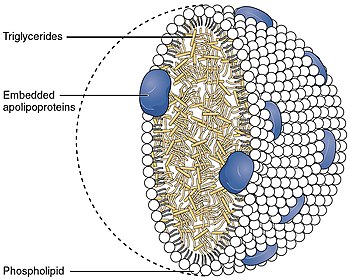
Lipid
Lipids are a broad group of organic compounds which include
Lipids may be broadly defined as
Although the term "lipid" is sometimes used as a synonym for fats, fats are a subgroup of lipids called triglycerides. Lipids also encompass molecules such as fatty acids and their derivatives (including tri-, di-, monoglycerides, and phospholipids), as well as other sterol-containing metabolites such as cholesterol.[6] Although humans and other mammals use various biosynthetic pathways both to break down and to synthesize lipids, some essential lipids cannot be made this way and must be obtained from the diet.
History
In 1815, Henri Braconnot classified lipids (graisses) in two categories, suifs (solid greases or tallow) and huiles (fluid oils).[7] In 1823, Michel Eugène Chevreul developed a more detailed classification, including oils, greases, tallow, waxes, resins, balsams and volatile oils (or essential oils).[8][9][10]
The first synthetic triglyceride was reported by
In 1827, William Prout recognized fat ("oily" alimentary matters), along with protein ("albuminous") and carbohydrate ("saccharine"), as an important nutrient for humans and animals.[13][14]
For a century, chemists regarded "fats" as only simple lipids made of fatty acids and glycerol (glycerides), but new forms were described later.
The terms lipoid, lipin, lipide and lipid have been used with varied meanings from author to author.
The word lipide, which stems etymologically from Greek λίπος, lipos 'fat', was introduced in 1923 by the French pharmacologist Gabriel Bertrand.[19] Bertrand included in the concept not only the traditional fats (glycerides), but also the "lipoids", with a complex constitution.[9] The word lipide was unanimously approved by the international commission of the Société de Chimie Biologique during the plenary session on July 3, 1923. The word lipide was later anglicized as lipid because of its pronunciation ('lɪpɪd). In French, the suffix -ide, from Ancient Greek -ίδης (meaning 'son of' or 'descendant of'), is always pronounced (ɪd).
In 1947, T. P. Hilditch defined "simple lipids" as greases and waxes (true waxes, sterols, alcohols).[20][page needed]
Categories
Lipids have been classified into eight categories by the Lipid MAPS consortium[3] as follows:
Fatty acyls


Fatty acyls, a generic term for describing fatty acids, their conjugates and derivatives, are a diverse group of molecules synthesized by chain-elongation of an
Examples of biologically important fatty acids include the
Glycerolipids
Glycerolipids are composed of mono-, di-, and tri-substituted glycerols,[30] the best-known being the fatty acid triesters of glycerol, called triglycerides. The word "triacylglycerol" is sometimes used synonymously with "triglyceride". In these compounds, the three hydroxyl groups of glycerol are each esterified, typically by different fatty acids. Because they function as an energy store, these lipids comprise the bulk of storage fat in animal tissues. The hydrolysis of the ester bonds of triglycerides and the release of glycerol and fatty acids from adipose tissue are the initial steps in metabolizing fat.[31]: 630–1
Additional subclasses of glycerolipids are represented by glycosylglycerols, which are characterized by the presence of one or more
Glycerophospholipids
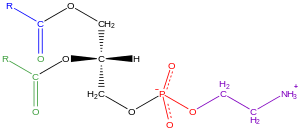
Glycerophospholipids, usually referred to as
Examples of glycerophospholipids found in biological membranes are phosphatidylcholine (also known as PC, GPCho or lecithin), phosphatidylethanolamine (PE or GPEtn) and phosphatidylserine (PS or GPSer). In addition to serving as a primary component of cellular membranes and binding sites for intra- and intercellular proteins, some glycerophospholipids in eukaryotic cells, such as phosphatidylinositols and phosphatidic acids are either precursors of or, themselves, membrane-derived second messengers.[31]: 844 Typically, one or both of these hydroxyl groups are acylated with long-chain fatty acids, but there are also alkyl-linked and 1Z-alkenyl-linked (plasmalogen) glycerophospholipids, as well as dialkylether variants in archaebacteria.[38]
Sphingolipids
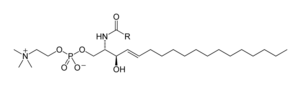
Sphingolipids are a complicated family of compounds
The major phosphosphingolipids of mammals are sphingomyelins (ceramide phosphocholines),[40] whereas insects contain mainly ceramide phosphoethanolamines[41] and fungi have phytoceramide phosphoinositols and mannose-containing headgroups.[42] The glycosphingolipids are a diverse family of molecules composed of one or more sugar residues linked via a glycosidic bond to the sphingoid base. Examples of these are the simple and complex glycosphingolipids such as cerebrosides and gangliosides.
Sterols

