Racemases, epimerases
This category (EC 5.1) includes (
| EC number | Description | Examples |
|---|---|---|
| EC 5.1.1 | Acting on Amino Acids and Derivative | alanine racemase, methionine racemase |
| EC 5.1.2 | Acting on Hydroxy Acids and Derivatives | lactate racemase, tartrate epimerase |
| EC 5.1.3 | Acting on Carbohydrates and Derivatives | ribulose-phosphate 3-epimerase, UDP-glucose 4-epimerase
|
| EC 5.1.99 | Acting on Other Compounds | methylmalonyl CoA epimerase, hydantoin racemase |
Cis-trans isomerases
This category (EC 5.2) includes enzymes that catalyze the isomerization of
This category is not broken down any further. All entries presently include:

Intramolecular oxidoreductases
This category (EC 5.3) includes intramolecular oxidoreductases. These isomerases catalyze the transfer of electrons from one part of the molecule to another. In other words, they catalyze the oxidation of one part of the molecule and the concurrent reduction of another part.[8] Sub-categories of this class are:

| EC number | Description | Examples |
|---|---|---|
| EC 5.3.1 | Interconverting Aldoses and Ketoses | Triose-phosphate isomerase, Ribose-5-phosphate isomerase
|
| EC 5.3.2 | Interconverting Keto- and Enol-Groups | Phenylpyruvate tautomerase, Oxaloacetate tautomerase |
| EC 5.3.3 | Transposing C=C Double Bonds | Steroid Delta-isomerase, L-dopachrome isomerase |
| EC 5.3.4 | Transposing S-S Bonds | Protein disulfide-isomerase |
| EC 5.3.99 | Other Intramolecular Oxidoreductases | Allene-oxide cyclase
|
Intramolecular transferases
This category (EC 5.4) includes intramolecular transferases (mutases). These isomerases catalyze the transfer of functional groups from one part of a molecule to another.[8] Phosphotransferases (EC 5.4.2) were categorized as transferases (EC 2.7.5) with regeneration of donors until 1983.[9] This sub-class can be broken down according to the functional group the enzyme transfers:

| EC number | Description | Examples |
|---|---|---|
| EC 5.4.1 | Transferring Acyl Groups | Lysolecithin acylmutase, Precorrin-8X methylmutase |
| EC 5.4.2 | Phosphotransferases (Phosphomutases) | Phosphoglucomutase, Phosphopentomutase |
| EC 5.4.3 | Transferring Amino Groups | Beta-lysine 5,6-aminomutase, Tyrosine 2,3-aminomutase |
| EC 5.4.4 | Transferring hydroxy groups | (hydroxyamino)benzene mutase, Isochorismate synthase |
| EC 5.4.99 | Transferring Other Groups | Methylaspartate mutase, Chorismate mutase |
Intramolecular lyases
This category (EC 5.5) includes intramolecular lyases. These enzymes catalyze "reactions in which a group can be regarded as eliminated from one part of a molecule, leaving a double bond, while remaining covalently attached to the molecule."[8] Some of these catalyzed reactions involve the breaking of a ring structure.
This category is not broken down any further. All entries presently include:

Mechanisms of isomerases
Ring expansion and contraction via tautomers

A classic example of ring opening and contraction is the isomerization of glucose (an aldehyde with a six-membered ring) to fructose (a ketone with a five-membered ring). The conversion of D-glucose-6-phosphate to D-fructose-6-phosphate is catalyzed by glucose-6-phosphate isomerase, an intramolecular oxidoreductase. The overall reaction involves the opening of the ring to form an aldose via acid/base catalysis and the subsequent formation of a cis-endiol intermediate. A ketose is then formed and the ring is closed again.
Glucose-6-phosphate first binds to the
Epimerization

An example of epimerization is found in the Calvin cycle when D-ribulose-5-phosphate is converted into D-xylulose-5-phosphate by
Intramolecular transfer

