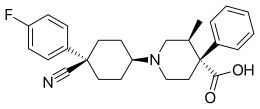
Levocabastine
 | |
| Clinical data | |
|---|---|
| Trade names | Livostin |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | Ophthalmic, intranasal[1] |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Levocabastine (trade name Livostin or Livocab, depending on the region) is a selective second-generation
Janssen Pharmaceutica in 1979. It is used for allergic conjunctivitis.[3]
As well as acting as an antihistamine, levocabastine has also subsequently been found to act as a potent and selective antagonist for the neurotensin receptor NTS2, and was the first drug used to characterise the different neurotensin subtypes.[4][5] This has made it a useful tool for the study of this receptor.[6][7][8][9][10]
The
pharmaceutical drug Bilina is a combination of Levocabastine, benzalkonium chloride, and other components and is typically used in a 0.5 mg/ml suspension as eye-drops, dispensed in 4ml bottles for the treatment of allergic conjunctivitis or similar allergic ocular conditions.[11]
References
- ^ "Livostin Nasal Spray". RxMed: Pharmaceutical Information. Retrieved 13 November 2005.
- ^ "Livostin - levocabastine hydrochloride suspension". DailyMed. U.S. National Library of Medicine. Retrieved 4 January 2016.
- S2CID 8681108.
- S2CID 23692347.
- PMID 2888670.
- S2CID 5802578.
- PMID 8795617.
- PMID 16148226.
- S2CID 24790151.
- S2CID 19774998.
- ^ "Levocabastine ophthalmic". vademecum.es. Retrieved 11 September 2014.
External links
- "Levocabastine". Drug Information Portal. U.S. National Library of Medicine.
- "Levocabastine hydrochloride". Drug Information Portal. U.S. National Library of Medicine.
