Marine fungi
| Part of a series of overviews on |
| Marine life |
|---|
 |
Marine fungi are
Different marine habitats support very different fungal communities. Fungi can be found in niches ranging from ocean depths and coastal waters to
Overview
Terrestrial fungi play critical roles in nutrient cycling and food webs and can shape macroorganism communities as parasites and mutualists. Although estimates for the number of fungal species on the planet range from 1.5 to over 5 million, likely fewer than 10% of fungi have been identified so far. To date, a relatively small percentage of described species are associated with marine environments, with ~1,100 species retrieved exclusively from the marine environment. Nevertheless, fungi have been found in nearly every marine habitat explored, from the surface of the ocean to kilometers below ocean sediments. Fungi are hypothesized to contribute to
Fungi represent a large and diverse group of microorganisms in microbiological communities in the marine environment and have an important role in nutrient cycling.[7] They are divided into two major groups; obligate marine fungi and facultative marine fungi.[8] Obligate marine fungi are adapted to reproduce in the aquatic environment, while facultative marine fungi can grow in aquatic as well as terrestrial environments.[8] Marine fungi are called marine-derived fungi when their facultative or obligate state is not certain.[9]
Marine fungal species occur as
Factors that influence whether or not marine fungi are present in any particular location include the water temperature, its salinity, the water movement, the presence of suitable substrates for colonization, the presence of propagules in the water, interspecific competition, pollution and the oxygen content of the water.[5]
Some marine fungi which have ventured into the sea from terrestrial habitats include species that burrow into sand grains, living in the pores. Others live inside stony corals, and may become pathogenic if the coral is stressed by rising sea temperatures.[3][self-published source?][12]
In 2011 the
The secondary metabolites produced by marine fungi have high potential for use in biotechnological, medical and industrial applications.[14]
Evolution
In contrast to
The earliest fossils possessing features typical of fungi date to the
For much of the
The evolutionary adaptation from an aquatic to a terrestrial lifestyle necessitated a diversification of ecological strategies for obtaining nutrients, including
The growth of fungi as
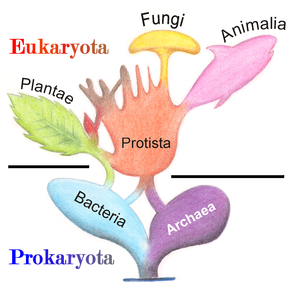
Fungi were considered to be part of the plant kingdom until the mid-20th century. By the middle of the 20th century Fungi were considered a distinct kingdom, and the newly recognized kingdom Fungi becoming the third major kingdom of multicellular eukaryotes with kingdom Plantae and kingdom Animalia, the distinguishing feature between these kingdoms being the way they obtain nutrition.[28]
Marine plants
Mangroves
The greatest number of known species of marine fungi are from
Other plants
The
Eelgrass, Zostera marina, is sometimes affected by seagrass wasting disease. The primary cause of this seems to be pathogenic strains of the protist Labyrinthula zosterae, but it is thought that fungal pathogens also contribute and may predispose the eelgrass to disease.[31][32]
Wood

Many marine fungi are very specific as to which species of floating and submerged wood they colonise. A range of species of fungi colonise beech, while oak supports a different community. When a fungal propagule lands on a suitable piece of wood, it will grow if no other fungi are present. If the wood is already colonised by another fungal species, growth will depend on whether that fungus produces antifungal chemicals and whether the new arrival can resist them. The chemical properties of colonizing fungi also affect the animal communities that graze on them: in one study, when hyphae from five different species of marine fungi were fed to nematodes, one species supported less than half the number of nematodes per mg of hyphae than did the others.[33]
Detection of fungi in wood may involve incubation at a suitable temperature in a suitable water medium for a period of six months to upward of eighteen months.[33]
Lichens

Lichens are mutualistic associations between fungi, usually an ascomycete with a basidiomycete,[34] and an alga or a cyanobacterium. Several lichens, including Arthopyrenia halodytes, Pharcidia laminariicola, Pharcidia rhachiana and Turgidosculum ulvae, are found in marine environments.[2] Many more occur in the splash zone, where they occupy different vertical zones depending on how tolerant they are to submersion.[35] Lichen-like fossils have been found in the Doushantuo Formation in China dating back about 600 million years ago.[36]
Fungi from Verrucariales also form marine lichens with the brown algae Petroderma maculiforme,[37] and have a symbiotic relationship with seaweed like (rockweed) and Blidingia minima, where the algae are the dominant components. The fungi is thought to help the rockweeds to resist desiccation when exposed to air.[38][39] In addition, lichens can also use yellow-green algae (Heterococcus) as their symbiotic partner.[40]
