Lignin
Lignin is a class of complex
History
Lignin was first mentioned in 1813 by the Swiss botanist
Lignin is present in red algae, which suggest that the common ancestor of plants and red algae also synthesised lignin. This finding also suggests that the original function of lignin was structural as it plays this role in the red alga Calliarthron, where it supports joints between calcified segments.[7]
Composition and structure
The composition of lignin varies from species to species. An example of composition from an aspen[8] sample is 63.4% carbon, 5.9% hydrogen, 0.7% ash (mineral components), and 30% oxygen (by difference),[9] corresponding approximately to the formula (C31H34O11)n.
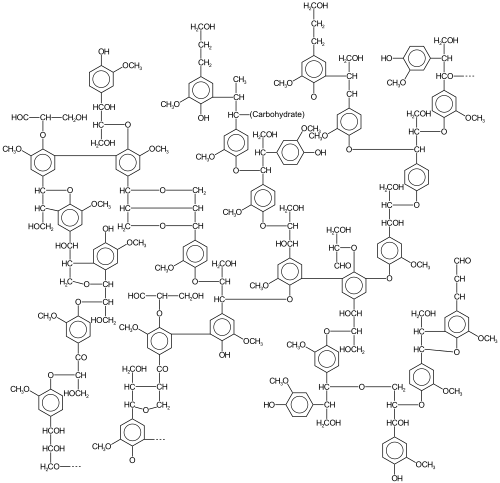
Lignin is a collection of highly
The relative amounts of the precursor "monomers" (lignols or monolignols) vary according to the plant source.
Lignin's

Many grasses have mostly G, while some palms have mainly S.[13] All lignins contain small amounts of incomplete or modified monolignols, and other monomers are prominent in non-woody plants.[14]
Biological function
Lignin fills the spaces in the cell wall between cellulose, hemicellulose, and pectin components, especially in vascular and support tissues: xylem tracheids, vessel elements and sclereid cells.[citation needed]
Lignin plays a crucial part in conducting water and aqueous nutrients in
It is
Finally, lignin also confers disease resistance by accumulating at the site of pathogen infiltration, making the plant cell less accessible to cell wall degradation.[20]
Economic significance

Global commercial production of lignin is a consequence of papermaking. In 1988, more than 220 million tons of paper were produced worldwide.[21] Much of this paper was delignified; lignin comprises about 1/3 of the mass of lignocellulose, the precursor to paper. Lignin is an impediment to papermaking as it is colored, it yellows in air, and its presence weakens the paper. Once separated from the cellulose, it is burned as fuel. Only a fraction is used in a wide range of low volume applications where the form but not the quality is important.[22]
Mechanical, or high-yield
In
Lignin removed by the
Given that it is the most prevalent biopolymer after cellulose, lignin has been investigated as a feedstock for biofuel production and can become a crucial plant extract in the development of a new class of biofuels.[27][28]
Biosynthesis
Lignin
The
Biodegradation
In contrast to other bio-polymers (e.g. proteins, DNA, and even cellulose), lignin resists degradation. It is immune to both acid- and base-catalyzed hydrolysis. The degradability varies with species and plant tissue type. For example, syringyl (S) lignin is more susceptible to degradation by fungal decay as it has fewer aryl-aryl bonds and a lower redox potential than guaiacyl units.[31][32] Because it is cross-linked with the other cell wall components, lignin minimizes the accessibility of cellulose and hemicellulose to microbial enzymes, leading to a reduced digestibility of biomass.[15]
Some ligninolytic enzymes include heme peroxidases such as lignin peroxidases, manganese peroxidases, versatile peroxidases, and dye-decolourizing peroxidases as well as copper-based laccases. Lignin peroxidases oxidize non-phenolic lignin, whereas manganese peroxidases only oxidize the phenolic structures. Dye-decolorizing peroxidases, or DyPs, exhibit catalytic activity on a wide range of lignin model compounds, but their in vivo substrate is unknown. In general, laccases oxidize phenolic substrates but some fungal laccases have been shown to oxidize non-phenolic substrates in the presence of synthetic redox mediators.[33][34]
Lignin degradation by fungi
Well-studied ligninolytic enzymes are found in
Lignin degradation by bacteria
Bacteria lack most of the enzymes employed by fungi to degrade lignin, and lignin derivatives (aliphatic acids, furans, and solubilized phenolics) inhibit the growth of bacteria.
In the environment, lignin can be degraded either biotically via bacteria or abiotically via photochemical alteration, and oftentimes the latter assists in the former.
