JWH-193
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
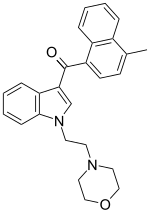
JWH-193 is a drug from the
naphthoylindole families which acts as a cannabinoid receptor agonist. It was invented by the pharmaceutical company Sanofi-Winthrop in the early 1990s. JWH-193 has a binding affinity at the CB1 receptor of 6 nM, binding around seven times more tightly than the parent compound JWH-200,[1]
though with closer to twice the potency of JWH-200 in activity tests.
In the United States, all CB1 receptor agonists of the 3-(1-naphthoyl)indole class such as JWH-193 are
Schedule I Controlled Substances.[2]
Related compounds
A structural isomer of JWH-193 with the methyl group on the indole ring instead of the naphthoyl ring, was also found to be of similarly increased potency over JWH-200.[3][4]

