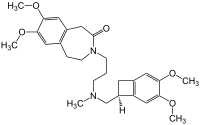
Ivabradine
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ɪˈvæbrədiːn/ |
| Trade names | Corlanor, Procoralan, others |
| Other names | S-16257 |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 40% |
| Protein binding | 70% |
| Metabolism | Liver (first-pass) >50%, CYP3A4-mediated |
| Elimination half-life | 2 hours |
| Excretion | Kidney and fecal |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Ivabradine, sold under the brand name Procoralan among others, is a
Ivabradine acts by allowing negative chronotropy in the sinoatrial structure, thus reducing the heart rate via specific inhibition of the pacemaker current. It operates by a mechanism different from that of beta blockers and calcium channel blockers, which are two commonly prescribed antianginal classes of cardiac drugs. Ivabradine has no apparent inotropic properties and may be a cardiotonic agent.
Medical uses
It is used for the symptomatic treatment of chronic stable
Chest pain
Ivabradine is as effective as the beta blocker atenolol and comparable with amlodipine in the management of chronic stable angina, as demonstrated by improvements in total exercise duration in non-inferiority trials, hence it can be an alternative therapy for those who cannot tolerate conventional therapies.[5][6] In people not sufficiently managed with beta blockers for their angina, adding ivabradine can further reduce heart rate and improve total exercise duration.[7]
Heart failure
It is used in combination with beta blockers in people with heart failure with LVEF lower than 35 percent inadequately controlled by beta blockers alone and whose heart rate exceeds 70 beats per minute.[8] In people not sufficiently managed with beta blockers for their heart failure adding ivabradine decreases the risk of hospitalization for heart failure.[2]
Tachycardia
The clinical use of ivabradine is predicated on its mechanism of action on sinoatrial nodal tissue where it selectively inhibits the funny current (If) and results in a decrease in heart rate.[9]
Ivabradine’s most frequent application in electrophysiology is for the treatment of inappropriate sinus tachycardia. Its use for inappropriate sinus tachycardia is not a European Medicines Agency or Food and Drug Administration approved indication for ivabradine.[9]
It has been used experimentally for the treatment of postural orthostatic tachycardia syndrome in patients with long COVID.[10]
Contraindications
Ivabradine is contraindicated in
Use of ivabradine with verapamil or diltiazem is contraindicated.[12]
Adverse effects
Overall, 14.5% of patients taking ivabradine experience luminous phenomena (by patients described as sensations of enhanced brightness in a fully maintained visual field). This is probably due to blockage of Ih ion channels in the retina, which are very similar to cardiac If. These symptoms are mild, transient, and fully reversible. In clinical studies, about 1% of all patients had to discontinue the drug because of these sensations, which occurred on average 40 days after the drug was started.[5]
In a large clinical trial, bradycardia (unusually slow heart rate) occurred in 2% and 5% of patients taking ivabradine at doses of 7.5 and 10 mg respectively (compared to 4.3% in those taking atenolol).[5] Headaches were reported in 2.6 to 4.8 percent of cases.[5] Other common adverse drug reactions (1–10% of patients) include first-degree AV block, ventricular extrasystoles, dizziness and/or blurred vision.[13]
Mechanism of action
Ivabradine acts on the If (f is for "
Ivabradine binds to HCN4 receptors (potassium/sodium hyperpolarization-activated cyclic nucleotide-gated channel 4), utilizing Y506, F509 and I510 residues.[16]
Clinical trials
Coronary artery disease
The BEAUTIFUL study randomised over 10917 patients having stable coronary artery disease and left ventricle dysfunction (ejection fraction < 40%). Ivabradine did not show a significant reduction in the primary composite endpoint of cardiovascular death, admission to hospital for acute myocardial infarction, and admission to hospital for new onset or worsening heart failure. However, in a prespecified subgroup of patients with a baseline heart rate of more than 70 bpm, ivabradine significantly reduced the following secondary endpoints:[17]
- Coronary events by 22% (P=0.023)
- Fatal and nonfatal myocardial infarction by 36% (P=0.001)
- Coronary revascularization by 30% (P=0.016).
