Jelly roll fold
The jelly roll or Swiss roll fold is a
Structure
The basic jelly roll structure consists of eight
Viral proteins

A large number of
Single jelly roll capsid proteins
Single jelly roll capsid (JRC) proteins are found in at least sixteen distinct viral
Another group of viruses uses single jelly roll proteins in their capsids, but in the vertical rather than horizontal orientation. These viruses are evolutionarily related to the large group of double jelly-roll viruses known as the
Double jelly roll proteins
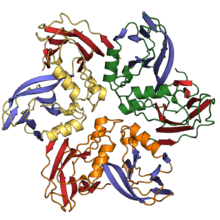
Double jelly roll capsid proteins consist of two single jelly roll folds connected by a short linker region. They are found in both
Double jelly roll proteins are believed to have evolved from single jelly roll proteins by
Initially, it was believed that double jelly roll proteins are unique to viruses, because they were not observed in cellular proteins.[11] However, in 2022, comparison of protein structures revealed several families of bona fide cellular proteins with the double jelly roll fold [25]
Non-capsid proteins
Single jelly rolls also occur in non-capsid viral proteins, including minor components of the assembled
Cellular proteins
Both single and double jelly roll folds are found in proteins of cellular origin.
More broadly, the members of the extremely diverse cupin superfamily are also often described as jelly rolls; though the common core of the cupin domain structure contains only six beta strands, many cupins have eight.[32] Examples include the non-heme dioxygenase enzymes[33][34] (including alpha-ketoglutarate-dependent hydroxylases) and JmjC-family histone demethylases.[35][36]
Cellular proteins with the double jelly roll fold include glycoside hydrolases of the DUF2961 family, peptide:N-glycosidase F (PNGases F) and peptidylglycine alpha-amidating monooxygenase.[25]

A notable difference between PNGases F and the other double jelly roll proteins is the absence of the α-helices, which follow the F and F' strands in capsid proteins and DUF2961. The equivalent regions are variable in the PNGases F and contain either long loops or insertions. By contrast, jelly-roll domains of DUF2961 proteins contain an insertion of short β-hairpins upstream of the G and G' strands of the double jelly roll fold. Importantly, DUF2961 family proteins form trimers resembling viral capsomers.[25]
Evolution
Comparative studies of proteins classified as jelly roll and
History and nomenclature
The name "jelly roll" was first used for the structure composed of an elaboration on the
References
- ^ PMID 25195746.
- ^ PMID 7020376.
- ^ PMID 1447783.
- ^ PMID 23408879.
- ^ PMID 24244135.
- S2CID 4341051.
- PMID 6854630.
- ^ PMID 15574324.
- ^ S2CID 2767388.
- PMID 21450811.
- ^ PMID 22440622.
- ^ PMID 28265094.
- PMID 23850154.
- ^ PMID 26320579.
- ^ PMID 30862777.
- ^ .
- ^ PMID 18775333.
- PMID 28716906.
- ^ S2CID 31542714.
- PMID 32132243.
- S2CID 221182789.
- PMID 29636073.
- PMID 21742267.
- PMID 23812617.
- ^ PMID 35078938.
- PMID 37319262.
- PMID 11684019.
- PMID 25813344.
- PMID 11796220.
- PMID 14725774.
- S2CID 39174409.
- PMID 11264412.
- PMID 17301803.
- ^ PMID 23142576.
- S2CID 15273763.
- S2CID 2616900.
External links
- Antiparallel β Domains, a section from Anatomy and Taxonomy of Protein Structure by Jane S. Richardson
- The Jelly Roll of Life by Jacqueline Humphries at Small Things Considered, a blog sponsored by the American Society for Microbiology