Chorismate mutase is an intramolecular transferase and it catalyzes the conversion of chorismate to prephenate, used as a precursor for L-tyrosine and L-phenylalanine in some plants and bacteria. This reaction is a Claisen rearrangement that can proceed with or without the isomerase, though the rate increases 106 fold in the presence of chorismate mutase. The reaction goes through a chair transition state with the substrate in a trans-diaxial position.[12] Experimental evidence indicates that the isomerase selectively binds the chair transition state, though the exact mechanism of catalysis is not known. It is thought that this binding stabilizes the transition state through electrostatic effects, accounting for the dramatic increase in the reaction rate in the presence of the mutase or upon addition of a specifically-placed cation in the active site.[13]
Intramolecular oxidoreduction

The role of isomerase in human disease
Isomerase plays a role in human disease. Deficiencies of this enzyme can cause disorders in humans.
Phosphohexose isomerase deficiency
Phosphohexose Isomerase Deficiency (PHI) is also known as phosphoglucose isomerase deficiency or Glucose-6-phosphate isomerase deficiency, and is a hereditary enzyme deficiency. PHI is the second most frequent erthoenzyopathy in glycolysis besides pyruvate kinase deficiency, and is associated with non-spherocytic haemolytic anaemia of variable severity.[15][16] This disease is centered on the glucose-6-phosphate protein. This protein can be found in the secretion of some cancer cells.[17] PHI is the result of a dimeric enzyme that catalyses the reversible interconversion of fructose-6-phosphate and gluose-6-phosphate.[15]
PHI is a very rare disease with only 50 cases reported in literature to date.[15]
Diagnosis is made on the basis of the clinical picture in association with biochemical studies revealing erythrocyte GPI deficiency (between 7 and 60% of normal) and identification of a mutation in the GPI gene by molecular analysis.[15]
The deficiency of phosphohexose isomerase can lead to a condition referred to as hemolytic syndrome. As in humans, the hemolytic syndrome, which is characterized by a diminished erythrocyte number, lower hematocrit, lower hemoglobin, higher number of reticulocytes and plasma bilirubin concentration, as well as increased liver- and spleen-somatic indices, was exclusively manifested in homozygous mutants.[16]
Triosephosphate isomerase deficiency
The disease referred to as triosephosphate isomerase deficiency (TPI), is a severe autosomal recessive inherited multisystem disorder of glycolytic metabolism.[18] It is characterized by hemolytic anemia and neurodegeneration, and is caused by anaerobic metabolic dysfunction. This dysfunction results from a missense mutation that effects the encoded TPI protein.[19] The most common mutation is the substitution of gene, Glu104Asp, which produces the most severe phenotype, and is responsible for approximately 80% of clinical TPI deficiency.[18]
TPI deficiency is very rare with less than 50 cases reported in literature.[20] Being an autosomal recessive inherited disease, TPI deficiency has a 25% recurrence risk in the case of heterozygous parents.[18][20] It is a congenital disease that most often occurs with hemolytic anemia and manifests with jaundice.[18] Most patients with TPI for Glu104Asp mutation or heterozygous for a TPI null allele and Glu104Asp have a life expectancy of infancy to early childhood. TPI patients with other mutations generally show longer life expectancy. To date, there are only two cases of individuals with TPI living beyond the age of 6. These cases involve two brothers from Hungary, one who did not develop neurological symptoms until the age of 12, and the older brother who has no neurological symptoms and suffers from anemia only.[21]
Individuals with TPI show obvious symptoms after 6–24 months of age. These symptoms include: dystonia, tremor, dyskinesia, pyramidal tract signs, cardiomyopathy and spinal motor neuron involvement.[18] Patients also show frequent respiratory system bacterial infections.[18]