Lichen-like fossils consisting of coccoid cells (
Not to be confused with lichens are Mycophycobiosis, similar to lichens in being a symbiosis of an algae and a fungus, in mycophycobiosis the algae forms the external, multicellular structure housing the fungus. The reproduction of both partners is always disjoint. [49]
Algae and phytoplankton
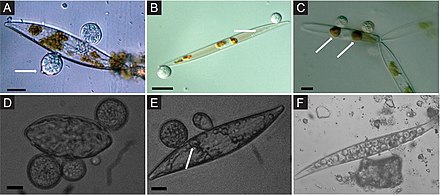
Marine fungi associated with algae are largely unexplored, despite their ecological role and potential industrial applications. For example, it has been shown that fungi associated with algae produce many bioactive
Almost one-third of all known marine fungal species are associated with algae.[59] The most commonly described fungi associated with algae belong to the Ascomycota and are represented by a wide diversity of genera such as Acremonium, Alternaria, Aspergillus, Cladosporium, Phoma, Penicillium, Trichoderma, Emericellopsis, Retrosium, Spathulospora, Pontogenia and Sigmoidea.[55][60][57][58][61][62][63][9]
Invertebrates
The
Vertebrates
Marine sediment
Ascomycota, Basidiomycota, and Chytridiomycota have been observed in marine sediments ranging in depth from 0 to 1740 meters beneath the ocean floor. One study analyzed subsurface samples of marine sediment between these depths and isolated all observable fungi. Isolates showed that most subsurface fungal diversity was found between depths of 0 to 25 meters below the sea floor with Fusarium oxysporum and Rhodotorula mucilaginosa being the most prominent. Overall, the ascomycota are the dominant subsurface phylum.[68] Almost all fungal species recovered have also been observed in terrestrial sediments with spore-sourcing indicating terrestrial origin.[68][69]
Contrary to previous beliefs, deep subsurface marine fungi actively grow and germinate, with some studies showing increased growth rates under high hydrostatic pressures. Though the methods by which marine fungi are able to survive the extreme conditions of the seafloor and below is largely unknown, Saccharomyces cerevisiae shines some light onto adaptations that make it possible. This fungus strengthens its outer membrane in order to endure higher hydrostatic pressures.[68][clarification needed]
Several sediment-dwelling marine fungi are involved in biogeochemical processes. Fusarium oxysporum and Fusarium solani are denitrifiers both in marine and terrestrial environments.[68][70] Some are co-denitrifying, fixing nitrogen into nitrous oxide and dinitrogen.[69] Still others process organic matter including carbohydrate, proteins, and lipids. Ocean crust fungi, like those found around hydrothermal vents, decompose organic matter, and play various roles in manganese and arsenic cycling.[6]
Sediment-bound marine fungi played a major role in breaking down oil spilled from the Deepwater Horizons disaster in 2010. Aspergillus, Penicillium, and Fusarium species, among others, can degrade high-molecular-weight hydrocarbons as well as assist hydrocarbon-degrading bacteria.[6]
Arctic marine fungi
Marine fungi have been observed as far north as the Arctic Ocean. Chytridiomycota, the dominant parasitic fungal organism in Arctic waters, take advantage of phytoplankton blooms in brine channels caused by warming temperatures and increased light penetration through the ice. These fungi parasitize diatoms, thereby controlling algal blooms and recycling carbon back into the microbial food web. Arctic blooms also provide conducive environments for other parasitic fungi. Light levels and seasonal factors, such as temperature and salinity, also control chytrid activity independently of phytoplankton populations. During periods of low temperatures and phytoplankton levels, Aureobasidium and Cladosporium populations overtake those of chytrids within the brine channels.[72]
Food webs and the mycoloop

Human uses
Biomass processors
Medical
Marine fungi produce antiviral and antibacterial compounds as metabolites with upwards of 1,000 having realized and potential uses as anticancer, anti-diabetic, and anti-inflammatory drugs.[77][78]
The antiviral properties of marine fungi were realized in 1988 after their compounds were used to successfully treat the H1N1 flu virus. In addition to H1N1, antiviral compounds isolated from marine fungi have been shown to have virucidal effects on
Mangrove-associated fungi have prominent antibacterial effects on several common pathogenic human bacteria including, Staphylococcus aureus and Pseudomonas aeruginosa. High competition between organisms within mangrove niches lead to increases in antibacterial substances produced by these fungi as defensive agents.[79] Penicillium and Aspergillus species are the largest producers of antibacterial compounds among the marine fungi.[80]
Various deep-sea marine fungi species have recently been shown to produce anti-cancer metabolites. One study uncovered 199 novel cytotoxic compounds with anticancer potential. In addition to cytotoxic metabolites, these compounds have structures capable of disrupting cancer-activated telomerases via DNA binding. Others inhibit the topoisomerase enzyme from continuing to aid in the repair and replication of cancer cells.[78]
See also
References
- S2CID 45289302.