Pyrolysis
Chemical analysis
The conventional method for lignin quantitation in the pulp industry is the Klason lignin and acid-soluble lignin test, which is standardized procedures. The cellulose is digested thermally in the presence of acid. The residue is termed Klason lignin. Acid-soluble lignin (ASL) is quantified by the intensity of its
A solution of hydrochloric acid and
Thermochemolysis (chemical break down of a substance under vacuum and at high temperature) with tetramethylammonium hydroxide (TMAH) or cupric oxide[46] has also been used to characterize lignins. The ratio of syringyl lignol (S) to vanillyl lignol (V) and cinnamyl lignol (C) to vanillyl lignol (V) is variable based on plant type and can therefore be used to trace plant sources in aquatic systems (woody vs. non-woody and angiosperm vs. gymnosperm).[47] Ratios of carboxylic acid (Ad) to aldehyde (Al) forms of the lignols (Ad/Al) reveal diagenetic information, with higher ratios indicating a more highly degraded material.[31][32] Increases in the (Ad/Al) value indicate an oxidative cleavage reaction has occurred on the alkyl lignin side chain which has been shown to be a step in the decay of wood by many white-rot and some soft rot fungi.[31][32][48][49][50]
Lignin and its models have been well examined by 1H and 13C NMR spectroscopy. Owing to the structural complexity of lignins, the spectra are poorly resolved and quantitation is challenging.[51]
References
- ^ ISBN 978-3527306732.
- ISBN 978-0-471-23896-6. Retrieved 2007-10-14.
- ^ de Candolle, M.A.P. (1813). Theorie Elementaire de la Botanique ou Exposition des Principes de la Classification Naturelle et de l'Art de Decrire et d'Etudier les Vegetaux. Paris: Deterville. See p. 417.
- ^ ISBN 978-0-12-647480-0.
- ^ PMID 14503002.
- ^ "Lignin". Encyclopedia Brittanica. 2023-10-05. Retrieved 2023-10-26.
- S2CID 17409200.
- ^ In the referenced article, the species of aspen is not specified, only that it was from Canada.
- ^ Hsiang-Hui King; Peter R. Solomon; Eitan Avni; Robert W. Coughlin (Fall 1983). "Modeling Tar Composition in Lignin Pyrolysis" (PDF). Symposium on Mathematical Modeling of Biomass Pyrolysis Phenomena, Washington, D.C., 1983. p. 1. Archived from the original (PDF) on 2017-08-08. Retrieved 2024-01-29.
- S2CID 236200196.
- PMID 11449052.
- ^ "Lignin and its Properties: Glossary of Lignin Nomenclature". Dialogue/Newsletters Volume 9, Number 1. Lignin Institute. July 2001. Archived from the original on 2007-10-09. Retrieved 2007-10-14.
- S2CID 27962271.
- PMID 11423146. Archived from the originalon 2021-04-28. Retrieved 2018-12-29.
- ^ a b K.V. Sarkanen & C.H. Ludwig, eds. (1971). Lignins: Occurrence, Formation, Structure, and Reactions. New York: Wiley Intersci.
- PMID 11722770.
- ISBN 978-0030500039.
- ISBN 978-0471245209.
- .
- PMID 19649200.
- ISBN 978-3527306732.
- ^ Higson, A; Smith, C (25 May 2011). "NNFCC Renewable Chemicals Factsheet: Lignin". Archived from the original on 20 July 2011.
- ^ "Uses of lignin from sulfite pulping". Archived from the original on 2007-10-09. Retrieved 2007-09-10.
- ISBN 978-3527306732.
- ISBN 978-3527306732.
- ^ "Frost & Sullivan: Full Speed Ahead for the Lignin Market with High-Value Opportunities as early as 2017".
- ^ Folkedahl, Bruce (2016), "Cellulosic ethanol: what to do with the lignin", Biomass, retrieved 2016-08-10.
- ^ Abengoa (2016-04-21), The importance of lignin for ethanol production, retrieved 2016-08-10.
- S2CID 20529001.
- PMID 16023847.
- ^ PMID 12568554.
- ^ .
- ^ OCLC 841913543.
- ^ PMID 27544286.
- S2CID 8767248.
- PMID 28363966.
- PMID 20400590.
- . Retrieved 2018-11-27.
- ^ "Persistence of Soil Organic Matter as an Ecosystem Property". ResearchGate. Retrieved 2018-11-27.
- ISBN 978-0124059405.
- ISBN 978-0-8412-1346-3.
- ^ "TAPPI. T 222 om-02 – Acid-insoluble lignin in wood and pulp" (PDF).
- ^ Harkin, John M. (November 1966). "Lignin production and detection in wood" (PDF). U.S. Forest Service Research. Note FPL-0148. Archived from the original (PDF) on 2020-03-05. Retrieved 2012-12-30.
- PMID 12228544.
- S2CID 95157574.
- ISSN 0003-2700.
- ISSN 0016-7037.
- .
- PMID 11409955.
- .
- ISBN 978-1574444865.
Further reading
- Freudenberg, K. & Nash, A. C., eds. (1968). Constitution and Biosynthesis of Lignin. Berlin: Springer-Verlag.
