Treatment and management of COVID-19
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|
The treatment and management of COVID-19 combines both
Most cases of COVID-19 are mild. In these, supportive care includes medication such as
Although several medications have been approved in different countries as of April 2022, not all countries have these medications. Patients with mild to moderate symptoms who are in the risk groups can take
Several experimental treatments are being actively studied in clinical trials.[20] These include the antivirals molnupiravir (developed by Merck),[21] and nirmatrelvir/ritonavir (developed by Pfizer).[22][23] Others were thought to be promising early in the pandemic, such as hydroxychloroquine and lopinavir/ritonavir, but later research found them to be ineffective or even harmful,[24][needs update][25][26] like fluvoxamine, a cheap and widely available antidepressant;[27] As of December 2020, there was not enough high-quality evidence to recommend so-called early treatment.[25][26] In December 2020, two monoclonal antibody-based therapies were available in the United States, for early use in cases thought to be at high risk of progression to severe disease.[26] The antiviral remdesivir has been available in the U.S., Canada, Australia, and several other countries, with varying restrictions; however, it is not recommended for people needing mechanical ventilation, and has been discouraged altogether by the World Health Organization (WHO),[28] due to limited evidence of its efficacy.[24] In November 2021, the UK approved the use of molnupiravir as a COVID treatment for vulnerable patients recently diagnosed with the disease.[29]
The WHO, the
General support
Taking over-the-counter drugs such as paracetamol or ibuprofen, drinking fluids, taking honey to ease a cough, and resting may help alleviate symptoms.[7][35][36][37]
Medications

In the early months of the pandemic, many ICU doctors faced with the virus ventured to prescribe conjectured treatments because of the unprecedented circumstances.[38] However, the standard of care for most intractable illnesses is that, as it develops over years, doctors build a body of research that tests various theories, compares and contrasts dosages, and measures one drug's power against another.[38]
Antiviral development for SARS-CoV-2 has been disappointing.[39] In January 2020, research into potential treatments started,[40] and several antiviral drugs were in clinical trials.[41][42] In February 2020 with 'no known effective' treatments, the WHO recommended volunteers take part in trials of the effectiveness and safety of potential treatments.[43] Antiviral medications were tried in people with severe disease.[1] As of March 2020 several medications were already approved for other uses or were already in advanced testing.[44] As of April 2020 trials were investigating whether existing medications could be used effectively against the body's immune reaction to SARS-CoV-2 infection.[45][46] As of May 2020 several antiviral drugs were under investigation for COVID-19, though none had been shown to be clearly effective on mortality in published randomized controlled trials.[45]
As of February 2021, in the European Union, the use of dexamethasone and remdesivir were authorized.[47] Despite being controversial at the beginning of the pandemic in 2020,[48] corticosteroids like dexamethasone showed clinical benefit in treating COVID-19, once randomized controlled trials were performed in 2020.[49][50] As of February 2021, the
As of February 2021 there were Emergency Use Authorizations for baricitinib, bamlanivimab, bamlanivimab/etesevimab, and casirivimab/imdevimab.[53]
As of July 2021, outpatient drugs budesonide and tocilizumab showed promising results in some patients but remained under investigation.[54][55][56] As of July 2021, a large number of drugs had been considered for treating COVID-19 patients.[57] As of November 2022, there was moderate-certainty evidence suggesting that dexamethasone, and systemic corticosteroids in general, probably cause a slight reduction in all-cause mortality (up to 30 days) in hospitalized patients with COVID‐19, the evidence was very uncertain at 120 days.[58]
In March 2022, the
The WHO recommendations on which medications should or should not be used to treat Covid-19 are continuously updated. As of July 2022, WHO strongly recommended for non-severe cases
For patients in a life-threatening stage of the illness and in the presence of poor prognostic predictors, early antiviral treatment is essential.[61]
Ineffective
As of 2020, several treatments had been investigated and found to be ineffective or unsafe, and are thus were not recommended for use; these include baloxavir marboxil, lopinavir/ritonavir, ruxolitinib, chloroquine, hydroxychloroquine, interferon β-1a, and colchicine.[14] As of 2021, favipiravir and nafamostat had shown mixed results but were still in clinical trials in some countries.[62][63][64]
