Keratan sulfate
Keratan sulfate (KS), also called keratosulfate, is any of several
Structure
Like other
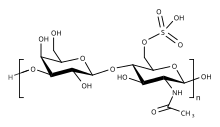
The basic repeating disaccharide unit within keratan sulfate is -3
- A linkage region, at one end of which the KS chain is linked to the core protein.
- A repeat region, composed of the -3Galβ1-4GlcNAcβ1- repeating disaccharide unit and
- A chain capping region, occurring at the opposite end of the KS chain to the protein linkage region.
The monosaccharide mannose is found within the linkage region of keratan sulfate type I (KSI). Disaccharides within the repeating region of KSII may be fucosylated and N-Acetylneuraminic acid caps the end of all keratan sulfate type II (KSII) chains and up to 70% of KSI type chains.[4]
KS classes
The designations KSI and KSII were originally assigned on the basis of the tissue type from which the keratan sulfate was isolated. KSI was isolated from
Corneal KSI
The amount of KS found in the cornea is 10 fold higher than it is in cartilage and 2-4 times higher than it is in other tissues.[10] It is produced by corneal keratocytes[11] and is thought to play a role of a dynamic buffer of corneal hydration. In a rare progressive disorder called macular corneal dystrophy (MCDC), the synthesis of keratan sulfate is either absent (MCDC type I) or abnormal (MCDC type II).[12]
Non-corneal KSI
KSII
See also
- N-Acetylglucosamine
- Galactose
- Lumican
- Morquio syndrome is marked by the accumulation of KS
- Proteoglycan 4
References
External links
- Chondroitin and keratin sulfate at the Duke University Health System's Orthopedics program
- Keratan+sulfate at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
