Kiliani–Fischer synthesis
The Kiliani–Fischer synthesis, named for
Classic Kiliani–Fischer synthesis
The original version of the Kiliani–Fischer synthesis proceeds through
Improved version
More recently, an improved reduction method has been developed that produces somewhat higher yields of the larger sugars. Instead of conversion of the cyanohydrin to a lactone, the cyanohydrin is
. Due to the presence of water, the imine quickly hydrolyzes to form an aldehyde, thus the final sugars are produced in just two steps rather than three. The separation of the isomers is then performed at the stage of the sugar products themselves rather than at the lactone intermediates. The special catalyst is needed to avoid further reduction of the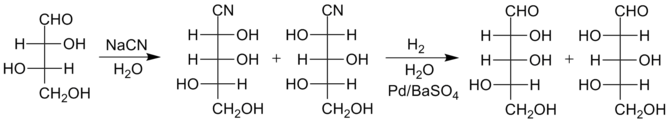
Uses and limitations
Both enantiomers of glyceraldehyde are commercially available, so one can access any stereoisomer of any chain-length aldose by an appropriate number of repeated applications of the Kiliani–Fischer synthesis. The triose D-glyceraldehyde (1) leads to the tetroses D-erythrose (2a) and D-threose (2b). Those lead to the pentoses D-ribose (3a) and D-arabinose (3b), and D-xylose (3c) and D-lyxose (3d), respectively. The next iteration leads to the hexoses D-allose (4a) and D-altrose (4b), D-glucose (4c) and D-mannose (4d), D-gulose (4e) and D-idose (4f), and D-galactose (4g) and D-talose (4h). The D-heptoses and beyond are available by continuing the sequence, and enantiomeric L series is available by starting the sequence with L-glyceraldehyde.

In practice, the Kiliani–Fischer synthesis is usually used for production of sugars that are difficult or impossible to obtain from natural sources. While it does provide access to every possible stereoisomer of any desired aldose, the process is limited by its low yield and use of toxic reagents. In addition, the process requires having a supply of the previous sugar in the series, which may itself require substantial synthetic work if it is not readily available. For example, if successive iterations of the Kiliani–Fischer synthesis are used, the overall yield drops approximately exponentially for each additional iteration.
The process only provides direct access to aldoses, whereas some sugars of interest may instead be ketoses. Some ketoses may be accessible from similar aldoses by isomerization via an
See also
- Homologation reactions
- Wohl degradation
- Ruff degradation
References
- Carey, Francis A. (2006). Organic Chemistry, Sixth Edition, New York, NY: McGraw-Hill. ISBN 0-07-111562-5.