During the early part of 2020 convalescent plasma, plasma from persons who recovered from SARS-CoV-2 infection, was frequently used with anecdotal successes in reports and small case series.[65] However subsequent trials found no consistent evidence of benefit.[66][67] However, conflicting outcomes from trials could be understood by noting that they transfused insufficient therapeutic doses of CCP.[68]
As of February 2021, in the United States, only remdesivir had FDA approval for certain COVID-19 patients,[69] and while early research had suggested a benefit in preventing death and shortening illness duration, this was not borne out by subsequent trials.[24]
On 16 April 2021, the FDA revoked the emergency use authorization (EUA) for the investigational monoclonal antibody therapy bamlanivimab, when administered alone, to be used for the treatment of mild-to-moderate COVID-19 in adults and certain pediatric patients.[70]
As of July 2022, WHO strongly recommended against treating non-severe cases with convalescent plasma, hydroxychloroquine, lopinavir-ritonavir or colchicine and recommended conditionally against corticosteroids or ivermectin or fluvoxamine or nirmatrelvir and ritonavir WHO also strongly recommended against treating severe cases with hydroxychloroquine or lopinavir-ritonavir or Baricitinib and conditionally recommended against ruxolitinib or tofacitinib, ivermectin or convalescent plasma.[60]
As of September 2022, oral treatment of outpatients with metformin, ivermectin, and fluvoxamine were found to be ineffective in a large randomized, controlled trial.[71]
Adjuvant anticoagulation
In general there is no good evidence that anticoagulants have any benefit in the treatment of COVID-19, other than poor quality evidence suggesting a possible effect on all-cause mortality.[72]
Respiratory support
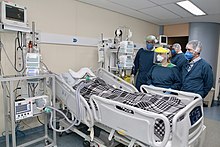
People seriously ill with COVID-19 may require respiratory support. Depending on the severity, oxygen therapy, mechanical ventilation, and intravenous fluids may be required.[73]
Mechanical ventilation
Most cases of COVID-19 are not severe enough to require mechanical ventilation or alternatives, but a percentage of cases are.[74][75] Some of the people acutely ill with COVID-19 experience deterioration of their lungs and acute respiratory distress syndrome (ARDS) and/or respiratory failure. Due to the high risk of death, urgent respiratory support including mechanical ventilation is often required in these people.[76] Mechanical ventilation becomes more complex as ARDS develops in COVID-19 and oxygenation becomes increasingly difficult.[77]
People who undergo mechanical ventilation are at risk of ventilator-associated lung injury or of worsening an existing lung injury, this damage is called ventilatory-induced lung injury (VILI).[76] The mechanism of this injury is thought to be due to trauma to the lungs caused by aerated regions of the lungs being over swollen (overdistension of the aerated alveoli) and atelectrauma (force on the alveolar that could lead to lung collapse).[76]
Ventilators capable of
Other approaches to mechanical ventilation including avoiding
The administration of inhaled nitric oxide to people being ventilated is not recommended, and evidence around this practice is weak.[82]
Extracorporeal membrane oxygenation
Extracorporeal membrane oxygenation (ECMO) is an artificial lung technology that has been used since the 1980s to treat respiratory failure and acute respiratory distress syndrome when conventional mechanical ventilation fails. In this complex procedure, blood is removed from the body via large cannulae, moved through a membrane oxygenator that performs the lung functions of oxygen delivery and carbon dioxide removal, and then returned to the body. The Extracorporeal Life Support Organization (ELSO) maintains a registry of outcomes for this technology, and as of September 2020 it has been used in less than 120,000 patients over 435 ECMO centers worldwide with 40% mortality for adult respiratory patients.[83]
Initial use of ECMO in COVID-19 patients from China early in the pandemic suggested poor outcomes, with less than 90% mortality.[84] In March 2020, the ELSO registry began collecting data on the worldwide use of ECMO for patients with COVID-19 and reporting this data on the ELSO website in real time. In September 2020, the outcomes of 1,035 COVID-19 patients supported with ECMO from 213 experienced centers in 36 different countries were published in The Lancet, and demonstrated 38% mortality, which is similar to many other respiratory diseases treated with ECMO. The mortality is also similar to the 35% mortality seen in the EOLIA trial, the largest randomized controlled trial for ECMO in ARDS.[85] This registry based, multi-center, multi-country data provide provisional support for the use of ECMO for COVID-19 associated acute hypoxemic respiratory failure. Given that this is a complex technology that can be resource intense, guidelines exist for the use of ECMO during the COVID-19 pandemic.[86][87][88]
Psychological support
