Materials science
This article includes a list of general references, but it lacks sufficient corresponding inline citations. (August 2023) |

Materials science is an interdisciplinary field of researching and discovering materials. Materials engineering is an engineering field of finding uses for materials in other fields and industries.
The intellectual origins of materials science stem from the Age of Enlightenment, when researchers began to use analytical thinking from chemistry, physics, and engineering to understand ancient, phenomenological observations in metallurgy and mineralogy.[1][2] Materials science still incorporates elements of physics, chemistry, and engineering. As such, the field was long considered by academic institutions as a sub-field of these related fields. Beginning in the 1940s, materials science began to be more widely recognized as a specific and distinct field of science and engineering, and major technical universities around the world created dedicated schools for its study.
Materials scientists emphasize understanding how the history of a material (processing) influences its structure, and thus the
Materials science is also an important part of forensic engineering and failure analysis – investigating materials, products, structures or components, which fail or do not function as intended, causing personal injury or damage to property. Such investigations are key to understanding, for example, the causes of various aviation accidents and incidents.
History

The material of choice of a given era is often a defining point. Phases such as
.Before the 1960s (and in some cases decades after), many eventual materials science departments were metallurgy or ceramics engineering departments, reflecting the 19th and early 20th-century emphasis on metals and ceramics. The growth of materials science in the United States was catalyzed in part by the
Fundamentals
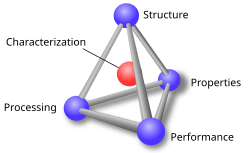
A material is defined as a substance (most often a solid, but other condensed phases can be included) that is intended to be used for certain applications.
The basis of materials science is studying the interplay between the structure of materials, the processing methods to make that material, and the resulting material properties. The complex combination of these produce the performance of a material in a specific application. Many features across many length scales impact material performance, from the constituent chemical elements, its microstructure, and macroscopic features from processing. Together with the laws of thermodynamics and kinetics materials scientists aim to understand and improve materials.
Structure
Structure is one of the most important components of the field of materials science. The very definition of the field holds that it is concerned with the investigation of "the relationships that exist between the structures and properties of materials".
Structure is studied in the following levels.
Atomic structure
Atomic structure deals with the atoms of the materials, and how they are arranged to give rise to molecules, crystals, etc. Much of the electrical, magnetic and chemical properties of materials arise from this level of structure. The length scales involved are in angstroms (Å). The chemical bonding and atomic arrangement (crystallography) are fundamental to studying the properties and behavior of any material.
Bonding
To obtain a full understanding of the material structure and how it relates to its properties, the materials scientist must study how the different atoms, ions and molecules are arranged and bonded to each other. This involves the study and use of
are also involved in the study of bonding and structure.Crystallography
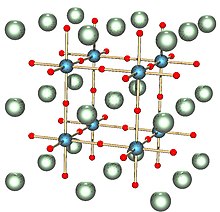
Crystallography is the science that examines the arrangement of atoms in crystalline solids. Crystallography is a useful tool for materials scientists. In single crystals, the effects of the crystalline arrangement of atoms is often easy to see macroscopically, because the natural shapes of crystals reflect the atomic structure. Further, physical properties are often controlled by crystalline defects. The understanding of crystal structures is an important prerequisite for understanding crystallographic defects. Mostly, materials do not occur as a single crystal, but in polycrystalline form, as an aggregate of small crystals or grains with different orientations. Because of this, the powder diffraction method, which uses diffraction patterns of polycrystalline samples with a large number of crystals, plays an important role in structural determination. Most materials have a crystalline structure, but some important materials do not exhibit regular crystal structure.[13] Polymers display varying degrees of crystallinity, and many are completely non-crystalline. Glass, some ceramics, and many natural materials are amorphous, not possessing any long-range order in their atomic arrangements. The study of polymers combines elements of chemical and statistical thermodynamics to give thermodynamic and mechanical descriptions of physical properties.
Nanostructure

Materials, which atoms and molecules form constituents in the nanoscale (i.e., they form nanostructure) are called nanomaterials. Nanomaterials are subject of intense research in the materials science community due to the unique properties that they exhibit.
Nanostructure deals with objects and structures that are in the 1 – 100 nm range.[14] In many materials, atoms or molecules agglomerate together to form objects at the nanoscale. This causes many interesting electrical, magnetic, optical, and mechanical properties.
In describing nanostructures, it is necessary to differentiate between the number of dimensions on the
Nanotextured surfaces have one dimension on the nanoscale, i.e., only the thickness of the surface of an object is between 0.1 and 100 nm.
Nanotubes have two dimensions on the nanoscale, i.e., the diameter of the tube is between 0.1 and 100 nm; its length could be much greater.
Finally, spherical nanoparticles have three dimensions on the nanoscale, i.e., the particle is between 0.1 and 100 nm in each spatial dimension. The terms nanoparticles and ultrafine particles (UFP) often are used synonymously although UFP can reach into the micrometre range. The term 'nanostructure' is often used, when referring to magnetic technology. Nanoscale structure in biology is often called ultrastructure.
Microstructure

