Lactone
Lactones are cyclic
Lactones are formed by intramolecular
Nomenclature
Greek prefixes in alphabetical order indicate ring size.
| Ring size (number of atoms in the ring) |
Systematic name | IUPAC name
|
Common name(s) | Structure |
|---|---|---|---|---|
| 3 | α-lactone | Oxiran-2-one | Acetolactone | |
| 4 | β-lactone | Oxetan-2-one |
|

|
| 5 | γ-lactone
|
Oxolan-2-one | γ-Butyrolactone | 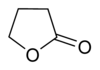
|
| 6 | δ-lactone | Oxan-2-one | 
| |
| 7 | ε-lactone | Oxepan-2-one |
|
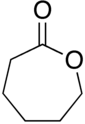
|
Lactones are usually named according to the precursor acid molecule (aceto = 2 carbon atoms, propio = 3, butyro = 4, valero = 5, capro = 6, etc.), with a -lactone suffix and a Greek letter prefix that specifies the number of carbon atoms in the heterocycle — that is, the distance between the relevant -OH and the -COOH groups along said backbone. The first carbon atom after the carbon in the -COOH group on the parent compound is labelled α, the second will be labeled β, and so forth. Therefore, the prefixes also indicate the size of the lactone ring: α-lactone = 3-membered ring, β-lactone = 4-membered, γ-lactone = 5-membered, δ-lactone = 6-membered, etc.
The other suffix used to denote a lactone is -olide, used in substance class names like butenolide, macrolide, cardenolide or bufadienolide.
To obtain the preferred IUPAC names, lactones are named as heterocyclic pseudoketones by adding the suffix 'one', 'dione', 'thione', etc. and the appropriate multiplicative prefixes to the name of the heterocyclic parent hydride.[4]
Etymology
The name lactone derives from the ring compound called
In 1880 the German chemist Wilhelm Rudolph Fittig extended the name "lactone" to all intramolecular carboxylic esters.[6]
Occurrence
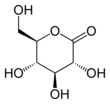
The most stable lactones are the 5-membered γ-lactones and 6-membered δ-lactones because, as in most organic cycles, 5 and 6 membered rings minimize the strain of
Many naturally-occurring lactones are γ- and δ-lactones, both saturated and unsaturated. They contribute to the aroma of fruits, butter, cheese, and other foods.
Macrocyclic lactones are also important natural products.
Lactone rings occur widely as building blocks in nature, such as in
).Synthesis

Many methods in ester synthesis can also be applied to that of lactones. Lactonization competes with polymerization for longer hydroxy acids, or the strained β‑lactones. γ‑Lactones, on the other hand, are so stable that 4-hydroxy acids (R-CH(OH)-(CH2)2-CO2H) spontaneously cyclise.
In one industrial synthesis of