Individuals may experience distress from quarantine, travel restrictions, side effects of treatment, or fear of the infection itself. To address these concerns, the National Health Commission of China published a national guideline for psychological crisis intervention on 27 January 2020.[89][90]
According to the Inter-Agency Standing Committee (IASC) Guidelines on Mental Health and Psychosocial Support, the pandemic produced long-term consequences. Deterioration of social networks and economies, survivor stigma, anger and aggression, and mistrust of official information are long-term consequences.[91]
In April 2020 The Lancet published a 14-page call for action focusing on the UK and stated conditions were such that a range of mental health issues was likely to become more common. BBC quoted Rory O'Connor in saying, "Increased social isolation, loneliness, health anxiety, stress, and an economic downturn are a perfect storm to harm people's mental health and wellbeing."[92][93]
Special populations
Concurrent treatment of other conditions
Early in the pandemic, theoretical concerns were raised about
People who use topical or systemic corticosteroids for respiratory conditions such as asthma or chronic obstructive pulmonary disease should continue taking them as prescribed even if they contract COVID-19.[49]
The principal for obstetric management of COVID-19 include rapid detection, isolation, and testing, profound preventive measures, regular monitoring of fetus as well as of uterine contractions, peculiar case-to-case delivery planning based on severity of symptoms, and appropriate post-natal measures for preventing infection.[99]
Patients with simultaneous Influenza infection
Patients with simultaneous SARS CoV2 and Influenza infection are more than twice as likely to die and more than four times as likely to need ventilation as patients with only COVID. It is recommended that patients admitted to hospital with COVID should be routinely tested to see if they also have Influenza. The public are advised to get vaccinated against both Influenza and COVID.[100]
Epidemiology
Severe cases are most common in older adults (those older than 60 years,[74] and especially those older than 80 years).[101] Many developed countries do not have enough hospital beds per capita, which limits a health system's capacity to handle a sudden spike in the number of COVID-19 cases severe enough to require hospitalisation.[102] This limited capacity is a significant driver behind calls to flatten the curve.[102] One study in China found 5% were admitted to intensive care units, 2.3% needed mechanical support of ventilation, and 1.4% died.[15] In China, approximately 30% of people in hospital with COVID-19 are eventually admitted to ICU.[103]
References
- ^ PMID 32106852.
- PMID 32044814.
- PMID 32171074.
- S2CID 236472654.
- ^ a b c d Motseki TP (7 June 2022). "COVID-19 Vaccination Guidelines". www.nih.gov. National Institutes of Health. Archived from the original on 19 January 2021. Retrieved 18 January 2021.
- PMID 32134116.
- ^ a b "Coronavirus". WebMD. Archived from the original on 1 February 2020. Retrieved 1 February 2020.
- PMID 32387333.
- ^ "Coronavirus recovery: breathing exercises". www.hopkinsmedicine.org. Johns Hopkins Medicine. Archived from the original on 11 October 2020. Retrieved 30 July 2020.
- PMID 32201353.
- ^ U.S. Centers for Disease Control and Prevention (5 April 2020). "What to Do if You Are Sick". U.S. Centers for Disease Control and Prevention (CDC). Archived from the original on 14 February 2020. Retrieved 24 April 2020.
- S2CID 227059995.
- ^ "Q&A: Dexamethasone and COVID-19". World Health Organization (WHO). Archived from the original on 11 October 2020. Retrieved 11 July 2020.
- ^ National COVID-19 Clinical Evidence Taskforce. Archivedfrom the original on 11 October 2020. Retrieved 11 July 2020.
- ^ PMID 32109013.
- PMID 32178774.
- PMID 33391477.
- ^ "COVID Treatment Guidelines: Clinical Management Summary". NIH Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. 8 April 2022. Archived from the original on 5 November 2021. Retrieved 19 April 2022.
- ^ Wise J (17 April 2022). "What Happened to Paxlovid, the COVID Wonder Drug?". Intelligencer. Archived from the original on 19 April 2022. Retrieved 19 April 2022.
- PMID 33849936.
- ^ Aripaka P (5 November 2021). "Britain approves Merck's COVID-19 pill in world first". Reuters. Archived from the original on 8 November 2021. Retrieved 8 November 2021.
- ^ Beasley D (5 November 2021). "Pfizer says its antiviral pill slashes risk of severe COVID-19 by 89%". Reuters. Archived from the original on 7 November 2021. Retrieved 8 November 2021.
- PMID 38032024.
- ^ PMID 32732190.
- ^ PMID 33175121.
- ^ a b c "COVID-19 Treatment Guidelines". www.nih.gov. National Institutes of Health. Archived from the original on 19 January 2021. Retrieved 18 January 2021./
- ^ Saima MS (2 November 2021). "Common Antidepressant Slashes Risk of COVID Death". Nature. Archived from the original on 8 November 2021. Retrieved 8 November 2021.