Microstructure is defined as the structure of a prepared surface or thin foil of material as revealed by a microscope above 25× magnification. It deals with objects from 100 nm to a few cm. The microstructure of a material (which can be broadly classified into metallic, polymeric, ceramic and composite) can strongly influence physical properties such as strength, toughness, ductility, hardness, corrosion resistance, high/low temperature behavior, wear resistance, and so on.[15] Most of the traditional materials (such as metals and ceramics) are microstructured.
The manufacture of a perfect
Macrostructure
Macrostructure is the appearance of a material in the scale millimeters to meters, it is the structure of the material as seen with the naked eye.
Properties
Materials exhibit myriad properties, including the following.
- Mechanical properties, see Strength of materials
- Chemical properties, see Chemistry
- Electrical properties, see Electricity
- Thermal properties, see Thermodynamics
- Optical properties, see Optics and Photonics
- Magnetic properties, see Magnetism
The properties of a material determine its usability and hence its engineering application.
Processing
Synthesis and processing involves the creation of a material with the desired micro-nanostructure. A material cannot be used in industry if no economically viable production method for it has been developed. Therefore, developing processing methods for materials that are reasonably effective and cost-efficient is vital to the field of materials science. Different materials require different processing or synthesis methods. For example, the processing of metals has historically defined eras such as the Bronze Age and Iron Age and is studied under the branch of materials science named physical metallurgy. Chemical and physical methods are also used to synthesize other materials such as polymers, ceramics, semiconductors, and thin films. As of the early 21st century, new methods are being developed to synthesize nanomaterials such as graphene.
Thermodynamics
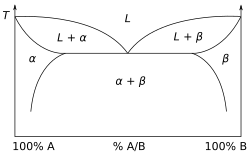
Thermodynamics is concerned with heat and temperature and their relation to energy and work. It defines macroscopic variables, such as internal energy, entropy, and pressure, that partly describe a body of matter or radiation. It states that the behavior of those variables is subject to general constraints common to all materials. These general constraints are expressed in the four laws of thermodynamics. Thermodynamics describes the bulk behavior of the body, not the microscopic behaviors of the very large numbers of its microscopic constituents, such as molecules. The behavior of these microscopic particles is described by, and the laws of thermodynamics are derived from, statistical mechanics.
The study of thermodynamics is fundamental to materials science. It forms the foundation to treat general phenomena in materials science and engineering, including chemical reactions, magnetism, polarizability, and elasticity.
Kinetics
Chemical kinetics is the study of the rates at which systems that are out of equilibrium change under the influence of various forces. When applied to materials science, it deals with how a material changes with time (moves from non-equilibrium to equilibrium state) due to application of a certain field. It details the rate of various processes evolving in materials including shape, size, composition and structure. Diffusion is important in the study of kinetics as this is the most common mechanism by which materials undergo change.[18] Kinetics is essential in processing of materials because, among other things, it details how the microstructure changes with application of heat.
Research
Materials science is a highly active area of research. Together with materials science departments, physics, chemistry, and many engineering departments are involved in materials research. Materials research covers a broad range of topics; the following non-exhaustive list highlights a few important research areas.
Nanomaterials
Nanomaterials describe, in principle, materials of which a single unit is sized (in at least one dimension) between 1 and 1000 nanometers (10−9 meter), but is usually 1 nm – 100 nm. Nanomaterials research takes a materials science based approach to nanotechnology, using advances in materials metrology and synthesis, which have been developed in support of microfabrication research. Materials with structure at the nanoscale often have unique optical, electronic, or mechanical properties. The field of nanomaterials is loosely organized, like the traditional field of chemistry, into organic (carbon-based) nanomaterials, such as fullerenes, and inorganic nanomaterials based on other elements, such as silicon. Examples of nanomaterials include fullerenes, carbon nanotubes, nanocrystals, etc.
Biomaterials

