L-DOPA
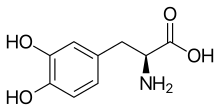 Skeletal formula of L-DOPA | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌɛlˈdoʊpə/, /ˌlɛvoʊˈdoʊpə/ |
| Trade names | Larodopa, Dopar, Inbrija, others |
| AHFS/Drugs.com | Professional Drug Facts |
| MedlinePlus | a619018 |
| License data | |
| Pregnancy category |
|
intravenous | |
| ATC code | |
| Legal status | |
| Legal status | |
renal 70–80% | |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
l-DOPA, also known as levodopa and l-3,4-dihydroxyphenylalanine, is made and used as part of the normal
l-DOPA has a counterpart with opposite chirality, d-DOPA. As is true for many molecules, the human body produces only one of these isomers (the l-DOPA form). The enantiomeric purity of l-DOPA may be analyzed by determination of the optical rotation or by chiral thin-layer chromatography.[7]
Medical use
l-DOPA crosses the protective
Once l-DOPA has entered the
In humans, conversion of l-DOPA to dopamine does not only occur within the
Inbrija (previously known as CVT-301) is an inhaled powder formulation of levodopa indicated for the intermittent treatment of "off episodes" in patients with Parkinson's disease currently taking carbidopa/levodopa.[12] It was approved by the United States Food and Drug Administration on December 21, 2018, and is marketed by Acorda Therapeutics.[13]
Coadministration of pyridoxine without a DDCI accelerates the peripheral decarboxylation of l-DOPA to such an extent that it negates the effects of l-DOPA administration, a phenomenon that historically caused great confusion.
In addition, l-DOPA, co-administered with a peripheral DDCI, is efficacious for the short-term treatment of
The two types of response seen with administration of l-DOPA are:
- The short-duration response is related to the half-life of the drug.
- The longer-duration response depends on the accumulation of effects over at least two weeks, during which ΔFosB accumulates in nigrostriatal neurons. In the treatment of Parkinson's disease, this response is evident only in early therapy, as the inability of the brain to store dopamine is not yet a concern.
Biological role
l-DOPA is produced from the amino acid l-tyrosine by the enzyme tyrosine hydroxylase. l-DOPA can act as an l-tyrosine mimetic and be incorporated into proteins by mammalian cells in place of L-tyrosine, generating protease-resistant and aggregate-prone proteins in vitro and may contribute to neurotoxicity with chronic l-DOPA administration.[18] It is also the precursor for the
l-DOPA can be directly metabolized by
l-Phenylalanine, l-tyrosine, and l-DOPA are all precursors to the biological
Marine adhesion
l-DOPA is a key
Side effects and adverse reactions
The side effects of l-DOPA may include:
- Hypertension, especially if the dosage is too high
- Arrhythmias, although these are uncommon
- Nausea, which is often reduced by taking the drug with food, although protein reduces drug absorption. l-DOPA is an amino acid, so protein competitively inhibits l-DOPA absorption.
- Gastrointestinal bleeding
- Disturbed respiration, which is not always harmful, and can actually benefit patients with upper airway obstruction
- Hair loss
- confusion
- Extreme emotional states, particularly anxiety, but also excessive libido
- Vivid dreams or insomnia
- Auditory or visual hallucinations
- Effects on learning; some evidence indicates it improves working memory, while impairing other complex functions
- Somnolence and narcolepsy
- A condition similar to stimulant psychosis
Although many adverse effects are associated with l-DOPA, in particular psychiatric ones, it has fewer than other
More serious are the effects of chronic l-DOPA administration in the treatment of Parkinson's disease, which include:
- End-of-dose deterioration of function
- "On/off" oscillations
- Freezing during movement
- Dose failure (drug resistance)
- Dyskinesia at peak dose (levodopa-induced dyskinesia)
- Possible dopamine dysregulation: The long-term use of l-DOPA in Parkinson's disease has been linked to the so-called dopamine dysregulation syndrome.[29]
Clinicians try to avoid these side effects and adverse reactions by limiting l-DOPA doses as much as possible until absolutely necessary.
The long term use of L-Dopa increases oxidative stress through
History
In work that earned him a
The 2001
Research
In 2015, a retrospective analysis comparing the incidence of
Role in plants and in the environment
In plants, L-DOPA functions as an
See also
- d-DOPA (Dextrodopa)
- l-DOPS(Droxidopa)
- Methyldopa (Aldomet, Apo-Methyldopa, Dopamet, Novomedopa, etc.)
- Dopamine (Intropan, Inovan, Revivan, Rivimine, Dopastat, Dynatra, etc.)
- Ciladopa
- Neuroleptic malignant syndrome
- Melanin (a metabolite)
References
- S2CID 96802274.
- ^ a b "Levodopa Use During Pregnancy". Drugs.com. 12 July 2019. Retrieved 27 September 2020.
- ^ PMID 35939305.
- PMID 18828673.
- PMID 24117106.
- PMID 26683006.
- S2CID 97903386.
- S2CID 22874032.
- PMID 28904446.
- ^ "Medicare D". Medicare. 2014. Retrieved 12 November 2015.
- ^ "Lodosyn", Drugs, nd, retrieved 12 November 2012
- ^ "Inbrija Prescribing Information" (PDF). Retrieved February 14, 2019.
- ^ "Acorda Therapeutics Announces FDA Approval of INBRIJA™ (levodopa inhalation powder)". ir.acorda.com. Retrieved 2019-02-14.
- PMID 21328278.
- PMID 19948186.
- PMID 15860375.
- PMID 24374199.
- S2CID 2288729.
- PMID 1281049. Archived from the original(PDF) on 2011-06-07. Retrieved 2008-10-16.
- PMID 6433900.
- S2CID 136967853.
- ^ "Study Reveals Details Of Mussels' Tenacious Bonds". Science Daily. Aug 16, 2006. Retrieved Sep 30, 2013.
- ^ "Mussel Adhesive Protein Mimetics". Archived from the original on 2006-05-29.
- S2CID 234474122.
- PMID 24936704.
- PMID 28344244.
- PMID 34423482.
- PMID 25110984.
- PMID 17988927.
- PMID 24653659.
- S2CID 32896604.
- PMID 13869404.
- PMID 5791298.
- ^ "Lasker Award". 1969. Archived from the original on 2016-01-05., accessed April 1, 2013
- ISBN 978-1-934559-87-1– via Google eBook.
- PMID 5637779.
- .
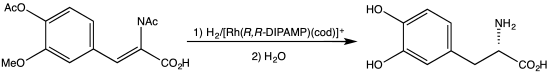
- ^ "Synthetic scheme for total synthesis of DOPA, L- (Monsanto)". UW Madison, Department of Chemistry. Retrieved Sep 30, 2013.
- .
- PMID 26524704.
- .
- PMID 37620733.
External links
- "Levodopa". Drug Information Portal. U.S. National Library of Medicine.