- PMID 33214186.
- ^ Reed J (4 November 2021). "Molnupiravir: First pill to treat Covid gets approval in UK". www.bbc.co.uk. Archived from the original on 4 November 2021. Retrieved 23 November 2021.
- ^ "Clinical management of COVID-19". World Health Organization (WHO). 27 May 2020. Archived from the original on 15 January 2021. Retrieved 18 January 2021.
- ^ "Coronavirus (COVID-19) | NICE". National Institute for Health and Care Excellence. Archived from the original on 20 January 2021. Retrieved 18 January 2021.
- PMID 32072569.
- ^ Farkas J (March 2020). COVID-19—The Internet Book of Critical Care (digital) (Reference manual). USA: EMCrit. Archived from the original on 11 March 2020. Retrieved 13 March 2020.
- Penn Libraries. 11 March 2020. Archivedfrom the original on 14 March 2020. Retrieved 13 March 2020.
- ^ "Home care for patients with suspected or confirmed COVID-19 and management of their contacts" (PDF). World Health Organization (WHO). 13 August 2020. Archived from the original on 21 January 2021. Retrieved 18 January 2021.
- ^ "Prevention & Treatment". U.S. Centers for Disease Control and Prevention (CDC). 15 February 2020. Archived from the original on 15 December 2019. Retrieved 21 January 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "How to look after yourself at home if you have coronavirus (COVID-19) or symptoms of COVID-19". NHS. 6 May 2022. Archived from the original on 23 May 2022. Retrieved 22 May 2022.
- ^ a b Dominus S (5 August 2020). "The Covid Drug Wars That Pitted Doctor vs. Doctor". The New York Times. Archived from the original on 1 July 2021. Retrieved 1 July 2021.
- PMID 34319150.
- ^ "Chinese doctors using plasma therapy on coronavirus, WHO says 'very valid' approach". Reuters. 17 February 2020. Archived from the original on 4 March 2020. Retrieved 19 March 2020.
- ^ Steenhuysen J, Kelland K (24 January 2020). "With Wuhan virus genetic code in hand, scientists begin work on a vaccine". Reuters. Archived from the original on 25 January 2020. Retrieved 25 January 2020.
- ^ Duddu P (19 February 2020). "Coronavirus outbreak: Vaccines/drugs in the pipeline for Covid-19". clinicaltrialsarena.com. Archived from the original on 19 February 2020.
- ^ Nebehay S, Kelland K, Liu R (5 February 2020). "WHO: 'no known effective' treatments for new coronavirus". Thomson Reuters. Archived from the original on 5 February 2020. Retrieved 5 February 2020.
- PMID 32127666.
- ^ S2CID 215752785.
- PMID 32284951.
- ^ "Treatments and vaccines for COVID-19: authorised medicines". European Medicines Agency. 11 January 2021. Archived from the original on 19 February 2021. Retrieved 20 February 2021.
- PMID 33532161.
- ^ National COVID-19 Clinical Evidence Taskforce. Archivedfrom the original on 25 September 2020. Retrieved 11 July 2020.
- PMID 32696108.
- ^ a b "FDA Authorizes Monoclonal Antibodies for Treatment of COVID-19". U.S. Food and Drug Administration (FDA) (Press release). 10 February 2021. Archived from the original on 10 February 2021. Retrieved 9 February 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "Coronavirus (COVID-19) Update: FDA Authorizes Monoclonal Antibodies for Treatment of COVID-19". U.S. Food and Drug Administration (FDA). 23 November 2020. Archived from the original on 25 January 2021. Retrieved 17 April 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ "COVID-19 Frequently Asked Questions: Drugs (Medicines)". U.S. Food and Drug Administration. 19 February 2021. Archived from the original on 19 February 2021. Retrieved 20 February 2021.
- PMID 33971376.
- S2CID 247725977.
- PMID 34240518.
- PMID 34234253.
- PMID 36385229.
- ^ Coronavirus treatments: What progress is being made? Archived 19 March 2022 at the Wayback Machine BBC
- ^ S2CID 251658284.
- PMID 35107300.
- PMID 34063964.
- PMID 34177298.
- PMID 34071185.
- PMID 33635310.
- PMID 35262370.
- PMID 35967398.
- PMID 35262370.
- ^ Zimmer C, Wu KJ, Corum J (16 July 2020). "Coronavirus Drug and Treatment Tracker". The New York Times. Archived from the original on 20 January 2021. Retrieved 3 November 2021.