TPI is detected through deficiency of enzymatic activity and the build-up of dihyroxyacetone phosphate(DHAP), which is a toxic substrate, in erythrocytes.[18][20] This can be detected through physical examination and a series of lab work. In detection, there is generally myopathic changes seen in muscles and chronic axonal neuropathy found in the nerves.[18] Diagnosis of TPI can be confirmed through molecular genetics.[18] Chorionic villus DNA analysis or analysis of fetal red cells can be used to detect TPI in antenatal diagnosis.[18]
Treatment for TPI is not specific, but varies according to different cases. Because of the range of symptoms TPI causes, a team of specialist may be needed to provide treatment to a single individual. That team of specialists would consists of pediatricians, cardiologists, neurologists, and other healthcare professionals, that can develop a comprehensive plan of action.[22]
Supportive measures such as red cell transfusions in cases of severe anaemia can be taken to treat TPI as well. In some cases, spleen removal (splenectomy) may improve the anaemia. There is no treatment to prevent progressive neurological impairment of any other non-haematological clinical manifestation of the diseases.[23]
Industrial applications
By far the most common use of isomerases in industrial applications is in sugar manufacturing. Glucose isomerase (also known as xylose isomerase) catalyzes the conversion of D-xylose and D-glucose to D-xylulose and D-fructose. Like most sugar isomerases, glucose isomerase catalyzes the interconversion of aldoses and ketoses.[24]
The conversion of glucose to fructose is a key component of
The isomerization of xylose to xylulose has its own commercial applications as interest in biofuels has increased. This reaction is often seen naturally in bacteria that feed on decaying plant matter. Its most common industrial use is in the production of ethanol, achieved by the fermentation of xylulose. The use of hemicellulose as source material is very common. Hemicellulose contains xylan, which itself is composed of xylose in β(1,4) linkages.[27] The use of glucose isomerase very efficiently converts xylose to xylulose, which can then be acted upon by fermenting yeast. Overall, extensive research in genetic engineering has been invested into optimizing glucose isomerase and facilitating its recovery from industrial applications for re-use.
Glucose isomerase is able to catalyze the isomerization of a range of other sugars, including D-
Membrane-associated isomerases
Some isomerases associate with
References
- ISBN 9780323144605.
- ^ ISBN 978-0-9678550-9-7.
- ISBN 978-0-7637-2197-8.
- ISBN 978-3-527-66548-8.
- ^ PMID 3521737.
- PMID 5726186.
- ISBN 978-0-321-59695-6.
- ^ ISBN 978-0-12-227164-9.
- ^ The Enzyme List Class 5 - Isomerases (PDF). Nomenclature Committee of the International Union of Biochemistry and Molecular Biology (NC-IUBMB). 2010.
- PMID 15342241.
- PMID 3987693.
- ISBN 978-1-118-34896-3.
- PMID 10960481.
- PMID 17250851.
- ^ PMID 10916680.
- ^ PMID 8417789.
- S2CID 34123426.
- ^ a b c d e f g h i j Orosz PF. "Triose phosphate-isomerase deficiency". Orphanet. Retrieved 14 November 2013.
- PMID 16980388.
- ^ PMID 12023819. Archived from the original(PDF) on 2013-12-03. Retrieved 2013-11-27.
- S2CID 3110178.
- ^ "Triosephosphate Isomerase Deficiency". NORD. Retrieved 14 December 2013.
- ^ "Triose phosphate isomerase deficiency -TPI" (PDF). Retrieved 26 November 2013.
- ^ PMID 8801434.
- ^ Baker S (1976). "Pure fructose syrups". Process Biochemistry. 11: 20–25.
- ^ Antrim RL, Colilla W, Schnyder BJ (1979). "Glucose isomerase production of high fructose syrups". Applied Biochemistry and Bioengineering. 2: 97–155.
- PMID 6446306.
- ^ Chen WP (August–September 1980). "Glucose isomerase". Process Biochemistry. 15: 36–41.
- ^ Superfamilies of single-pass transmembrane lyases in Membranome database

![{\displaystyle {\ce {A-B}}\quad {\xrightarrow[{\text{ isomerase }}]{}}\quad {\ce {B-A}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/3e7114e4d3961ea355ca13869f9ea72fc1b38e08)