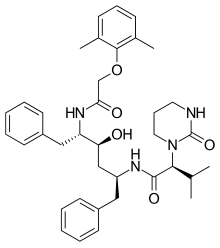
Lopinavir
 | |
 | |
| Clinical data | |
|---|---|
| Other names | ABT-378 |
| AHFS/Drugs.com | International Drug Names |
| MedlinePlus | a602015 |
| License data | |
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Unknown |
| Protein binding | 98-99% |
| Metabolism | Liver |
| Elimination half-life | 5 to 6 hours |
| Excretion | Mostly fecal |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Lopinavir is an anti
It was patented in 1995 and approved for medical use in 2000.
Side effects
Side effects, interactions, and contraindications have only been evaluated in the drug combination lopinavir/ritonavir. They include nausea, vomiting, and stomach aches.[citation needed]
Pharmacology
Lopinavir is highly bound to plasma proteins (98–99%).[4]
Reports are contradictory regarding lopinavir penetration into the cerebrospinal fluid (CSF). Anecdotal reports state that lopinavir cannot be detected in the CSF; however, a study of paired CSF-plasma samples from 26 patients receiving lopinavir/ritonavir found lopinavir CSF levels above the IC50 in 77% of samples.[5]
Research
A 2014 study indicates that lopinavir is effective against the
Lopinavir was found to inhibit
A long-acting injectable formulation of lopinavir is under clinical trial aiming at monthly dosing (NCT05850728).
References
- ^ "FDA Approved Drug Products: Kaletra". Retrieved 30 April 2004.
- ISBN 9783527607495.
- PMC 7689305.
- ^ Kaletra (lopinavir/ritonavir) capsules; (lopinavir/ritonavir) oral solution. Prescribing information. April 2009
- S2CID 3162858.
- ^ HIV drug used to reverse effects of virus that causes cervical cancer University of Manchester, 17 February 2014.
- PMID 34335531.
- PMID 25073485.
- PMID 24841269.
- PMID 32187464.
External links
- "Lopinavir". Drug Information Portal. U.S. National Library of Medicine.
