Ketoconazole
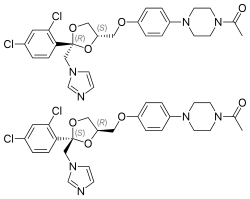 (2R,4S)-(+)-ketoconazole (top) (2S,4R)-(−)-ketoconazole (bottom) | |
 Ball-and-stick model of (2R,4S)-(+)-ketoconazole | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌkiːtoʊˈkoʊnəˌzoʊl, -zɒl/[1][2] |
| Trade names | Nizoral, others |
| Other names | R-41400; KW-1414 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682816 |
| License data |
|
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | By mouth: 37–97%[8] |
| Protein binding | 84 to 99% |
| Metabolism | Extensive liver (predominantly oxidation, O-dealkylation) |
| Metabolites | N-deacetyl ketoconazole |
| Elimination half-life | Biphasic |
| Excretion | Bile duct (major) and kidney[9] |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture[9][10] |
| |
| |
| | |


Ketoconazole, sold under the brand name Nizoral among others, is an
Common
Ketoconazole was patented in 1977 by Belgian pharmaceutical company
Medical uses
Topical antifungal
Topically administered ketoconazole is usually prescribed for fungal infections of the skin and mucous membranes, such as
Systemic antifungal
Ketoconazole has activity against many kinds of fungi that may cause human disease, such as
Ketoconazole is used orally in dosages of 200 to 400 mg per day in the treatment of superficial and deep fungal infections.[25]
Off-label uses
Hair loss

Ketoconazole shampoo in conjunction with an oral
Limited clinical studies suggest ketoconazole shampoo used either alone[27][28] or in combination with other treatments[29] may be useful in reducing hair loss in some cases.[30]
Hormonal
The side effects of ketoconazole are sometimes harnessed in the treatment of non-fungal conditions. While ketoconazole blocks the synthesis of the sterol
Ketoconazole has been used to prevent the
Contraindications
Oral ketoconazole has various contraindications, such as concomitant use with certain other drugs due to known drug interactions.[6] Other contraindications of oral ketoconazole include liver disease, adrenal insufficiency, and known hypersensitivity to oral ketoconazole.[6]
Side effects
Gastrointestinal
Vomiting, diarrhea, nausea, constipation, abdominal pain, upper abdominal pain, dry mouth,
Endocrine
The drug may cause adrenal insufficiency so the level of the adrenocortical hormones should be monitored while taking it.[13][36] Oral ketoconazole at a dosage range of 400 to 2,000 mg/day has been found to result in a rate of gynecomastia of 21%.[37]
Liver
In July 2013, the US Food and Drug Administration (FDA) issued a warning that taking ketoconazole by mouth can cause severe liver injuries and adrenal gland problems: adrenal insufficiency and worsening of other related to the gland conditions.[13] It recommends oral tablets should not be a first-line treatment for any fungal infection. It should be used for the treatment of certain fungal infections, known as endemic mycoses, only when alternative antifungal therapies are not available or tolerated.[13] As contraindication it should not be used in people with acute or chronic liver disease.[13]
Hypersensitivity
Anaphylaxis after the first dose may occur.[medical citation needed] Other cases of hypersensitivity include urticaria.[11][6]
Topical formulations
The topical formulations have not been associated with liver damage, adrenal problems, or drug interactions. These formulations include creams, shampoos, foams, and gels applied to the skin, unlike the ketoconazole tablets, which are taken by mouth.[13]
Pregnancy
Ketoconazole is categorized as
Overdose
In the event of an
Interactions
The concomitant use of the following medications is contraindicated with ketoconazole tablets:[6][36]
- methadone, disopyramide, dronedarone
- irinotecan, lurasidone, colchicine
- alprazolam, oral midazolam, oral triazolam
- felodipine, ranolazine, tolvaptan, eplerenone
- HMG-CoA reductase inhibitors: lovastatin, simvastatin
- Others: cisapride, nisoldipine, dofetilide, pimozide
And is not recommended:[6][36]
- carbamazepine, phenytoin
- gastric acid suppressants: proton pump inhibitors
- sucralfate
- efavirenz, nevirapine
Ritonavir is known for increasing activity of the ketoconazole so it is recommended to reduce dosage.[6]
There is also a list of drugs which significantly decrease systemic exposure to the ketoconazole and drugs whose systemic exposure is increased by the ketoconazole.[6][36]
Pharmacology
Pharmacodynamics
Antifungal activity
This section needs additional citations for verification. (May 2013) |
As an antifungal, ketoconazole is structurally similar to
Resistance to ketoconazole has been observed in a number of clinical fungal isolates, including Candida albicans. Experimentally, resistance usually arises as a result of mutations in the sterol biosynthesis pathway. Defects in the sterol 5-6 desaturase enzyme reduce the toxic effects of azole inhibition of the 14-alpha demethylation step.
