Magnetism

| Articles about |
| Electromagnetism |
|---|
 |
Magnetism is the class of physical attributes that occur through a magnetic field, which allows objects to attract or repel each other. Because both electric currents and magnetic moments of elementary particles give rise to a magnetic field, magnetism is one of two aspects of electromagnetism.
The most familiar effects occur in
All substances exhibit some type of magnetism. Magnetic materials are classified according to their bulk susceptibility.[1] Ferromagnetism is responsible for most of the effects of magnetism encountered in everyday life, but there are actually several types of magnetism. Paramagnetic substances, such as aluminium and oxygen, are weakly attracted to an applied magnetic field; diamagnetic substances, such as copper and carbon, are weakly repelled; while antiferromagnetic materials, such as chromium, have a more complex relationship with a magnetic field.[vague] The force of a magnet on paramagnetic, diamagnetic, and antiferromagnetic materials is usually too weak to be felt and can be detected only by laboratory instruments, so in everyday life, these substances are often described as non-magnetic.
The strength of a magnetic field always decreases with distance from the magnetic source,[2] though the exact mathematical relationship between strength and distance varies. Many factors can influence the magnetic field of an object including the magnetic moment of the material, the physical shape of the object, both the magnitude and direction of any electric current present within the object, and the temperature of the object.
History



Magnetism was first discovered in the ancient world when people noticed that
In ancient China, the earliest literary reference to magnetism lies in a 4th-century BC book named after its author, Guiguzi.[8] The 2nd-century BC annals, Lüshi Chunqiu, also notes: "The lodestone makes iron approach; some (force) is attracting it."[9] The earliest mention of the attraction of a needle is in a 1st-century work Lunheng (Balanced Inquiries): "A lodestone attracts a needle."[10] The 11th-century
In 1600,
An understanding of the relationship between electricity and magnetism began in 1819 with work by Hans Christian Ørsted, a professor at the University of Copenhagen, who discovered, by the accidental twitching of a compass needle near a wire, that an electric current could create a magnetic field. This landmark experiment is known as Ørsted's Experiment. Jean-Baptiste Biot and Félix Savart, both of whom in 1820 came up with the Biot–Savart law giving an equation for the magnetic field from a current-carrying wire. Around the same time, André-Marie Ampère carried out numerous systematic experiments and discovered that the magnetic force between two DC current loops of any shape is equal to the sum of the individual forces that each current element of one circuit exerts on each other current element of the other circuit.
In 1831, Michael Faraday discovered that a time-varying magnetic flux induces a voltage through a wire loop. In 1835, Carl Friedrich Gauss hypothesized, based on Ampère's force law in its original form, that all forms of magnetism arise as a result of elementary point charges moving relative to each other.[12] Wilhelm Eduard Weber advanced Gauss' theory to Weber electrodynamics.
From around 1861,
Electromagnetism has continued to develop into the 21st century, being incorporated into the more fundamental theories of
Sources
Magnetism, at its root, arises from three sources:
- Electric current
- elementary particles
- Changing electric fields
The magnetic properties of materials are mainly due to the magnetic moments of their atoms' orbiting electrons. The magnetic moments of the nuclei of atoms are typically thousands of times smaller than the electrons' magnetic moments, so they are negligible in the context of the magnetization of materials. Nuclear magnetic moments are nevertheless very important in other contexts, particularly in nuclear magnetic resonance (NMR) and magnetic resonance imaging (MRI).
Ordinarily, the enormous number of electrons in a material are arranged such that their magnetic moments (both orbital and intrinsic) cancel out. This is due, to some extent, to electrons combining into pairs with opposite intrinsic magnetic moments as a result of the
Sometimes—either spontaneously, or owing to an applied external magnetic field—each of the electron magnetic moments will be, on average, lined up. A suitable material can then produce a strong net magnetic field.