A biomaterial is any matter, surface, or construct that interacts with biological systems. The study of biomaterials is called bio materials science. It has experienced steady and strong growth over its history, with many companies investing large amounts of money into developing new products. Biomaterials science encompasses elements of medicine, biology, chemistry, tissue engineering, and materials science.
Biomaterials can be derived either from nature or synthesized in a laboratory using a variety of chemical approaches using metallic components,
Electronic, optical, and magnetic
Semiconductors, metals, and ceramics are used today to form highly complex systems, such as integrated electronic circuits, optoelectronic devices, and magnetic and optical mass storage media. These materials form the basis of our modern computing world, and hence research into these materials is of vital importance.
Semiconductors are a traditional example of these types of materials. They are materials that have properties that are intermediate between conductors and insulators. Their electrical conductivities are very sensitive to the concentration of impurities, which allows the use of doping to achieve desirable electronic properties. Hence, semiconductors form the basis of the traditional computer.
This field also includes new areas of research such as superconducting materials, spintronics, metamaterials, etc. The study of these materials involves knowledge of materials science and solid-state physics or condensed matter physics.
Computational materials science
With continuing increases in computing power, simulating the behavior of materials has become possible. This enables materials scientists to understand behavior and mechanisms, design new materials, and explain properties formerly poorly understood. Efforts surrounding
Industry

Radical
Besides material characterization, the material scientist or engineer also deals with extracting materials and converting them into useful forms. Thus ingot casting, foundry methods, blast furnace extraction, and electrolytic extraction are all part of the required knowledge of a materials engineer. Often the presence, absence, or variation of minute quantities of secondary elements and compounds in a bulk material will greatly affect the final properties of the materials produced. For example, steels are classified based on 1/10 and 1/100 weight percentages of the carbon and other alloying elements they contain. Thus, the extracting and purifying methods used to extract iron in a blast furnace can affect the quality of steel that is produced.
Solid materials are generally grouped into three basic classifications: ceramics, metals, and polymers. This broad classification is based on the empirical makeup and atomic structure of the solid materials, and most solids fall into one of these broad categories.[22] An item that is often made from each of these materials types is the beverage container. The material types used for beverage containers accordingly provide different advantages and disadvantages, depending on the material used. Ceramic (glass) containers are optically transparent, impervious to the passage of carbon dioxide, relatively inexpensive, and are easily recycled, but are also heavy and fracture easily. Metal (aluminum alloy) is relatively strong, is a good barrier to the diffusion of carbon dioxide, and is easily recycled. However, the cans are opaque, expensive to produce, and are easily dented and punctured. Polymers (polyethylene plastic) are relatively strong, can be optically transparent, are inexpensive and lightweight, and can be recyclable, but are not as impervious to the passage of carbon dioxide as aluminum and glass.
Ceramics and glasses

Another application of materials science is the study of ceramics and glasses, typically the most brittle materials with industrial relevance. Many ceramics and glasses exhibit covalent or ionic-covalent bonding with SiO2 (silica) as a fundamental building block. Ceramics – not to be confused with raw, unfired clay – are usually seen in crystalline form. The vast majority of commercial glasses contain a metal oxide fused with silica. At the high temperatures used to prepare glass, the material is a viscous liquid which solidifies into a disordered state upon cooling. Windowpanes and eyeglasses are important examples. Fibers of glass are also used for long-range telecommunication and optical transmission. Scratch resistant Corning Gorilla Glass is a well-known example of the application of materials science to drastically improve the properties of common components.
Engineering ceramics are known for their stiffness and stability under high temperatures, compression and electrical stress. Alumina, silicon carbide, and tungsten carbide are made from a fine powder of their constituents in a process of sintering with a binder. Hot pressing provides higher density material. Chemical vapor deposition can place a film of a ceramic on another material. Cermets are ceramic particles containing some metals. The wear resistance of tools is derived from cemented carbides with the metal phase of cobalt and nickel typically added to modify properties.
Ceramics can be significantly strengthened for engineering applications using the principle of
Composites

Another application of materials science in industry is making composite materials. These are structured materials composed of two or more macroscopic phases.
Applications range from structural elements such as steel-reinforced concrete, to the thermal insulating tiles, which play a key and integral role in NASA's
Other examples can be seen in the "plastic" casings of television sets, cell-phones and so on. These plastic casings are usually a composite material made up of a
Polymers
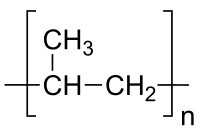

Polyvinyl chloride (PVC) is widely used, inexpensive, and annual production quantities are large. It lends itself to a vast array of applications, from
Specialty plastics are materials with unique characteristics, such as ultra-high strength, electrical conductivity, electro-fluorescence, high thermal stability, etc.
The dividing lines between the various types of plastics is not based on material but rather on their properties and applications. For example,
Metal alloys