Antihormonal activity
As an
Ketoconazole, along with miconazole, has been found to act as an antagonist of the glucocorticoid receptor.[45][46]
Ketoconazole is a
Oral ketoconazole has been used clinically as a steroidogenesis inhibitor in men, women, and children at dosages of 200 to 1,200 mg/day.
Ketoconazole has been found to displace dihydrotestosterone and estradiol from sex hormone-binding globulin in vitro, but this was not found to be relevant in vivo.[48]
Other activities
Ketoconazole has been found to inhibit the activity of the cation channel TRPM5.[55]
Pharmacokinetics
When administered orally, ketoconazole is best absorbed at highly acidic levels, so antacids or other causes of decreased stomach acid levels will lower the drug's absorption. Absorption can be increased by taking it with an acidic beverage, such as cola.[56] Ketoconazole is very lipophilic and tends to accumulate in fatty tissues.
Chemistry
Ketoconazole is a
History
Ketoconazole was discovered in 1976 at
Due to incidence of serious
Ketoconazole HRA was approved for use in the European Union for treatment of Cushing's syndrome in November 2013.[7][63]
Society and culture
Generic names
Ketoconazole is the
Brand names
Ketoconazole has been marketed under a large number of brand names.[57][58][64][65]
Availability
Ketoconazole is available widely throughout the world.[58][65]
In 2013, the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) recommended that a ban be imposed on the use of oral ketoconazole for systemic use in humans throughout the European Union, after concluding that the risk of serious liver injury from systemic ketoconazole outweighs its benefits.[66]
Research
As of March 2019, oral levoketoconazole (developmental code name COR-003, tentative brand name Recorlev) is phase III clinical trials for the treatment of Cushing's syndrome.[67] Oral levoketoconazole may have a lower risk of liver toxicity than oral ketoconazole.[68]
Veterinary use
Ketoconazole is sometimes prescribed as an antifungal by veterinarians for use in pets, often as unflavored tablets that may need to be cut to smaller size for correct dosage.[69]
References
- ^ "Ketoconazole". Merriam-Webster.com Dictionary.
- ^ "Ketoconazole". Dictionary.com Unabridged (Online). n.d.
- FDA. Retrieved 22 October 2023.
- ^ "Ketoconazole HRA 200mg Tablets - Summary of Product Characteristics (SmPC)". (emc). 18 September 2017. Archived from the original on 2 August 2020. Retrieved 1 April 2020.
- ^ "Ketoconazole 2% w/w Shampoo - Summary of Product Characteristics (SmPC)". (emc). 5 October 2015. Archived from the original on 3 August 2020. Retrieved 1 April 2020.
- ^ a b c d e f g h i j "Ketoconazole tablet". DailyMed. 26 June 2018. Archived from the original on 5 August 2020. Retrieved 5 January 2020.
- ^ a b "Ketoconazole HRA EPAR". European Medicines Agency (EMA). 23 April 2012. Archived from the original on 3 August 2020. Retrieved 1 April 2020.
- ^ ISBN 978-0-203-90831-0.
- ^ a b c "Assessment report: Ketoconazole HRA" (PDF). European Medicines Agency (EMA). Archived (PDF) from the original on 27 August 2016. Retrieved 26 August 2016.
- ^ S2CID 26531459.
- ^ a b c d e f g h "Ketoconazole Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Archived from the original on 28 December 2010. Retrieved 23 March 2019.
- ^ ISBN 9780857113382.