- ^ a b c d Species of Higher Marine Fungi Archived 2013-04-22 at the Wayback Machine University of Mississippi. Retrieved 2012-02-05.
- ^ a b c "Cool New Paper: Marine Fungi". Teaching Biology. Archived from the original on March 16, 2012.
- PMID 30889385.
- ^ a b c E. B. Gareth Jones (2000). "Marine fungi: some factors influencing biodiversity" (PDF). Fungal Diversity. 4: 53–73.
- ^ doi:10.1128/mBio.01189-18..
 Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ Tisthammer, K.H.; Cobian, G.M.; Amend, A.S. Global biogeography of marine fungi is shaped by the environment. Fungal Ecol. 2016, 19, 39–46.
- ^ a b Raghukumar, S. The marine environment and the role of fungi. In Fungi in Coastal and Oceanic Marine Ecosystems: Marine Fungi; Springer International Publishing: Cham, Switzerland, 2017; pp. 17–38.
- ^ doi:10.3390/microorganisms8010052..
 Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ Jones, E.B.G.; Suetrong, S.; Sakayaroj, J.; Bahkali, A.H.; Abdel-Wahab, M.A.; Boekhout, T.; Pang, K.-L. Classification of marine Ascomycota, Basidiomycota, Blastocladiomycota and Chytridiomycota. Fungal Divers. 2015, 73, 1–72.
- ^ Debbab, A.; Aly, A.H.; Proksch, P. Mangrove derived fungal endophytes–a chemical and biological perception. Fungal Divers. 2013, 61, 1–27.
- .
- PMID 22457985.
- ^ Marine Fungi Retrieved 2012-02-06.
- ISBN 978-0-19-517234-8.
- S2CID 25586788.
- S2CID 6689439.
- S2CID 4302864.
- PMID 17010206.
- ^ "CK12-Foundation". flexbooks.ck12.org. Retrieved 2020-05-19.
- PMID 16151185.
- ^ Taylor and Taylor, pp. 84–94 and 106–107.
- PMID 20525580.
- ISBN 978-0-521-30899-1.
- PMID 12208998.
- ^ PMID 1837147.
- ISBN 978-0-7923-4981-5.
- ^ "Fungus - Evolution and phylogeny of fungi". Encyclopedia Britannica. Retrieved 2020-05-19.
- .
- PMID 14657360.
- ^ Disease Analysis in San Juan Archipelago Archived 2013-09-12 at the Wayback Machine Friday Harbor Laboratories Seagrass Lab. Retrieved 2012-02-06.
- ISBN 9780080525617.
- ^ ISBN 9780521308991.
- PMID 27445309.
- ^ Freshwater and marine lichen-forming fungi Retrieved 2012-02-06.
- S2CID 27083645.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 21653406.
- ^ "Mutualisms between fungi and algae – New Brunswick Museum". Archived from the original on 18 September 2018. Retrieved 4 October 2018.
- from the original on 7 October 2018. Retrieved 7 October 2018.
- PMID 23402662.
- S2CID 27083645.
- (PDF) from the original on 22 December 2016. Retrieved 22 December 2016.
- S2CID 17181699.
- S2CID 129180481.
- from the original on 1 January 2013. Retrieved 2 January 2013.
- .
- .
- S2CID 208034624.
- .
- PMID 26754171.
- ^ Overy, D.P.; Bayman, P.; Kerr, R.G.; Bills, G.F. An assessment of natural product discovery from marine (sensu stricto) and marine-derived fungi. Mycology 2014, 5, 145–167.
- ^ Flewelling, A.J.; Currie, J.; Gray, C.A.; Johnson, J.A. Endophytes from marine macroalgae: Promising sources of novel natural products. Curr. Sci. 2015, 109, 88–111.