The magnetic behavior of a material depends on its structure, particularly its
Types
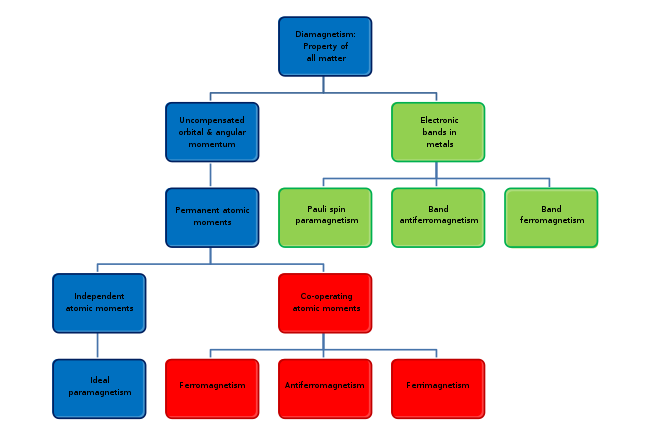
Diamagnetism
Diamagnetism appears in all materials and is the tendency of a material to oppose an applied magnetic field, and therefore, to be repelled by a magnetic field. However, in a material with paramagnetic properties (that is, with a tendency to enhance an external magnetic field), the paramagnetic behavior dominates.[15] Thus, despite its universal occurrence, diamagnetic behavior is observed only in a purely diamagnetic material. In a diamagnetic material, there are no unpaired electrons, so the intrinsic electron magnetic moments cannot produce any bulk effect. In these cases, the magnetization arises from the electrons' orbital motions, which can be understood classically as follows:
When a material is put in a magnetic field, the electrons circling the nucleus will experience, in addition to their Coulomb attraction to the nucleus, a Lorentz force from the magnetic field. Depending on which direction the electron is orbiting, this force may increase the centripetal force on the electrons, pulling them in towards the nucleus, or it may decrease the force, pulling them away from the nucleus. This effect systematically increases the orbital magnetic moments that were aligned opposite the field and decreases the ones aligned parallel to the field (in accordance with Lenz's law). This results in a small bulk magnetic moment, with an opposite direction to the applied field.
This description is meant only as a heuristic; the Bohr–Van Leeuwen theorem shows that diamagnetism is impossible according to classical physics, and that a proper understanding requires a quantum-mechanical description.
All materials undergo this orbital response. However, in paramagnetic and ferromagnetic substances, the diamagnetic effect is overwhelmed by the much stronger effects caused by the unpaired electrons.
Paramagnetism
In a paramagnetic material there are unpaired electrons; i.e., atomic or molecular orbitals with exactly one electron in them. While paired electrons are required by the Pauli exclusion principle to have their intrinsic ('spin') magnetic moments pointing in opposite directions, causing their magnetic fields to cancel out, an unpaired electron is free to align its magnetic moment in any direction. When an external magnetic field is applied, these magnetic moments will tend to align themselves in the same direction as the applied field, thus reinforcing it.
Ferromagnetism
A ferromagnet, like a paramagnetic substance, has unpaired electrons. However, in addition to the electrons' intrinsic magnetic moment's tendency to be parallel to an applied field, there is also in these materials a tendency for these magnetic moments to orient parallel to each other to maintain a lowered-energy state. Thus, even in the absence of an applied field, the magnetic moments of the electrons in the material spontaneously line up parallel to one another.
Every ferromagnetic substance has its own individual temperature, called the Curie temperature, or Curie point, above which it loses its ferromagnetic properties. This is because the thermal tendency to disorder overwhelms the energy-lowering due to ferromagnetic order.
Ferromagnetism only occurs in a few substances; common ones are
Magnetic domains
The magnetic moments of atoms in a
When a domain contains too many molecules, it becomes unstable and divides into two domains aligned in opposite directions so that they stick together more stably.
When exposed to a magnetic field, the domain boundaries move, so that the domains aligned with the magnetic field grow and dominate the structure (dotted yellow area), as shown at the left. When the magnetizing field is removed, the domains may not return to an unmagnetized state. This results in the ferromagnetic material's being magnetized, forming a permanent magnet.
When magnetized strongly enough that the prevailing domain overruns all others to result in only one single domain, the material is
Antiferromagnetism
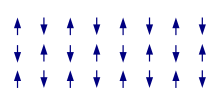
In an
In some materials, neighboring electrons prefer to point in opposite directions, but there is no geometrical arrangement in which each pair of neighbors is anti-aligned. This is called a canted antiferromagnet or spin ice and is an example of geometrical frustration.
Ferrimagnetism
Like ferromagnetism, ferrimagnets retain their magnetization in the absence of a field. However, like antiferromagnets, neighboring pairs of electron spins tend to point in opposite directions. These two properties are not contradictory, because in the optimal geometrical arrangement, there is more magnetic moment from the sublattice of electrons that point in one direction, than from the sublattice that points in the opposite direction.
Most ferrites are ferrimagnetic. The first discovered magnetic substance, magnetite, is a ferrite and was originally believed to be a ferromagnet; Louis Néel disproved this, however, after discovering ferrimagnetism.
