Lactose
| |
| Names | |
|---|---|
| IUPAC name
β-D-Galactopyranosyl-(1→4)-D-glucose
| |
| Systematic IUPAC name
(2R,3R,4S,5R,6S)-2-(Hydroxymethyl)-6-{[(2R,3S,4R,5R,6R)-4,5,6-trihydroxy-2-(hydroxymethyl)oxan-3-yl]oxy}oxane-3,4,5-triol | |
| Other names
Milk sugar
Lactobiose 4-O-β-D-Galactopyranosyl-D-glucose | |
| Identifiers | |
3D model (
JSmol ) |
|
| 90841 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.000.509 |
| EC Number |
|
| 342369 | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H22O11 | |
| Molar mass | 342.297 g·mol−1 |
| Appearance | White solid |
| Density | 1.525 g/cm3 |
| Melting point | 252 °C (anhydrous)[1] 202 °C (monohydrate)[1] |
| 195 g/L[2][3] | |
Chiral rotation ([α]D)
|
+55.4° (anhydrous) +52.3° (monohydrate) |
| Thermochemistry | |
Std enthalpy of (ΔcH⦵298)combustion |
5652 kJ/mol, 1351 kcal/mol, 16.5 kJ/g, 3.94 kcal/g |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 357.8 °C (676.0 °F; 631.0 K)[4] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lactose, or milk sugar, is a
Structure and reactions


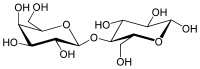
Lactose is a disaccharide derived from the condensation of galactose and glucose, which form a β-1→4 glycosidic linkage. Its systematic name is β-D-galactopyranosyl-(1→4)-D-glucose. The glucose can be in either the α-pyranose form or the β-pyranose form, whereas the galactose can have only the β-pyranose form: hence α-lactose and β-lactose refer to the anomeric form of the glucopyranose ring alone.
Detection reactions for lactose are the Woehlk-
Lactose is
Lactulose is a commercial product, used for treatment of constipation.Occurrence and isolation
Lactose comprises about 2–8% of milk by weight. Several million tons are produced annually as a by-product of the
Dairy products such as yogurt and cheese contain very little lactose. This is because the bacteria used to make these products breaks down lactose through the use of lactase.
Metabolism
Infant mammals nurse on their mothers to drink milk, which is rich in lactose. The intestinal villi secrete the enzyme lactase (β-D-galactosidase) to digest it. This enzyme cleaves the lactose molecule into its two subunits, the simple sugars glucose and galactose, which can be absorbed. Since lactose occurs mostly in milk, in most mammals, the production of lactase gradually decreases with maturity due to weaning; the removal of lactose from the diet removes the metabolic pressure to continue to produce lactase for its digestion.
Many people with ancestry in
Biological properties
The sweetness of lactose is 0.2 to 0.4, relative to 1.0 for sucrose.[15] For comparison, the sweetness of glucose is 0.6 to 0.7, of fructose is 1.3, of galactose is 0.5 to 0.7, of maltose is 0.4 to 0.5, of sorbose is 0.4, and of xylose is 0.6 to 0.7.[15]
When lactose is completely digested in the
The glycemic index of lactose is 46 to 65.[15][16] For comparison, the glycemic index of glucose is 100 to 138, of sucrose is 68 to 92, of maltose is 105, and of fructose is 19 to 27.[15][16]
Lactose has relatively low
Applications
Its mild flavor and easy handling properties have led to its use as a carrier and stabiliser of aromas and pharmaceutical products.[5] Lactose is not added directly to many foods, because its solubility is less than that of other sugars commonly used in food. Infant formula is a notable exception, where the addition of lactose is necessary to match the composition of human milk.
Lactose is not fermented by most
Yeast belonging to the genus Kluyveromyces have a unique industrial application, as they are capable of fermenting lactose for ethanol production. Surplus lactose from the whey by-product of dairy operations is a potential source of alternative energy.[18]
Another significant lactose use is in the pharmaceutical industry. Lactose is added to tablet and capsule drug products as an ingredient because of its physical and functional properties.[
History
The first crude isolation of lactose, by Italian physician Fabrizio Bartoletti (1576–1630), was published in 1633.[20] In 1700, the Venetian pharmacist Lodovico Testi (1640–1707) published a booklet of testimonials to the power of milk sugar (saccharum lactis) to relieve, among other ailments, the symptoms of arthritis.[21] In 1715, Testi's procedure for making milk sugar was published by Antonio Vallisneri.[22] Lactose was identified as a sugar in 1780 by Carl Wilhelm Scheele.[23][9]
In 1812, Heinrich Vogel (1778–1867) recognized that glucose was a product of hydrolyzing lactose.[24] In 1856, Louis Pasteur crystallized the other component of lactose, galactose.[25] By 1894, Emil Fischer had established the configurations of the component sugars.[26]
Lactose was named by the French chemist Jean Baptiste André Dumas (1800–1884) in 1843.[27] In 1856, Pasteur named galactose "lactose".[28] In 1860, Marcellin Berthelot renamed it "galactose", and transferred the name "lactose" to what is now called lactose.[29] It has a formula of C12H22O11 and the hydrate formula C12H22O11·H2O, making it an isomer of sucrose.
See also
References
- ^ ISBN 978-0-8493-3829-8.
- ^ "D-Lactose".
- . ds
- ^ Sigma Aldrich
- ^ ISBN 978-3-527-30673-2.