- ^ U.S. Food and Drug Administration. 26 July 2013. Archivedfrom the original on 2 December 2013. Retrieved 23 November 2013.
- ^ ISBN 9783527607495.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Ketoconazole - Drug Usage Statistics". ClinCalc. Archived from the original on 8 July 2020. Retrieved 14 January 2024.
- ^ "Oral ketoconazole (Nizoral) 200 mg tablets". Therapeutic Goods Administration (TGA). 10 October 2013. Archived from the original on 2 July 2015. Retrieved 23 March 2019.
- ^ S2CID 206695486.
- China Food and Drug Administration. 25 June 2015. Archivedfrom the original on 2 July 2015. Retrieved 2 July 2015.
- ^ ISBN 978-1-4377-2003-7.
- ISBN 9780723435716.
- ISBN 978-1-4377-2003-7.
- ^ a b c d Finkel R, Cubeddu LX, Clark MA (2009). Pharmacology (4th ed.). Baltimore: Lippincott Williams & Wilkins. p. 411.
- ISBN 978-0-7216-9652-2.
- ^ ISBN 978-0-7817-1750-2.
- from the original on 12 December 2015.
- S2CID 30635892.
- from the original on 29 August 2021. Retrieved 4 July 2019.
- S2CID 20886812.
- S2CID 209331373.
- ^ "MedScape". Ectopic Cortisol Production Derived From Malignant Testicular Masses: Treatment and Management. Nature Publishing Group. Archived from the original on 13 May 2018. Retrieved 18 April 2015.
- ^ PMID 6304148.
- ISBN 9780781772075.
- PMID 3023421.
- PMID 16986003.
- ^ a b c d e "Nizoral (Ketoconazole): Side Effects, Interactions, Warning, Dosage & Uses". RxList. Archived from the original on 7 April 2019. Retrieved 7 April 2019.
- S2CID 22938364.
- ^ a b "Ketoconazole (Nizoral) Use During Pregnancy". Drugs.com. Archived from the original on 10 April 2020. Retrieved 24 May 2020.
- S2CID 41187361.
- S2CID 8440973.
- PMID 9043118.
- PMID 2652864.
- S2CID 42845713.
- from the original on 11 February 2020. Retrieved 4 July 2019.
- PMID 6135709.
- S2CID 21455699.
- S2CID 39334280.
- ^ S2CID 9148909.
- ^ ISBN 978-1-4419-1435-4.
- ^ PMID 29256130.
- PMID 3536461.
- PMID 8174717.
- ^ PMID 2194539.
- ISSN 0171-2004.
- PMID 29316407.
- PMID 7486898.
- ^ ISBN 978-1-4757-2085-3. Archivedfrom the original on 10 January 2023. Retrieved 11 April 2017.
- ^ ISBN 978-3-88763-075-1. Archivedfrom the original on 10 January 2023. Retrieved 22 July 2018.
- PMID 490531.
- ISBN 978-0-8155-1856-3. Archivedfrom the original on 10 January 2023. Retrieved 14 June 2019.
- ^ ISBN 978-0-7817-8355-2. Archivedfrom the original on 10 January 2023. Retrieved 11 April 2017.
- ^ "Ketoconazole-containing medicines". European Medicines Agency (EMA). 25 July 2013. Archived from the original on 22 February 2024. Retrieved 22 February 2024.
- ^ "Ketoconazole HRA recommended for approval in Cushing's syndrome". European Medicines Agency (EMA). 26 September 2014. Archived from the original on 22 February 2024. Retrieved 22 February 2024.
- ^ ISBN 978-94-011-4439-1.
- ^ a b c "Ketoconazole". Archived from the original on 23 July 2018. Retrieved 22 July 2018.
- ^ "European Medicines Agency recommends suspension of marketing authorisations for oral ketoconazole" (Press release). European Medicines Agency (EMA). 26 July 2013. Archived from the original on 14 January 2014.
- ^ "Levoketoconazole - Strongbridge Biopharma - AdisInsight". Archived from the original on 23 January 2020. Retrieved 14 June 2019.
- PMID 27600150.
- ^ KuKanich B (January 2008). "A review of selected systemic antifungal drugs for use in dogs and cats". Veterinary Medicine. Archived from the original on 5 October 2013.