Superparamagnetism
When a ferromagnet or ferrimagnet is sufficiently small, it acts like a single magnetic spin that is subject to Brownian motion. Its response to a magnetic field is qualitatively similar to the response of a paramagnet, but much larger.
Nagaoka magnetism
Japanese physicist Yosuke Nagaoka conceived of a type of magnetism in a square, two-dimensional lattice where every lattice node had one electron. If one electron was removed under specific conditions, the lattice's energy would be minimal only when all electrons' spins were parallel.
A variation on this was achieved experimentally by arranging the atoms in a triangular moiré lattice of molybdenum diselenide and tungsten disulfide monolayers. Applying a weak magnetic field and a voltage led to ferromagnetic behavior when 100-150% more electrons than lattice nodes were present. The extra electrons delocalized and paired with lattice electrons to form doublons. Delocalization was prevented unless the lattice electrons had aligned spins. The doublons thus created localized ferromagnetic regions. The phenomenon took place at 140 millikelvins.[16]
Other types of magnetism
Electromagnet
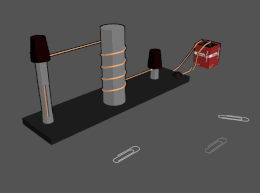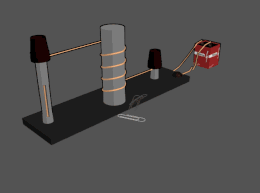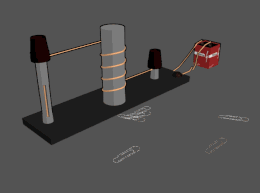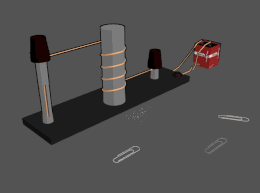
An
The main advantage of an electromagnet over a
Electromagnets are widely used as components of other electrical devices, such as
Magnetism, electricity, and special relativity
As a consequence of Einstein's theory of special relativity, electricity and magnetism are fundamentally interlinked. Both magnetism lacking electricity, and electricity without magnetism, are inconsistent with special relativity, due to such effects as
All observations on electromagnetism apply to what might be considered to be primarily magnetism, e.g. perturbations in the magnetic field are necessarily accompanied by a nonzero electric field, and propagate at the speed of light.[21]
Magnetic fields in a material
In vacuum,
where μ0 is the vacuum permeability.
In a material,
The quantity μ0M is called magnetic polarization.
If the field H is small, the response of the magnetization M in a
the constant of proportionality being called the magnetic susceptibility. If so,
In a hard magnet such as a ferromagnet, M is not proportional to the field and is generally nonzero even when H is zero (see Remanence).
Magnetic force
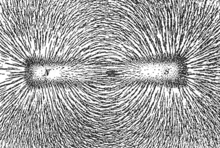
The phenomenon of magnetism is "mediated" by the magnetic field. An electric current or magnetic dipole creates a magnetic field, and that field, in turn, imparts magnetic forces on other particles that are in the fields.
Maxwell's equations, which simplify to the
The same situations that create magnetic fields—charge moving in a current or in an atom, and intrinsic magnetic dipoles—are also the situations in which a magnetic field has an effect, creating a force. Following is the formula for moving charge; for the forces on an intrinsic dipole, see magnetic dipole.
When a charged particle moves through a magnetic field B, it feels a Lorentz force F given by the cross product:[22]
where
- is the electric charge of the particle, and
- v is the vectorof the particle
Because this is a cross product, the force is
where is the angle between v and B.
One tool for determining the direction of the velocity vector of a moving charge, the magnetic field, and the force exerted is labeling the index finger "V"[dubious ], the middle finger "B", and the thumb "F" with your right hand. When making a gun-like configuration, with the middle finger crossing under the index finger, the fingers represent the velocity vector, magnetic field vector, and force vector, respectively. See also right-hand rule.
