Lead(II) oxide

| |
| |
| Names | |
|---|---|
| IUPAC name
Lead(II) oxide
| |
| Other names | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.013.880 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3288 2291 3077 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| PbO | |
| Molar mass | 223.20 g/mol |
| Appearance | red or yellow powder |
| Density | 9.53 g/cm3 |
| Melting point | 888 °C (1,630 °F; 1,161 K) |
| Boiling point | 1,477 °C (2,691 °F; 1,750 K) |
| 0.017 g/L[1] | |
| Solubility | insoluble in dilute alkalis, alcohol soluble in concentrated alkalis soluble in HCl, ammonium chloride |
| 4.20×10−5 cm3/mol | |
| Structure | |
Tetragonal, tP4
| |
| P4/nmm, No. 129 | |
| Hazards | |
| GHS labelling: | |
  
| |
| Danger | |
| H302, H332, H351, H360Df, H362, H373, H410 | |
| P201, P202, P260, P261, P263, P264, P270, P271, P273, P281, P301+P312, P304+P312, P304+P340, P308+P313, P312, P314, P330, P391, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LDLo (lowest published)
|
1400 mg/kg (dog, oral)[2] |
| Safety data sheet (SDS) | ICSC 0288 |
| Related compounds | |
Other anions
|
Lead(II) sulfide Lead selenide Lead telluride |
Other cations
|
Carbon monoxide Silicon monoxide Germanium monoxide Tin(II) oxide |
Lead(II,II,IV) oxide
Lead dioxide | |
Related compounds
|
Thallium(III) oxide Bismuth(III) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lead(II) oxide, also called lead monoxide, is the inorganic compound with the molecular formula
Types
Lead oxide exists in two types:
- Red tetragonal (α-PbO), obtained at temperatures below 486 °C (907 °F)
- Yellow orthorhombic (β-PbO), obtained at temperatures above 486 °C (907 °F)
Synthesis
PbO may be prepared by heating lead metal in air at approximately 600 °C (1,100 °F). At this temperature it is also the end product of decomposition of other
Thermal decomposition of lead(II) nitrate or lead(II) carbonate also results in the formation of PbO:
PbO is produced on a large scale as an intermediate product in refining raw lead ores into metallic lead. The usual lead ore is galena (lead(II) sulfide). At a temperature of around 1,000 °C (1,800 °F) the sulfide is converted to the oxide:[5]
- 2 PbS + 3 O
2 → 2 PbO + 2 SO2
From lead
There are two principal methods to make lead monoxide both of which resemble combustion of the lead at high temperature:[6]
- Barton pot method.
- The refined molten lead droplets are oxidized in a vessel under a forced air flow which carries them out to the separation system (e.g. cyclonic separators) for further processing.[6][7]: 245 Oxides produced by this method are mostly a mixture of α-PbO and β-PbO. The overall reaction is:
2Pb + O22PbO
- Ball mill method
- The lead balls are oxidized in a cooled rotating drum. The oxidation is achieved by collisions of the balls. Just like in Barton pot method, the supply of air and separators may also be used.[6][7]: 245
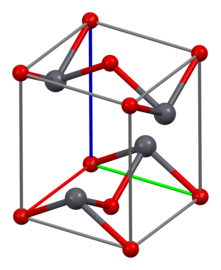
Structure
As determined by
-
Pb coordinates square-pyramidally
-
O coordinates distorted-tetrahedrally
-
Unit cell
-
3×3×3 unit cells
-
Along thea axis
-
Along thec axis
Reactions
Metallic lead is obtained by reducing PbO with carbon monoxide at around 1,200 °C (2,200 °F):[13]
- PbO + CO → Pb + CO2
The red and yellow forms of this material are related by a small change in enthalpy:
- PbO(red) → PbO(yellow) ΔH = 1.6 kJ/mol
PbO is
6O(OH)
6]4+
. With strong bases, PbO dissolves to form plumbite (also called plumbate(II)) salts:[14]
- PbO + H2O + OH−
→ [Pb(OH)
3]−
Applications
The kind of lead in
The consumption of lead, and hence the processing of PbO, correlates with the number of automobiles, because lead remains the key component of automotive
Niche or declining uses
A mixture of PbO with
PbO was one of the raw materials for
In powdered tetragonal litharge form, it can be mixed with linseed oil and then boiled to create a weather-resistant sizing used in gilding. The litharge would give the sizing a dark red color that made the gold leaf appear warm and lustrous, while the linseed oil would impart adhesion and a flat durable binding surface.
PbO is used in certain condensation reactions in organic synthesis.[18]
PbO is the input photoconductor in a video camera tube called the
Health issues

Lead oxide may be fatal if swallowed or inhaled. It causes irritation to skin, eyes, and respiratory tract. It affects gum tissue, the central nervous system, the kidneys, the blood, and the reproductive system. It can
References
- ^ Blei(II)-oxid. Merck
- ^ "Lead compounds (as Pb)". Immediately Dangerous to Life or Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ^ ISBN 978-3527306732.
- ^ ISBN 978-0-08-037941-8.
- S2CID 96393940.
- ^ ISSN 0378-7753.
- ^ )
- ISBN 0-19-855370-6[page needed]
- ISBN 978-0-7923-7521-0.
- ISBN 978-0-7099-2250-6.
- .
- ^ "ICSD Entry: 94333". Cambridge Structural Database: Access Structures. Cambridge Crystallographic Data Centre. Retrieved 2021-06-01.
- ^ Lead Processing @ Universalium.academic.ru. Alt address: Lead processing @ Enwiki.net.
- ISBN 0-12-352651-5[page needed]
- ^ Chapter 9, "Lead Compounds", in the book Ceramic and Glass Materials: Structure, Properties and Processing, published by Springer, year 2008.
- ISBN 9780819441447– via Google Books.
- ISBN 978-3527306732.
- ^ Corson, B. B. (1936). "1,4-Diphenylbutadiene". Organic Syntheses. 16: 28; Collected Volumes, vol. 2, p. 229.
- ^ "Lead(II) oxide". International Occupational Safety and Health Information Centre. Archived from the original on 2011-12-15. Retrieved 2009-06-06.


![{\displaystyle {\ce {PbO2->[{293 °C}] Pb12O19 ->[{351 °C}] Pb12O17 ->[{375 °C}] Pb3O4 ->[{605 °C}] PbO}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/65b5cadcc73921d39d6f69f4b6d59a4227e2b780)