Knorr pyrrole synthesis
| Knorr pyrrole synthesis | |
|---|---|
| Named after | Ludwig Knorr |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000497 |
The Knorr pyrrole synthesis is a widely used

Method
The mechanism requires zinc and acetic acid as catalysts. It will proceed at room temperature. Because α-aminoketones self-condense very easily, they must be prepared in situ. The usual way of doing this is from the relevant oxime, via the Neber rearrangement.[5][6]
The original Knorr synthesis employed two equivalents of
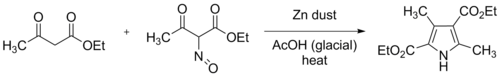
Modern practice is to add the oxime solution resulting from the nitrosation and the zinc dust gradually to a well-stirred solution of ethyl acetoacetate in glacial acetic acid. The reaction is
Knorr's pyrrole can be derivatized in a number of useful manners. One equivalent of
Levi and Zanetti extended the Knorr synthesis in 1894 to the use of
The mechanism of the Knorr pyrrole synthesis begins with condensation of the amine and ketone to give an imine. The imine then

Related synthesis
There are a number of important syntheses of pyrroles that are operated in the manner of the Knorr Synthesis, despite having mechanisms of very different connectivity between the starting materials and the pyrrolic product.
George Kleinspehn reported that the Fischer–Fink connectivity could be forced to occur exclusively, by the use of diethyl oximinomalonate in the synthesis, with 2,4-pentanedione, or its 3-alkyl substituted derivatives. Yields were high, around 60%, and this synthesis eventually came to be one of the most important in the repertory.[13] Yields were significantly improved, by the use of preformed diethyl aminomalonate (prepared by the hydrogenolysis of diethyl oximinomalonate in ethanol, over Pd/C), and adding a mixture of diethyl aminomalonate and the β-diketone to actively boiling glacial acetic acid.[14]
Meanwhile, Johnson had extended the Fischer-Fink synthesis by reacting 2-oximinoacetoacetate esters (ethyl, benzyl, or tertiary-butyl), with 3-alkyl substituted 2,4-pentanediones.[15] The Kleinspehn synthesis was extended under David Dolphin by the use of unsymmetrical β-diketones (such as 3-alkyl substituted 2,4-hexanediones), which preferentially reacted initially at the less hindered acetyl group and afforded the corresponding 5-methylpyrrole-2-carboxylate esters. N,N-Dialkyl 2-oximinoacetoacetamides also were found to give pyrroles when reacted under Knorr conditions with 3-substituted-2,4-pentanediones, in yields comparable to the corresponding esters (around 45%). However, when unsymmetrical diketones were used, it was found that the acetyl group from the acetoacetamide was retained in the product, and one of the acyl groups from the diketone had been lost.[16] This same mechanism occurs to a minor extent in the acetoacetate ester systems, and had previously been detected radiochemically by Harbuck and Rapoport.[17] Most of the above-described syntheses have application in the synthesis of porphyrins, bile pigments, and dipyrrins.
References
- .
- .
- .
- Wiley. pp. 287 ff.
- ; Collected Volumes, vol. 2, p. 202.
- ; Collected Volumes, vol. 3, p. 513.
- .
- .
- La Gazzetta Chimica Italiana(in Italian). 24 (1): 546–554.
- .
- doi:10.1139/v66-149.
- .
- .
- .
- .
- .
- .