- ^ "Coronavirus (COVID-19) Update: FDA Revokes Emergency Use Authorization for Monoclonal Antibody Bamlanivimab". U.S. Food and Drug Administration (FDA) (Press release). 16 April 2021. Archived from the original on 16 April 2021. Retrieved 16 April 2021.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- S2CID 251658284.
- PMID 35244208.
- ^ "Overview of novel coronavirus (2019-nCoV)—Summary of relevant conditions". The BMJ. Archived from the original on 31 January 2020. Retrieved 1 February 2020.
- ^ from the original on 18 March 2020. Retrieved 18 March 2020.
- ^ "Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected" (PDF). World Health Organization (WHO). 28 January 2020. Archived (PDF) from the original on 26 February 2020. Retrieved 18 March 2020.
- ^ PMID 33784416.
- PMID 32203709.
- from the original on 11 October 2020. Retrieved 1 June 2020.
- PMID 20197533.
- ^ PMID 35767435.
- PMID 32278367.
- PMID 32222812.
- ^ "Extracorporeal Life Support Organization - ECMO and ECLS > Registry > Statistics > International Summary". www.elso.org. Archived from the original on 23 September 2020. Retrieved 28 September 2020.
- PMID 32279018.
- S2CID 44106489.
- PMID 32243267.
- PMID 32358233.
- PMID 32203711.
- PMID 32032543.
- PMID 32035030.
- ^ "Inter-Agency Standing Committee Guidelines on Mental Health and Psychosocial support" (PDF). MH Innovation. Archived (PDF) from the original on 31 March 2020. Retrieved 28 March 2020.
- ^ Roxby P (16 April 2020). "Coronavirus: 'Profound' mental health impact prompts calls for urgent research". BBC. Archived from the original on 11 October 2020.
- PMID 32304649.
"A fragmented research response, characterised by small-scale and localised initiatives, will not yield the clear insights necessary to guide policymakers or the public
- ^ "Patients taking ACE-i and ARBs who contract COVID-19 should continue treatment, unless otherwise advised by their physician". Archived from the original on 21 March 2020. Retrieved 21 March 2020.
- ^ "Patients taking ACE-i and ARBs who contract COVID-19 should continue treatment, unless otherwise advised by their physician". American Heart Association (Press release). 17 March 2020. Archived from the original on 24 March 2020. Retrieved 25 March 2020.
- ^ de Simone G. "Position Statement of the ESC Council on Hypertension on ACE-Inhibitors and Angiotensin Receptor Blockers". Council on Hypertension of the European Society of Cardiology. Archived from the original on 24 March 2020. Retrieved 24 March 2020.
- ^ "New Evidence Concerning Safety of ACE Inhibitors, ARBs in COVID-19". Pharmacy Times. 28 April 2020. Archived from the original on 11 October 2020. Retrieved 2 May 2020.
- ^ "FDA advises patients on use of non-steroidal anti-inflammatory drugs (NSAIDs) for COVID-19". U.S. Food and Drug Administration (FDA). 19 March 2020. Archived from the original on 27 March 2020. Retrieved 27 March 2020.
- PMID 33209759.
- ^ McKie R (27 March 2022). "Patients with Covid and flu double the risk of dying, say scientists". The Guardian. Archived from the original on 27 March 2022.
- hdl:20.1000/100.
- ^ a b Scott D (16 March 2020). "Coronavirus is exposing all of the weaknesses in the US health system High health care costs and low medical capacity made the US uniquely vulnerable to the coronavirus". Vox. Archived from the original on 18 March 2020. Retrieved 18 March 2020.
- ^ "Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID-19)". U.S. Centers for Disease Control and Prevention (CDC). 6 April 2020. Archived from the original on 2 March 2020. Retrieved 19 April 2020.
External links
Treatment guidelines
- "JHMI Clinical Recommendations for Available Pharmacologic Therapies for COVID-19" (PDF). Johns Hopkins Medicine.
- "Bouncing Back From COVID-19: Your Guide to Restoring Movement" (PDF). Johns Hopkins Medicine. Archived from the original (PDF) on 15 February 2021. Retrieved 5 December 2020.
- "Guidelines on the Treatment and Management of Patients with COVID-19". Infectious Diseases Society of America (IDSA).
- "Coronavirus Disease 2019 (COVID-19) Treatment Guidelines". National Institutes of Health.
- hdl:10665/334125. WHO/2019-nCoV/Corticosteroids/2020.1.
- World Health Organization (2022). Therapeutics and COVID-19: living guideline (Report). World Health Organization (WHO). WHO-2019-nCoV-therapeutics-2022.1.