Sterols, such as
Sterols are
Prenols

Saccharolipids
Polyketides
Polyketides are synthesized by polymerization of
Biological functions
Component of biological membranes
Plant thylakoid membranes have the largest lipid component of a non-bilayer forming monogalactosyl diglyceride (MGDG), and little phospholipids; despite this unique lipid composition, chloroplast thylakoid membranes have been shown to contain a dynamic lipid-bilayer matrix as revealed by magnetic resonance and electron microscope studies.[59]
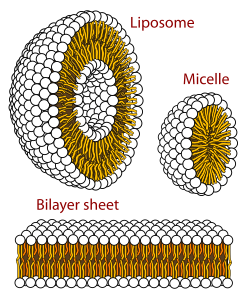
A biological membrane is a form of
The formation of lipids into protocell membranes represents a key step in models of abiogenesis, the origin of life.[64]
Energy storage
Triglycerides, stored in adipose tissue, are a major form of energy storage both in animals and plants. They are a major source of energy in aerobic respiration. The complete oxidation of fatty acids releases about 38 kJ/g (9 kcal/g), compared with only 17 kJ/g (4 kcal/g) for the oxidative breakdown of carbohydrates and proteins. The adipocyte, or fat cell, is designed for continuous synthesis and breakdown of triglycerides in animals, with breakdown controlled mainly by the activation of hormone-sensitive enzyme lipase.[65] Migratory birds that must fly long distances without eating use triglycerides to fuel their flights.[2]: 619
Signaling
Evidence has emerged showing that
Other functions
The "fat-soluble" vitamins (
Metabolism
The major dietary lipids for humans and other animals are animal and plant triglycerides, sterols, and membrane phospholipids. The process of lipid metabolism synthesizes and degrades the lipid stores and produces the structural and functional lipids characteristic of individual tissues.
Biosynthesis
In animals, when there is an oversupply of dietary carbohydrate, the excess carbohydrate is converted to triglycerides. This involves the synthesis of fatty acids from
The synthesis of
Triglyceride synthesis takes place in the endoplasmic reticulum by metabolic pathways in which acyl groups in fatty acyl-CoAs are transferred to the hydroxyl groups of glycerol-3-phosphate and diacylglycerol.[2]: 733–9
Degradation
Nutrition and health
Most of the fat found in food is in the form of triglycerides, cholesterol, and phospholipids. Some dietary fat is necessary to facilitate absorption of fat-soluble vitamins (
In contrast, it is now well-established that consumption of
A few studies have suggested that total dietary fat intake is linked to an increased risk of obesity.
See also
- Solid lipid nanoparticle – Novel drug delivery system
- Simple lipid
- Emulsion test
- Lipid microdomain
- Membrane lipid – Lipid molecules on cell membrane
- Lipidomics – large scale study of an organism's lipid metabolism using high-end chemical analysis techniques
- Lipidome – Totality of lipids in cells
- Protein–lipid interaction
- Phenolic lipid – Class of organic compounds, a class of natural products composed of long aliphatic chains and phenolic rings that occur in plants, fungi and bacteria
References
- ISBN 978-0-393-97378-5.
- ^ ISBN 978-0-7167-8724-2.
- ^ PMID 19098281.
- PMID 21939287.
- PMID 23429269.

- ISBN 978-0-13-981176-0.
- ^ Braconnot H (31 March 1815). "Sur la nature des corps gras". Annales de chimie. 2 (XCIII): 225–277.
- ^ Chevreul ME (1823). Recherches sur les corps gras d'origine animale. Paris: Levrault.
- ^ ISBN 978-1466551466.
- ISBN 978-1482242317.
- ^ Pelouze TJ, Gélis A (1844). "Mémoire sur l'acide butyrique". Annales de Chimie et de Physique. 10: 434.
- ^ Comptes rendus hebdomadaires des séances de l'Académie des Sciences, Paris, 1853, 36, 27; Annales de Chimie et de Physique 1854, 41, 216
- ^ Leray C. "Chronological history of lipid center". Cyberlipid Center. Archived from the original on 13 October 2017. Retrieved 1 December 2017.
- ^ Prout W (1827). "On the ultimate composition of simple alimentary substances, with some preliminary remarks on the analysis of organised bodies in general". Phil. Trans.: 355–388.
- ISBN 978-1483164793.
- ^ Rosenbloom J, Gies WJ (1911). "Suggestion to teachers of biochemistry. I. A proposed chemical classification of lipins, with a note on the intimate relation between cholesterols and bile salts". Biochem. Bull. 1: 51–56.
- S2CID 75844378.
- ISBN 978-0857097866.
- ^ Bertrand G (1923). "Projet de reforme de la nomenclature de Chimie biologique". Bulletin de la Société de Chimie Biologique. 5: 96–109.
- ^ Hilditch, Thomas Percy (1956). The Chemical Constitution of Natural Fats. Wiley.
- ISBN 978-0-444-51139-3.
- ISBN 978-0-12-373895-0.
- ISBN 978-0-314-04467-9.
- PMID 3428918.
- ^ ISBN 978-0-471-17053-2.
- S2CID 1625062.
- ^ Furse S (2 December 2011). "A Long Lipid, a Long Name: Docosahexaenoic Acid". The Lipid Chronicles.
- ^ "DHA for Optimal Brain and Visual Functioning". DHA/EPA Omega-3 Institute.
- PMID 18751909.
- PMID 14654091.
- ^ ISBN 978-0-8053-3931-4.
- ^ PMID 17599463.
- S2CID 2678053.
- ^ "The Structure of a Membrane". The Lipid Chronicles. 5 November 2011. Retrieved 31 December 2011.
- S2CID 26822092.
- PMID 10878232.
- PMID 17954212.
- PMID 7859340.
- ISBN 978-0-444-51138-6.
- PMID 8415797.
- PMID 1739742.
- PMID 18508430.
- PMID 12648773.
- PMID 12543708.
- .
- ISBN 978-1-4051-3066-0.
- PMID 17161336.
- ^ PMID 12735695.
- PMID 17349800.
- PMID 2691341.
- PMID 16019076.
- ^ PMID 16479018.