- ^ Miao, F.; Zuo, J.; Liu, X.; Ji, N. Algicidal activities of secondary metabolites of marine macroalgal-derived endophytic fungi. J. Oceanol. Limnol. 2019, 37, 112–121.
- ^ Gnavi, G.; Garzoli, L.; Poli, A.; Prigione, V.; Burgaud, G.; Varese, G.C. The culturable mycobiota of Flabellia petiolata: First survey of marine fungi associated to a Mediterranean green alga. PLoS ONE 2017, 12, e0175941.
- ^ a b Kohlmeyer, J.; Volkmann-Kohlmeyer, B. Illustrated key to the filamentous higher marine fungi. Bot. Mar. 1991, 34, 1–61.
- ^ Stanley, S.J. Observations on the seasonal occurrence of marine endophytic and parasitic fungi. Can. J. Bot. 1992, 70, 2089–2096.
- ^ a b Zuccaro, A.; Schoch, C.L.; Spatafora, J.W.; Kohlmeyer, J.; Draeger, S.; Mitchell, J.I. Detection and identification of fungi intimately associated with the brown seaweed Fucus serratus. Appl. Environ. Microbiol. 2008, 74, 931–941.
- ^ a b Lee, S.; Park, M.S.; Lee, H.; Kim, J.J.; Eimes, J.A.; Lim, Y.W. Fungal diversity and enzyme activity associated with the macroalgae, Agarum clathratum. Mycobiology 2019, 47, 50–58.
- ^ Balabanova, L.; Slepchenko, L.; Son, O.; Tekutyeva, L. Biotechnology potential of marine fungi degrading plant and algae polymeric substrates. Front. Microbiol. 2018, 9, 15–27.
- ^ Stanley, S.J. Observations on the seasonal occurrence of marine endophytic and parasitic fungi. Can. J. Bot. 1992, 70, 2089–2096. [Google Scholar] [CrossRef]
- ^ Zuccaro, A.; Summerbell, R.C.; Gams, W.; Schroers, H.-J.; Mitchell, J.I. A new Acremonium species associated with Fucus spp., and its affinity with a phylogenetically distinct marine Emericellopsis clade. Stud. Mycol. 2004, 50, 283–297.
- ^ Kohlmeyer, J.; Kohlmeyer, E. Marine Mycology: The Higher Fungi; Elsevier: Amsterdam, the Netherlands, 2013.
- ^ Wainwright, B.J.; Bauman, A.G.; Zahn, G.L.; Todd, P.A.; Huang, D. Characterization of fungal biodiversity and communities associated with the reef macroalga Sargassum ilicifolium reveals fungal community differentiation according to geographic locality and algal structure. Mar. Biodivers. 2019, 49, 2601–2608.
- PMID 11052082.
- PMID 29304709.
- ^ Gaydos, Joseph K.; Balcomb, Kenneth C.; Osborne, Richard W.; Dierauf, Leslie. A Review of Potential Infectious Disease Threats to Southern Resident Killer Whales (Orcinus orca) (PDF) (Report). Archived from the original (PDF) on 2016-03-04. Retrieved 2012-09-28.
- ^ Fungal infections of farmed salmon and trout Archived 2012-10-24 at the Wayback Machine Retrieved 2012-02-06.
- ^ PMID 25769836.
- ^ S2CID 9724720.
- PMID 2040619.
- S2CID 216033140..
 Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - PMID 26754171.
- doi:10.3389/fmicb.2014.00166..
 Modified text was copied from this source, which is available under a Creative Commons Attribution 3.0 International License
Modified text was copied from this source, which is available under a Creative Commons Attribution 3.0 International License - .
- doi:10.1111/1462-2920.13827..
 Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - doi:10.1007/s00442-020-04721-w..
 Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License
Modified text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License - ^ PMID 26204947.
- ^ PMID 29354097.
- PMID 27933244.
- PMID 26042616.
Further reading
- Gareth Jones, E. B. and Pang, Ka-Lai (2012) Marine Fungi: and Fungal-like Organisms Marine and Freshwater Botany Walter de Gruyter. ISBN 9783110264067.
- Raghukumar, Chandralata (2012) Biology of Marine Fungi Springer. ISBN 9783642233425.
- Raghukumar, Seshagiri (2017) Fungi in Coastal and Oceanic Marine Ecosystems Springer. ISBN 9783319543048.




