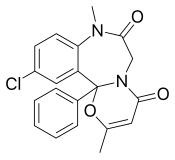
Ketazolam
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic |
| Elimination half-life | 26–200 hours |
| Excretion | Renal |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Ketazolam (marketed under the brand names Anseren, Ansieten, Ansietil, Marcen, Sedatival, Sedotime, Solatran and Unakalm) is a drug which is a
Therapeutic uses
It is used for the treatment of anxiety and has similar effectiveness compared to diazepam. Ketazolam also appears to produce reduced levels of side effects such as sedation compared with diazepam and the side effects when they occur tend to be milder.[3][4][5][6] Ketazolam is also an effective antispasmodic drug and is used for the treatment of spasticity.[7][8][9]
Availability
Ketazolam is not approved for sale in
Tolerance and physical dependence
Chronic use of ketazolam as with other
Contraindications and special caution
Benzodiazepines require special precaution if used in the elderly, during pregnancy, in children, alcohol or drug-dependent individuals and individuals with
Pharmacokinetics
Ketazolam breaks down in the blood to
Warnings
The U.S. Food and Drug Administration warns that in Spain, ketazolam marketed as Marcen may sometimes be mistakenly confused with Narcan.[16]
Legal status
Ketazolam is a List 3 drug under the Betäubungsmittelgesetz, like almost all benzodiazepines in Germany. Ketazolam is a List II drugs of the Opium Law in the Netherlands. Ketazolam is a Schedule IV drug under the Controlled Substances Act in the US.[17]
References
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-16.
- PMID 11443263.
- PMID 6108319.
- PMID 6105920.
- S2CID 26311555.
- S2CID 6597368.
- PMID 2874780.
- S2CID 45292155.
- PMID 6149738.
- ^ "Benzodiazepine Names". The Tranquilliser Recovery and Awareness Place. Archived from the original on 8 December 2008. Retrieved 26 August 2008.
- ^ "SOLATRAN 15 (capsules) - SOLATRAN 30 (capsules)". South African Electronic Package Inserts. 12 November 1985. Archived from the original on 2 June 2019. Retrieved 26 August 2008.
- ^ "Controlled Drugs and Substances Act". laws-lois.justice.gc.ca. Archived from the original on 2013-12-15.
- S2CID 38037200.
- PMID 19900604.
- PMID 6743768.
- ^ Food and Drug Administration (January 2006). "FDA Public Health Advisory - Consumers Filling U.S. Prescriptions Abroad May Get the Wrong Active Ingredient Because of Confusing Drug Names". FDA (USA). Archived from the original on 12 August 2006. Retrieved 26 August 2006.
- ^ Drug Enforcement Administration (USA). "Drug Scheduling". United States Government. Archived from the original on 27 May 2018. Retrieved 26 August 2008.