These results were seen in combination therapy with beta blockers, and were found to be safe and effective in improving coronary artery disease outcomes in patients with heart rates of 70 bpm or more.[18]
The SIGNIFY trial randomised 19102 patients with stable coronary artery disease and an elevated heart rate greater than 70 beats per minute were assigned to an intervention of ivabradine or placebo in addition to standard therapy. Ivabradine did not significantly improve the secondary outcomes in patient groups, however did demonstrate a reduction in heart rate. When compared to the SHIFT study, a reduction in cardiovascular death or hospital admission was also demonstrated and hence should be considered when additional therapy is in question.[19][20]
Chronic heart failure
In the SHIFT study, ivabradine significantly reduced the risk of the primary composite endpoint of hospitalization for worsening heart failure or cardiovascular death by 18% (P<0.0001) compared with placebo on top of optimal therapy.[21] These benefits were observed after 3 months of treatment. SHIFT also showed that administration of ivabradine to heart failure patients significantly reduced the risk of death from heart failure by 26% (P=0.014) and hospitalization for heart failure by 26% (P<0.0001). The improvements in outcomes were observed throughout all prespecified subgroups: female and male, with or without beta-blockers at randomization, patients below and over 65 years of age, with heart failure of ischemic or non-ischemic etiology, NYHA class II or class III, IV, with or without diabetes, and with or without hypertension.[22] A 2020 Cochrane review found no difference in cardiovascular mortality and serious adverse events between long-term treatment with ivabradine and placebo/usual care/no treatment in participants with heart failure with a reduced ejection fraction.[23]
A note of caution must be emphasised. Ivabradine, though indicated for chronic heart failure in patients who are clinically stable, is not indicated in acute heart failure where the enhanced heart rate represents cardiac reserve. Indiscriminate use of Ivabradine could destabilise these patients.
Society and culture
Approval
Ivabradine was approved by the European Medicines Agency in 2005, and by the United States Food and Drug Administration in 2015.[24]
Names
It is marketed by Amgen under the trade name Corlanor in the United States,[25] and by Servier in the rest of the world under the trade names Procoralan (worldwide), Coralan (in Hong Kong, Singapore, Australia and some other countries), Corlentor (in Armenia, Spain, Italy and Romania), Lancora (in Canada) and Coraxan (in Russia and Serbia). It is also marketed in India under the brand names Ivabrad, Ivabid. In Iran it's sold under the trademark of "bradix" . IVAMAC and Bradia. During its development, ivabradine was known as S-16257.
References
- ^ "Health Canada New Drug Authorizations: 2016 Highlights". Health Canada. 14 March 2017. Retrieved 7 April 2024.
- ^ PMID 27208050.
- S2CID 25305128.
- PMID 34414903.)
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link - ^ PMID 16214830.
- S2CID 25325838.
- PMID 19136486.
- PMID 22611136.
- ^ a b "The Clinical Use of Ivabradine". American College of Cardiology. Retrieved 2022-02-03.
- PMID 34127505.
- ^ "European Medicine Agency, Procoralan Summary of Product Characteristics" (PDF). Archived from the original (PDF) on 2012-05-19. Retrieved 2010-09-13.
- ^ "Press release: European Medicines Agency recommends measures to reduce risk of heart problems with Corlentor/Procoralan (ivabradine)". European Medicines Agency. 21 November 2014. Archived from the original on 21 June 2018. Retrieved 15 December 2014.
- ^ Anonymous (2006). "New medicines: Procoralan". Pharmaceutical Journal. 276 (7386): 131. Archived from the original on 2008-03-17. Retrieved 2007-10-14.
- PMID 8032660.
- PMID 16451297.
- PMID 23308150.
- S2CID 26282333.
- S2CID 249941448.
- S2CID 19002896.
- S2CID 21196422.
- ^ Stiles S. "SHIFT: Adding HR-slowing agent ivabradine to HF meds cuts mortality, hospitalization". TheHeart.org. Retrieved 1 April 2011.
- S2CID 21196422.
- PMID 33147368.
- ^ "FDA approves Corlanor to treat heart failure". www.fda.gov. Archived from the original on 2015-04-16. Retrieved 2015-04-16.
- ^ "Amgen and Servier announce product collaboration". www.servier.com. Archived from the original on 2016-03-04. Retrieved 2015-12-29.