In
Specific methods include

The γ-lactones
An alternative radical reaction yielding γ-lactones is the manganese-mediated coupling.
Reactions
Lactones exhibit the reactions characteristic of esters.
Hydrolysis and aminolysis
Heating a lactone with a base (
Lactones also react with amines to give the ring-opened alcohol and amide.
Reduction
Lactones can be reduced to diols using lithium aluminium hydride. For instance, gamma-lactones is reduced to butane-1,4-diol, (CH2(OH)-(CH2)2-CH2(OH).
Polymerization
Lactones readily form polyesters according to the formula, and have been shown to oligomerize without catalyst as well:[14][15]
The double lactone called lactide polymerizes to polylactic acid (polylactide). The resulting materials, polylactic acid, have many attractive properties.[16][17]
Uses
Flavors and fragrances
Lactones contribute significantly to the flavor of fruit, and of unfermented and fermented dairy products,
Macrocyclic lactones (cyclopentadecanolide, 15-pentadec-11/12-enolide) have odors similar to macrocyclic ketones of animal origin (muscone, civetone), but they can be prepared more easily, for example, by depolymerization of the corresponding linear polyesters. Replacement of a methylene unit by oxygen barely affects the odor of these compounds, and oxalactones with 15 – 17-membered rings are produced in addition to cyclopentadecanolide (e. g., 12-oxa-16-hexadecanolide).[9]
Prebiotic chemistry
Prebiotically plausible lactones, such as ε-caprolactone and δ-valerolactone, have been shown to oligomerize without the usage of catalysts forming oligomers that may have been relevant during the origin of life.[21]
Plastics
Polycaprolactone is an important plastic.
Dilactones
- Ellagic acid (Hexahydroxydiphenic acid dilactone)
- Flavogallonic acid dilactone can be found in Rhynchosia volubilis seeds and in Shorea laeviforia
- Lactide
- Tergallic acid dilactone can be found in Rhynchosia volubilis seeds
- Valoneic acid dilactone can be isolated from the heartwood of Shorea laeviforia
- Ethylene brassylate (Musk T), a widely used synthetic musk
See also
References and notes
- Compendium of Chemical Terminology, 2.3.3, International Union of Pure and Applied Chemistry, 2014-02-24, p. 817
- ^ Francis A. Carey; Robert M. Giuliano (2011), Organic Chemistry (8th ed.), McGraw-Hill, pp. 798–799
- ^ Steven A. Hardinger. "Illustrated Glossary of Organic Chemistry". Department of Chemistry & Biochemistry, UCLA.
- ISBN 978-0-85404-182-4.
- ^ Pelouze, J. (9 December 1844). "Mémoire sur l'acide lactique" [Memoir on lactic acid]. Comptes rendus (in French). 19: 1219–1227.
From p. 1223: "Indépendamment de la lactide dont je viens de rappeler l'existence dans les produits de la distllation de l'acide lactique, celui-ci donne encore, par sa décomposition, une autre substance, que je propose d'appeler lactone, parce qu'elle me paraît être à l'acide lactique ce que l'acétone est à l'acide acétique." (Independently of the lactide of which I have just recalled the existence in the products of the distillation of lactic acid, this [i.e., lactic acid] gives further, by its decomposition, another substance, which I propose to call lactone, because it seems to me to be to lactic acid what acetone is to acetic acid.)
- Reprinted: Pelouze, J. (1845). "Mémoire sur l'acide lactique" [Memoir on lactic acid]. Annales de Chimie et de Physique. 3rd series (in French). 13: 257–268. ; see p. 262.
- English translation: Pelouze, J. (January 15, 1845). "Researches on lactic acid". The Chemical Gazette. 3 (54): 29–35. ; see p. 31.
- Menten, Pierre de (2013). Dictionnaire de chimie: Une approche étymologique et historique [Dictionary of Chemistry: an etymological and historical approach] (in French). Brussels, Belgium: de boeck. p. 183. ISBN 9782804181758.
- . From p. 62: "Es ist wünschenswerth, für diese Gruppe von Verbindungen, deren bis jetzt einfachster Repräsentant der im Vorstehenden beschriebene Körper ist, eine allgemeine Bezeichnungsweise zu haben, und da der Name "Lactide" nicht anwendbar ist, weil dann das Lactid κατ εξοχην kein Lactid sein wurde, so schlagen wir als Gruppenbezeichnung den Namen "Lactone" vor". (It's desirable for this group of compounds — whose simplest representative until now has been the substance that's described in the preceding — to have a general designation, and since the name "lactide" isn't applicable because then the archetypal lactide would not be a lactide, we therefore suggest the name "lactone" as the designation of this group [of compounds].)
- .
- ^ a b c d Karl-Georg Fahlbusch; et al. (2007), "Flavors and Fragrances", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley, pp. 74‒78
- ^ Oliver, Garrett. "The Oxford Companion to Beer definition of barrel-aging". Craft Beer and Brewing.
- lead tetraacetate with ring-opening and finally reduction of the aldehyde to the alcohol with sodium borohydrideand intramolecular lactone formation
- ^ Organic Syntheses, Coll. Vol. 7, p.164 (1990); Vol. 64, p.175 (1986) Article link
- ^ Wilhelm Riemenschneider; Hermann M. Bolt (2007), "Esters, Organic", Ullmann's Encyclopedia of Industrial Chemistry (7th ed.), Wiley
- PMID 33067516.
- ISBN 978-0-470-29366-9.
- PMID 15584698.
- ^ ISBN 9783540493396. Retrieved 2 July 2015.
- ISBN 9781439853351. Retrieved 2 July 2015.
- ISBN 9781420017045. Retrieved 2 July 2015.
- PMID 31963928.