- S2CID 44858908.
- PMID 18473889.
- PMID 18387369.
- S2CID 235708094.
- ISBN 978-0-9514171-6-4
- S2CID 232761961.
- S2CID 360223.
- PMID 18216768.
- PMID 17051225.
- PMID 2087221.
- PMID 15867152.
- S2CID 10959497. Archived from the original(PDF) on 11 September 2008. Retrieved 15 March 2015.
- PMID 17878492.
- PMID 21743455.
- S2CID 31357937.
- PMID 15134755.
- PMID 21505029.
- S2CID 9194419.
- PMID 18951299.
- PMID 18751921.
- PMID 17981624.
- PMID 18691060.
- PMID 19035881.
- S2CID 23782770.
- PMID 1932043.
- PMID 375981.
- S2CID 7277949.
- S2CID 21937549.
- PMID 19237595.
- PMID 1550861.
- ^ "Steroids". Elmhurst. edu. Archived from the original on 23 October 2011. Retrieved 10 October 2013.
- S2CID 4043407.
- PMID 15952903.
- S2CID 46348092.
- PMID 16621811.
- PMID 15012203.
- ^ PMID 7023367.
- S2CID 4019443.
- ^ ISBN 978-0-12-095440-7.
- PMID 19022225.
- PMID 19328262.
- S2CID 20742062.
- PMID 18996687.
- PMID 18427401.
- S2CID 24998042.
- S2CID 34030743
- S2CID 260372605.
- PMID 18753397.
- PMID 16467233.

- PMID 16391215.

- ^ "The Nutrition Source". T. H. Chan School of Public Health. Harvard University.
- ^ "Fats and Cholesterol: Out with the Bad, In with the Good — What Should You Eat? – The Nutrition Source". Harvard School of Public Health.
Bibliography
- Bhagavan NV (2002). Medical Biochemistry. San Diego: Harcourt/Academic Press. ISBN 978-0-12-095440-7.
- Devlin TM (1997). Textbook of Biochemistry: With Clinical Correlations (4th ed.). Chichester: John Wiley & Sons. ISBN 978-0-471-17053-2.
- Stryer L, Berg JM, Tymoczko JL (2007). Biochemistry (6th ed.). San Francisco: W.H. Freeman. ISBN 978-0-7167-8724-2.
- van Holde KE, Mathews CK (1996). Biochemistry (2nd ed.). Menlo Park, California: Benjamin/Cummings Pub. Co. ISBN 978-0-8053-3931-4.
External links
Introductory
- List of lipid-related web sites
- Nature Lipidomics Gateway – Round-up and summaries of recent lipid research
- Lipid Library – General reference on lipid chemistry and biochemistry
- Cyberlipid.org – Resources and history for lipids.
- Molecular Computer Simulations – Modeling of Lipid Membranes
- Lipids, Membranes and Vesicle Trafficking – The Virtual Library of Biochemistry, Molecular Biology and Cell Biology
Nomenclature
Databases
- LIPID MAPS – Comprehensive lipid and lipid-associated gene/protein databases.
- LipidBank – Japanese database of lipids and related properties, spectral data and references.
General
- ApolloLipids – Provides dyslipidemia and cardiovascular disease prevention and treatment information as well as continuing medical education programs
- National Lipid Association – Professional medical education organization for health care professionals who seek to prevent morbidity and mortality stemming from dyslipidemias and other cholesterol-related disorders.