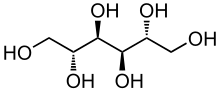
Mannitol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Osmitrol, Bronchitol, others |
| Other names | d-Mannitol, mannite, manna sugar |
| AHFS/Drugs.com | Monograph |
| License data | |
| Pregnancy category |
|
Intravenous, By mouth | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | ~7% |
| Metabolism | Liver, negligible |
| Elimination half-life | 100 minutes |
| Excretion | Kidney: 90% |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Mannitol is a type of
Common side effects from medical use include
The discovery of mannitol is attributed to
Uses

Medical uses
In the United States, mannitol is indicated for the reduction of intracranial pressure and treatment of cerebral edema and elevated intraocular pressure.[3]
In the European Union, mannitol is indicated for the treatment of cystic fibrosis (CF) in adults aged 18 years and above as an add-on therapy to best standard of care.[4]
Mannitol is used intravenously to reduce acutely raised intracranial pressure until more definitive treatment can be applied,
It may also be used for certain cases of
Intraoperative mannitol prior to vessel clamp release during renal transplant has been shown to reduce post-transplant kidney injury, but has not been shown to reduce graft rejection.[medical citation needed]
Mannitol acts as an osmotic laxative[3][19] in oral doses larger than 20 g,[20] and is sometimes sold as a laxative for children.[citation needed]
The use of mannitol, when inhaled, as a bronchial irritant as an alternative method of diagnosis of
Mannitol is commonly used in the circuit prime of a
Mannitol can also be used to temporarily encapsulate a sharp object (such as a helix on a lead for an
Mannitol is also the first drug of choice to treat acute
Food
Mannitol increases blood glucose to a lesser extent than
Analytical chemistry
Mannitol can be used to form a complex with boric acid. This increases the acid strength of the boric acid, permitting better precision in volumetric analysis of this acid.[27]
Other
Mannitol is the primary ingredient of mannitol salt agar, a bacterial growth medium, and is used in others.
Mannitol is used as a
Mannitol is a sugar alcohol "Mannitol has 50-70 percent of the relative sweetness of sugar, which means more must be used to equal the sweetness of sugar. Mannitol lingers in the intestines for a long time and therefore often causes bloating and diarrhea."<https://www.ynhh.org/services/nutrition/sugar-alcohol#:~:text=Mannitol%20has%2050%2D70%20percent,naturally%20in%20fruits%20and%20vegetables.>
Contraindications
Mannitol is contraindicated in people with anuria, severe hypovolemia, pre-existing severe pulmonary vascular congestion or pulmonary edema, irritable bowel syndrome (IBS), and active intracranial bleeding except during craniotomy.[3]
Adverse effects include hyponatremia and volume depletion leading to metabolic acidosis.[10]
Chemistry
Mannitol is an
Production
Mannitol is classified as a
Industrial synthesis
Mannitol is commonly produced via the
Biosyntheses
Mannitol is one of the most abundant energy and carbon storage molecules in nature, produced by a plethora of organisms, including bacteria, yeasts, fungi, algae, lichens, and many plants.
Natural extraction
Since mannitol is found in a wide variety of natural products, including almost all plants, it can be directly extracted from natural products, rather than chemical or biological syntheses. In fact, in China, isolation from
Traditionally, mannitol is extracted by the
Supercritical
History
Julije Domac elucidated the structure of hexene and mannitol obtained from Caspian manna. He determined the place of the double bond in hexene obtained from mannitol and proved that it is a derivative of a normal hexene. This also solved the structure of mannitol, which was unknown until then.[33][34][35][36]
Controversy
The three studies
Compendial status
See also
References
- FDA. Retrieved 22 October 2023.
- ^ "Regulatory Decision Summary - Aridol". Health Canada. 23 October 2014. Retrieved 7 June 2022.
- ^ a b c d "Osmitrol- mannitol injection, solution". DailyMed. 15 November 2018. Retrieved 28 October 2020.
- ^ a b "Bronchitol EPAR". European Medicines Agency (EMA). 17 September 2018. Retrieved 28 October 2020. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ ISBN 9781439876732. Archivedfrom the original on 10 September 2017.
- ^ ISBN 9789241547659.
- ^ PMID 23918314.
- ^ a b c d e f g "Mannitol". The American Society of Health-System Pharmacists. Archived from the original on 26 May 2015. Retrieved 8 January 2015.
- ^ "BRONCHITOL® (mannitol) inhalation powder Patient Site". bronchitol.com.
- ^ ISBN 9780931292170. Archivedfrom the original on 10 September 2017.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ISBN 9780323461122.
- ISBN 978-1437735789.
- ^ "THE 2017 PROHIBITED LIST INTERNATIONAL STANDARD" (PDF). January 2017. p. 5. Retrieved 7 July 2018.
- ^ "Mannitol (Intravenous Route)". Mayo Clinic.
- ^ Gu, J., Huang, H., Huang, Y. et al. Hypertonic saline or mannitol for treating elevated intracranial pressure in traumatic brain injury: a meta-analysis of randomized controlled trials. Neurosurg Rev 42, 499–509 (2019). https://doi.org/10.1007/s10143-018-0991-8
- S2CID 20258642.
- S2CID 199507024.
- ^ "Select Committee on GRAS Substances (SCOGS) Opinion: Mannitol". FDA.gov. April 2013. Archived from the original on 22 October 2014.
- from the original on 10 September 2017.
- PMID 21875541.
- ^ Veterinary Class Notes, Ophthalmology, The Ohio State University, provided by David Wilkie, DVM, DACVO
- ISBN 978-1461285229.
- ^ ISBN 0470659688.
- ^ a b Lawson P (2007). Mannitol. Blackwell Publishing Ltd. pp. 219–225.
- ^
Weiner ML, Kotkoskie LA (1999). Excipient Toxicity and Safety. Taylor & Francis. pp. 370. ISBN 9780824782108.
- ^ Belcher R, Nutten AJ (1960). Quantitative Inorganic Analysis (2nd ed.). London, UK: Butterworths. p. 194.
- ^ "Cut the Shit". December 2005. Archived from the original on 27 September 2016. Retrieved 4 September 2017.
- ^ Kearsley MW, Deis RC (2006). "Sorbitol and Mannitol". Sweeteners and Sugar Alternatives in Food Technology. Ames: Oxford. pp. 249–261.
{{cite book}}: CS1 maint: location missing publisher (link) - ^ S2CID 42103028.
- ^ .
- S2CID 260249165.
- PMID 22026131.
- ^ Domac J (1881). "Über das Hexylen aus Mannit". Sitzungsberichte der Kaiserlichen Akademie der Wissenschaften, Mathematisch-Naturwissenschaftliche Classe (in German). 23: 1038–1051.
- S2CID 94940823.
- .
- S2CID 43880412.
- S2CID 20678448.
- PMID 15035271.
- PMID 17322250.
- ^ British Pharmacopoeia Commission Secretariat (2009). "Index, BP 2009" (PDF). Archived from the original (PDF) on 11 April 2009. Retrieved 31 January 2010.
- ^ "Japanese Pharmacopoeia, Fifteenth Edition" (PDF). 2006. Archived from the original (PDF) on 22 July 2011. Retrieved 31 January 2010.
- ^ USP 32 (2008). "Mannitol Injection" (PDF). Archived from the original (PDF) on 6 July 2010. Retrieved 31 January 2010.
{{cite web}}: CS1 maint: numeric names: authors list (link)
External links
 Media related to Mannitol at Wikimedia Commons
Media related to Mannitol at Wikimedia Commons