- .
- ISSN 0003-2654.
- S2CID 208714341.
- ^ ISBN 978-0-85334-997-6
- ISBN 978-0-7514-0404-3
- PMID 24517206, DOI is open access
- ISBN 0-03-014813-8
- ^ Wade, Nicholas (2006-12-10), "Study Detects Recent Instance of Human Evolution", New York Times.
- ISBN 978-0-06-089408-5.
- ^ S2CID 10346203. Archived from the original(PDF) on Mar 2, 2019.
- ^ S2CID 14574754.
- ^ ISBN 978-1-4200-0431-1.
- ^ Ling, Charles (2008), Whey to Ethanol: A Biofuel Role for Dairy Cooperatives? (PDF), United States Department of Agriculture Rural Development.
- PMID 32141480.
- Sendivogius.] Dissolve it in [its] own water and coagulate. Repeat the operation until you have cream of whey, recalling, by [its] taste, only manna.)
In 1688, the German physician Michael Ettmüller (1644–1683) reprinted Bartoletti's preparation. See: Ettmüller, Michael, Opera Omnia … (Frankfurt am Main ("Francofurtum ad Moenum"), [Germany]: Johann David Zunner, 1688), book 2, page 163. Archived 2018-11-09 at the Wayback Machine From page 163: "Undd Bertholetus praeparat ex sero lactis remedium, quod vocat mannam S. [alchemical symbol for salt, salem] seri lactis vid. in Encyclopaed. p. 400. Praeparatio est haec: … " (Whence Bartoletti prepared from milk whey a medicine, which he called manna or salt of milk whey; see in [his] Encyclopedia [note: this is a mistake; the preparation appeared in Bartoletti's Methodus in dyspnoeam … ], p. 400. This is the preparation: … ) - ^ Lodovico Testi, De novo Saccharo Lactis [On the new milk sugar] (Venice, (Italy): Hertz, 1700).
- ^ Ludovico Testi (1715) "Saccharum lactis" (Milk sugar), Academiae Caesareo-Leopoldinae naturae curiosorum ephemerides, … , 3 : 69–79. The procedure was also published in Giornale de' letterati d'Italia in 1715.
- ^ See:
- Carl Wilhelm Scheele (1780) "Om Mjölk och dess syra" (About milk and its acid), Kongliga Vetenskaps Academiens Nya Handlingar (New Proceedings of the Royal Academy of Science), 1 : 116–124. From page 116: "Det år bekant, at Ko-mjölk innehåller Smör, Ost, Mjölk-såcker, … " (It is known, that cow's milk contains butter, cheese, milk-sugar, … )
- Carl Wilhelm Scheele (1780) "Om Mjölk-Såcker-Syra" (On milk-sugar acid), Kongliga Vetenskaps Academiens Nya Handlingar (New Proceedings of the Royal Academy of Science), 1 : 269–275. From pages 269–270: "Mjölk-Såcker år et sal essentiale, som uti Mjölken finnes uplöst, och som, för dess sötaktiga smak skull, fått namn af såcker." (Milk sugar is an essential salt, which is found dissolved in milk, and which, on account of its sweet taste, has the name of "sugar".)
- ^ See:
- Vogel (1812) "Sur le sucre liquide d'amidon, et sur la transmutation des matières douces en sucre fermentescible" (On the liquid sugar of starch, and on the transformation of sweet materials into fermentable sugars), Annales de chemie et de physique, series 1, 82 : 148–164; see especially pages 156–158.
- H. A. Vogel (1812) "Ueber die Verwandlung der Stärke und andrer Körper in Zucker" (On the conversion of starches and other substances into sugar), Annalen der Physik, new series, 42 : 123–134; see especially pages 129–131.
- ^ Pasteur (1856) "Note sur le sucre de lait" (Note on milk sugar), Comptes rendus, 42 : 347–351.
- ^ Fischer determined the configuration of glucose in:
- Emil Fischer (1891) "Ueber die Configuration des Traubenzuckers und seiner Isomeren" (On the configuration of grape sugar and its isomers), Berichte der Deutschen Chemischen Gesellschaft, 24 : 1836–1845.
- Emil Fischer (1891) "Ueber die Configuration des Traubenzuckers und seiner Isomeren. II" (On the configuration of grape sugar and its isomers), Berichte der Deutschen Chemischen Gesellschaft, 24 : 2683–2687.
- Emil Fischer and Robert S. Morrell (1894) "Ueber die Configuration der Rhamnose und Galactose" (On the configuration of rhamnose and galactose), Berichte der Deutschen chemischen Gesellschaft zu Berlin, 27 : 382–394. The configuration of galactose appears on page 385.
- ^ Dumas, Traité de Chimie, Appliquée aux Arts, volume 6 (Paris, France: Bechet Jeune, 1843), p. 293.
- ^ Pasteur (1856) "Note sur le sucre de lait" (Note on milk sugar), Comptes rendus, 42 : 347–351. From page 348: "Je propose de le nommer lactose." (I propose to name it lactose.)
- ^ Marcellin Berthelot, Chimie organique fondée sur la synthèse [Organic chemistry based on synthesis] (Paris, France: Mallet-Bachelier, 1860), vol. 2, pp. 248–249 and pp. 268–270.
External links
 Media related to Lactose at Wikimedia Commons
Media related to Lactose at Wikimedia Commons