Magnetic dipoles
A very common source of magnetic field found in nature is a
Magnetic monopoles
Since a bar magnet gets its ferromagnetism from electrons distributed evenly throughout the bar, when a bar magnet is cut in half, each of the resulting pieces is a smaller bar magnet. Even though a magnet is said to have a north pole and a south pole, these two poles cannot be separated from each other. A monopole—if such a thing exists—would be a new and fundamentally different kind of magnetic object. It would act as an isolated north pole, not attached to a south pole, or vice versa. Monopoles would carry "magnetic charge" analogous to electric charge. Despite systematic searches since 1931, as of 2010[update], they have never been observed, and could very well not exist.[23]
Nevertheless, some
Certain
Units
SI
| Symbol[25] | Name of quantity | Unit name | Symbol | Base units |
|---|---|---|---|---|
| E | energy | joule
|
J = C⋅V = W⋅s | kg⋅m2⋅s−2 |
| Q | electric charge | coulomb | C | A⋅s |
| I | electric current | ampere | A = C/s = W/V | A |
| J | electric current density | ampere per square metre | A/m2 | A⋅m−2 |
| U, ΔV; Δϕ; E, ξ | potential difference; voltage; electromotive force
|
volt | V = J/C | kg⋅m2⋅s−3⋅A−1 |
| R; Z; X | reactance
|
ohm
|
Ω = V/A | kg⋅m2⋅s−3⋅A−2 |
| ρ | resistivity
|
ohm metre
|
Ω⋅m | kg⋅m3⋅s−3⋅A−2 |
| P | electric power | watt | W = V⋅A | kg⋅m2⋅s−3 |
| C | capacitance | farad | F = C/V | kg−1⋅m−2⋅A2⋅s4 |
| ΦE | electric flux | volt metre | V⋅m | kg⋅m3⋅s−3⋅A−1 |
| E | electric field strength | volt per metre | V/m = N/C | kg⋅m⋅A−1⋅s−3 |
| D | electric displacement field | coulomb per square metre | C/m2 | A⋅s⋅m−2 |
| ε | permittivity | farad per metre | F/m | kg−1⋅m−3⋅A2⋅s4 |
| χe | electric susceptibility | (dimensionless) | 1 | 1 |
| p | electric dipole moment | coulomb metre | C⋅m | A⋅s⋅m |
| G; Y; B | conductance; admittance; susceptance
|
siemens | S = Ω−1 | kg−1⋅m−2⋅s3⋅A2 |
| κ, γ, σ | conductivity
|
siemens per metre | S/m | kg−1⋅m−3⋅s3⋅A2 |
| B | magnetic flux density, magnetic induction | tesla | T = Wb/m2 = N⋅A−1⋅m−1 | kg⋅s−2⋅A−1 |
| Φ, ΦM, ΦB | magnetic flux | weber | Wb = V⋅s | kg⋅m2⋅s−2⋅A−1 |
| H | magnetic field strength | ampere per metre | A/m | A⋅m−1 |
| F | magnetomotive force | ampere | A = Wb/H | A |
| R | magnetic reluctance | inverse henry | H−1 = A/Wb | kg−1⋅m−2⋅s2⋅A2 |
| P | magnetic permeance
|
henry | H = Wb/A | kg⋅m2⋅s-2⋅A-2 |
| L, M | inductance | henry | H = Wb/A = V⋅s/A | kg⋅m2⋅s−2⋅A−2 |
| μ | permeability | henry per metre | H/m | kg⋅m⋅s−2⋅A−2 |
| χ | magnetic susceptibility | (dimensionless) | 1 | 1 |
| m | magnetic dipole moment
|
square meter
|
A⋅m2 = J⋅T−1 | A⋅m2 |
| σ | mass magnetization | square meter per kilogram
|
A⋅m2/kg | A⋅m2⋅kg−1 |
Other
- unitof magnetic field (denoted B).
- oersted – the CGS unit of magnetizing field (denoted H)
- maxwell – the CGS unit for magnetic flux
- gamma – a unit of magnetic flux density that was commonly used before the tesla came into use (1.0 gamma = 1.0 nanotesla)
- μ0 – common symbol for the permeability of free space (4π × 10−7 newton/(ampere-turn)2)
Living things
Some
Interpretation of magnetism by means of relative velocities
In the years after 1820,
Gauss's force law states that the electromagnetic force experienced by a point charge, with trajectory , in the vicinity of another point charge, with trajectory , in a vacuum is equal to the central force
- ,
where is the distance between the charges and is the relative velocity. Wilhelm Eduard Weber confirmed Gauss's hypothesis in numerous experiments.[28][29][30] By means of Weber electrodynamics it is possible to explain the static and quasi-static effects in the non-relativistic regime of classical electrodynamics without magnetic field and Lorentz force.
