Lead dioxide

| |

| |
| Names | |
|---|---|
| IUPAC name
Lead(IV) oxide
| |
| Other names
Plumbic oxide
Plattnerite | |
| Identifiers | |
| ChemSpider | |
ECHA InfoCard
|
100.013.795 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1872 |
CompTox Dashboard (EPA)
|
|
| Properties | |
| PbO2 | |
| Molar mass | 239.1988 g/mol |
| Appearance | dark-brown, black powder |
| Density | 9.38 g/cm3 |
| Melting point | 290 °C (554 °F; 563 K) decomposes |
| insoluble | |
| Solubility | soluble in acetic acid insoluble in alcohol |
Refractive index (nD)
|
2.3 |
| Structure | |
| hexagonal | |
| Hazards | |
| GHS labelling: | |
   
| |
| Danger | |
| H272, H302, H332, H360, H372, H373, H410 | |
| P201, P202, P210, P220, P221, P260, P261, P264, P270, P271, P273, P280, P281, P301+P312, P304+P312, P304+P340, P308+P313, P312, P314, P330, P370+P378, P391, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Safety data sheet (SDS) | External MSDS |
| Related compounds | |
Other cations
|
Tin dioxide
|
Lead(II,IV) oxide
| |
Related compounds
|
Thallium(III) oxide Bismuth(III) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lead(IV) oxide, commonly known as lead dioxide, is an
Properties
Physical

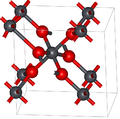
Lead dioxide has two major polymorphs, alpha and beta, which occur naturally as rare minerals scrutinyite and plattnerite, respectively. Whereas the beta form had been identified in 1845,[3] α-PbO2 was first identified in 1946 and found as a naturally occurring mineral 1988.[4]
The alpha form has
The symmetry of the beta form is
Chemical
Lead dioxide decomposes upon heating in air as follows:
The stoichiometry of the end product can be controlled by changing the temperature – for example, in the above reaction, the first step occurs at 290 °C, second at 350 °C, third at 375 °C and fourth at 600 °C. In addition, Pb2O3 can be obtained by decomposing PbO2 at 580–620 °C under an oxygen pressure of 1,400 atm (140 MPa). Therefore, thermal decomposition of lead dioxide is a common way of producing various lead oxides.[6]
Lead dioxide is an
- PbO2 + 2 NaOH + 2 H2O → Na2[Pb(OH)6]
It also reacts with basic oxides in the melt, yielding orthoplumbates M4[PbO4].
Because of the instability of its Pb4+ cation, lead dioxide reacts with hot acids, converting to the more stable Pb2+ state and liberating oxygen:[6]
- 2 PbO2 + 2 H2SO4 → 2 PbSO4 + 2 H2O + O2
- 2 PbO2 + 4 HNO3 → 2 Pb(NO3)2 + 2 H2O + O2
- PbO2 + 4 HCl → PbCl2 + 2 H2O + Cl2
However these reactions are slow.
Lead dioxide is well known for being a good oxidizing agent, with an example reactions listed below:[7]
- 2 MnSO4 + 5 PbO2 + 6 HNO3 → 2 HMnO4 + 2 PbSO4 + 3 Pb(NO3)2 + 2 H2O
- 2 Cr(OH)3 + 10 KOH + 3 PbO2 → 2 K2CrO4 + 3 K2PbO2 + 8 H2O
Electrochemical
Although the formula of lead dioxide is nominally given as PbO2, the actual oxygen to lead ratio varies between 1.90 and 1.98 depending on the preparation method. Deficiency of oxygen (or excess of lead) results in the characteristic metallic
Production
Chemical processes
Lead dioxide is produced commercially by several methods, which include oxidation of
- Pb3O4 + 4 HNO3 → PbO2 + 2 Pb(NO3)2 + 2 H2O
PbO2 reacts with sodium hydroxide to form the hexahydroxoplumbate(IV) ion [Pb(OH)6]2−, soluble in water.
Electrolysis
An alternative synthesis method is electrochemical: lead dioxide forms on pure lead, in dilute sulfuric acid, when polarized anodically at electrode potential about +1.5 V at room temperature. This procedure is used for large-scale industrial production of PbO2 anodes. Lead and copper electrodes are immersed in sulfuric acid flowing at a rate of 5–10 L/min. The electrodeposition is carried out galvanostatically, by applying a current of about 100 A/m2 for about 30 minutes.
The drawback of this method for the production of lead dioxide anodes is its softness, especially compared to the hard and brittle PbO2 which has a
Applications
Lead dioxide is used in the production of
Lead dioxide is used as an
Lead acid battery
The most important use of lead dioxide is as the cathode of
- Pb + PbO2 + 2 HSO−4 + 2 H+ → 2 PbSO4 + 2 H2O E° = +2.05 V
Safety
Lead compounds are poisons.[14] Lead dioxide is a strong oxidizer, so any contact of skin, eyes with either lead dioxide or its vapours may cause severe injury in the form of burns which can even lead to death.
PbO2 is not combustible, but it enhances flammability of other substances and the intensity of the fire. In case of a fire it gives off irritating and toxic fumes.[15]
Lead dioxide and other lead compound pose a huge environmental hazard when they are not disposed of properly. it is especially poisonous to aquatic life.[16]
References
- ISSN 0021-9584.
- ^ ISBN 978-3-11-011451-5.
- ^ Haidinger, W. (1845). "Zweite Klasse: Geogenide. II. Ordnung. Baryte VII. Bleibaryt. Plattnerit.". Handbuch der Bestimmenden Mineralogie (PDF) (in German). Vienna: Braumüller & Seidel. p. 500.
- ^ a b c Taggard, J. E. Jr.; et al. (1988). "Scrutinyite, natural occurrence of α-PbO2 from Bingham, New Mexico, U.S.A., and Mapimi, Mexico" (PDF). Canadian Mineralogist. 26: 905.
- .
- ^ ISBN 978-0-08-037941-8.
- ISBN 978-81-224-1384-7.
- ISBN 978-0-906048-26-9.
- ^ M. Baulder (1963). "Lead(IV) Oxide". In G. Brauer (ed.). Handbook of Preparative Inorganic Chemistry, 2nd Ed. Vol. 1. NY, NY: Academic Press. p. 758.
- ISBN 978-3-11-017770-1.
- ^ Sutcliffe, Arthur (1930). Practical Chemistry for Advanced Students (1949 ed.). London: John Murray.
- ^ "Plattnerite: Plattnerite mineral information and data". www.mindat.org. Retrieved 12 April 2018.
- ^ ISBN 978-1-84628-668-1.
- ^ "LEAD DIOXIDE". hazard.com. Retrieved 12 April 2018.
- ^ PubChem. "Lead dioxide". pubchem.ncbi.nlm.nih.gov. Retrieved 2022-12-15.
- ^ "Product and Company Identification" (PDF). ltschem.com. Retrieved 29 February 2024.