Since 1870,
Quantum-mechanical origin of magnetism
While heuristic explanations based on classical physics can be formulated, diamagnetism, paramagnetism and ferromagnetism can be fully explained only using quantum theory.[32][33] A successful model was developed already in 1927, by Walter Heitler and Fritz London, who derived, quantum-mechanically, how hydrogen molecules are formed from hydrogen atoms, i.e. from the atomic hydrogen orbitals and centered at the nuclei A and B, see below. That this leads to magnetism is not at all obvious, but will be explained in the following.
According to the Heitler–London theory, so-called two-body molecular -orbitals are formed, namely the resulting orbital is:
Here the last product means that a first electron, r1, is in an atomic hydrogen-orbital centered at the second nucleus, whereas the second electron runs around the first nucleus. This "exchange" phenomenon is an expression for the quantum-mechanical property that particles with identical properties cannot be distinguished. It is specific not only for the formation of chemical bonds, but also for magnetism. That is, in this connection the term exchange interaction arises, a term which is essential for the origin of magnetism, and which is stronger, roughly by factors 100 and even by 1000, than the energies arising from the electrodynamic dipole-dipole interaction.
As for the spin function , which is responsible for the magnetism, we have the already mentioned Pauli's principle, namely that a symmetric orbital (i.e. with the + sign as above) must be multiplied with an antisymmetric spin function (i.e. with a − sign), and vice versa. Thus:
- ,
I.e., not only and must be substituted by α and β, respectively (the first entity means "spin up", the second one "spin down"), but also the sign + by the − sign, and finally ri by the discrete values si (= ±1⁄2); thereby we have and . The "singlet state", i.e. the − sign, means: the spins are antiparallel, i.e. for the solid we have antiferromagnetism, and for two-atomic molecules one has diamagnetism. The tendency to form a (homoeopolar) chemical bond (this means: the formation of a symmetric molecular orbital, i.e. with the + sign) results through the Pauli principle automatically in an antisymmetric spin state (i.e. with the − sign). In contrast, the Coulomb repulsion of the electrons, i.e. the tendency that they try to avoid each other by this repulsion, would lead to an antisymmetric orbital function (i.e. with the − sign) of these two particles, and complementary to a symmetric spin function (i.e. with the + sign, one of the so-called "triplet functions"). Thus, now the spins would be parallel (ferromagnetism in a solid, paramagnetism in two-atomic gases).
The last-mentioned tendency dominates in the metals iron, cobalt and nickel, and in some rare earths, which are ferromagnetic. Most of the other metals, where the first-mentioned tendency dominates, are nonmagnetic (e.g. sodium, aluminium, and magnesium) or antiferromagnetic (e.g. manganese). Diatomic gases are also almost exclusively diamagnetic, and not paramagnetic. However, the oxygen molecule, because of the involvement of π-orbitals, is an exception important for the life-sciences.
The Heitler-London considerations can be generalized to the
The explanation of the phenomena is thus essentially based on all subtleties of quantum mechanics, whereas the electrodynamics covers mainly the phenomenology.
See also
- Coercivity
- Gravitomagnetism
- Magnetic hysteresis
- Magnetar
- Magnetic bearing
- Magnetic circuit
- Magnetic cooling
- Magnetic field viewing film
- Magnetic stirrer
- Switched-mode power supply
- Magnetic structure
- Micromagnetism
- Neodymium magnet
- Plastic magnet
- Rare-earth magnet
- Spin wave
- Spontaneous magnetization
- Vibrating-sample magnetometer
- Textbooks in electromagnetism
References
- )
- ISSN 1558-187X.
- ISBN 978-0-387-22967-6.
- ^ Platonis Opera, Meyer and Zeller, 1839, p. 989.
- ^ The location of Magnesia is debated; it could be the region in mainland Greece or Magnesia ad Sipylum. See, for example, "Magnet". Language Hat blog. 28 May 2005. Retrieved 22 March 2013.
- ^ Fowler, Michael (1997). "Historical Beginnings of Theories of Electricity and Magnetism". Retrieved 2008-04-02.
- ISBN 9781498761673.
- ^ The section "Fanying 2" (反應第二) of The Guiguzi: "其察言也,不失若磁石之取鍼,舌之取燔骨".
- S2CID 143585290.[...]: "La pierre d'aimant fait venir le fer ou elle l'attire."
un passage dans le Liu-che-tch'ouen-ts'ieou
From the section "Jingtong" (精通) of the "Almanac of the Last Autumn Month" (季秋紀): "慈石召鐵,或引之也]" - ^ In the section "A Last Word on Dragons" (亂龍篇 Luanlong) of the Lunheng: "Amber takes up straws, and a load-stone attracts needles" (頓牟掇芥,磁石引針).
- ^ Schmidl, Petra G. (1996–1997). "Two Early Arabic Sources On The Magnetic Compass". Journal of Arabic and Islamic Studies. 1: 81–132.
- ^ a b Gauss, Carl Friedrich (1867). Carl Friedrich Gauss Werke. Fünfter Band. Königliche Gesellschaft der Wissenschaften zu Göttingen. p. 617.
- ^ a b A. Einstein: "On the Electrodynamics of Moving Bodies", June 30, 1905.
- ISBN 9781420075021.
- ^
Catherine Westbrook; Carolyn Kaut; Carolyn Kaut-Roth (1998). MRI (Magnetic Resonance Imaging) in practice (2 ed.). Wiley-Blackwell. p. 217. ISBN 978-0-632-04205-0.
- ISSN 1059-1028. Retrieved 2024-02-08.
- ^ Purcell 2012, p. 320,584
- ^
Merzouki, Rochdi; Samantaray, Arun Kumar; Pathak, Pushparaj Mani (2012). Intelligent Mechatronic Systems: Modeling, Control and Diagnosis. Springer Science & Business Media. pp. 403–405. ISBN 978-1447146285.
- ^
Sturgeon, W. (1825). "Improved Electro Magnetic Apparatus". Trans. Royal Society of Arts, Manufactures, & Commerce. 43: 37–52. cited in
Miller, T.J.E (2001). Electronic Control of Switched Reluctance Machines. Newnes. p. 7. ISBN 978-0-7506-5073-1.
- ^ Griffiths 1998, chapter 12
- ISSN 1070-664X.
- ISBN 978-0-471-30932-1.
- S2CID 119061150..
- OCLC 38941224..
- ISBN 0-632-03583-8. pp. 14–15. Electronic version.
- PMID 1285705. Retrieved 29 March 2016.
- ISBN 978-1-987980-03-5.
- ^ Wilhelm Weber (2021). Andre Koch Torres Assis (ed.). Wilhelm Weber's Main Works in Electrodynamics Translated into English. Volume I: Gauss und Weber's Absolute System of Units. Apeiron Montreal.
- ^ Wilhelm Weber (2021). Andre Koch Torres Assis (ed.). Wilhelm Weber's Main Works in Electrodynamics Translated into English. Volume II: Weber's Fundamental Force and the Unification of the Laws of Coulomb, Ampere and Faraday. Apeiron Montreal.
- ^ Wilhelm Weber (2021). Andre Koch Torres Assis (ed.). Wilhelm Weber's Main Works in Electrodynamics Translated into English. Volume III: Measurement of Weber's Constant c, Diamagnetism, the Telegraph Equation and the Propagation of Electric Waves at Light Velocity. Apeiron Montreal.
- ^ Maxwell, James Clerk (1881). Treatise on Electricity and Magnetism. Volume 2. Vol. 2 (2 ed.). The Clarendon Press, Oxdord. p. 162.
- ^ "The Feynman Lectures on Physics Vol. II Ch. 34: The Magnetism of Matter". www.feynmanlectures.caltech.edu.
- ^ "The Feynman Lectures on Physics Vol. II Ch. 36: Ferromagnetism". www.feynmanlectures.caltech.edu.
Further reading
- David K. Cheng (1992). Field and Wave Electromagnetics. Addison-Wesley Publishing Company, Inc. ISBN 978-0-201-12819-2.
- Furlani, Edward P. (2001). Permanent Magnet and Electromechanical Devices: Materials, Analysis and Applications. OCLC 162129430.
- Griffiths, David J. (1998). Introduction to Electrodynamics (3rd ed.). Prentice Hall. OCLC 40251748.
- Kronmüller, Helmut. (2007). Handbook of Magnetism and Advanced Magnetic Materials, 5 Volume Set. John Wiley & Sons. OCLC 124165851.
- Purcell, Edward M. (2012). Electricity and magnetism (3rd ed.). Cambridge: Cambridge Univ. Press. ISBN 9781-10701-4022.
- Tipler, Paul (2004). Physics for Scientists and Engineers: Electricity, Magnetism, Light, and Elementary Modern Physics (5th ed.). W.H. Freeman. OCLC 51095685.
- Coey, J. M. D. (2019). Magnetism and Magnetic Materials. Cambridge University Press. ISBN 978-1108717519.


