The alloys of iron (steel, stainless steel, cast iron, tool steel, alloy steels) make up the largest proportion of metals today both by quantity and commercial value.
Iron alloyed with various proportions of carbon gives
Other significant metallic alloys are those of
Semiconductors
A
Of all the semiconductors in use today, silicon makes up the largest portion both by quantity and commercial value. Monocrystalline silicon is used to produce wafers used in the semiconductor and electronics industry. Gallium arsenide (GaAs) is the second most popular semiconductor used. Due to its higher electron mobility and saturation velocity compared to silicon, it is a material of choice for high-speed electronics applications. These superior properties are compelling reasons to use GaAs circuitry in mobile phones, satellite communications, microwave point-to-point links and higher frequency radar systems. Other semiconductor materials include germanium, silicon carbide, and gallium nitride and have various applications.
Relation with other fields

Materials science evolved, starting from the 1950s because it was recognized that to create, discover and design new materials, one had to approach it in a unified manner. Thus, materials science and engineering emerged in many ways: renaming and/or combining existing
The field of materials science and engineering is important both from a scientific perspective, as well as for applications field. Materials are of the utmost importance for engineers (or other applied fields) because usage of the appropriate materials is crucial when designing systems. As a result, materials science is an increasingly important part of an engineer's education.
Materials physics is the use of
The field is inherently
Emerging technologies
| Emerging technology | Status | Potentially marginalized technologies | Potential applications | Related articles |
|---|---|---|---|---|
| Aerogel | Hypothetical, experiments, diffusion,
early uses [31] |
Traditional insulation, glass | Improved insulation, insulative glass if it can be made clear, sleeves for oil pipelines, aerospace, high-heat & extreme cold applications | |
| Amorphous metal | Experiments | Kevlar | Armor | |
| Conductive polymers | Research, experiments, prototypes | Conductors | Lighter and cheaper wires, antistatic materials, organic solar cells | |
| Femtotechnology, picotechnology | Hypothetical | Present nuclear | New materials; nuclear weapons, power | |
| Fullerene | Experiments, diffusion | Synthetic diamond and carbon nanotubes (Buckypaper) | Programmable matter | |
| Graphene | Hypothetical, experiments, diffusion, | Silicon-based integrated circuit | Components with higher strength to weight ratios, transistors that operate at higher frequency, lower cost of display screens in mobile devices, storing hydrogen for fuel cell powered cars, filtration systems, longer-lasting and faster-charging batteries, sensors to diagnose diseases[34] | Potential applications of graphene |
| High-temperature superconductivity | Cryogenic receiver front-end (CRFE) RF and microwave filter systems for mobile phone base stations; prototypes in dry ice; Hypothetical and experiments for higher temperatures [35] | Copper wire, semiconductor integral circuits | No loss conductors, frictionless bearings, magnetic levitation, lossless high-capacity accumulators, electric cars, heat-free integral circuits and processors | |
| LiTraCon | Experiments, already used to make Europe Gate | Glass | Building skyscrapers, towers, and sculptures like Europe Gate | |
| Metamaterials | Hypothetical, experiments, diffusion [36] | Classical optics | Microscopes, cameras, metamaterial cloaking, cloaking devices | |
| Metal foam | Research, commercialization | Hulls | Space colonies, floating cities | |
| Multi function structures[37] | Hypothetical, experiments, some prototypes, few commercial | Composite materials | Wide range, e.g., self-health monitoring, self-healing material, morphing | |
| Nanomaterials: carbon nanotubes | Hypothetical, experiments, diffusion, | Structural steel and aluminium | Stronger, lighter materials, the space elevator | carbon fiber
|
| Programmable matter | Hypothetical, experiments[40][41] | catalysts
|
Wide range, e.g., claytronics, synthetic biology
|
|
| Quantum dots | Research, experiments, prototypes[42] | LCD, LED
|
Quantum dot laser, future use as programmable matter in display technologies (TV, projection), optical data communications (high-speed data transmission), medicine (laser scalpel) | |
| Silicene | Hypothetical, research | Field-effect transistors |
Subdisciplines
The main branches of materials science stem from the four main classes of materials: ceramics, metals, polymers and composites.
There are additionally broadly applicable, materials independent, endeavors.
- Materials characterization (spectroscopy, microscopy, diffraction)
- Computational materials science
- Materials informatics and selection
There are also relatively broad focuses across materials on specific phenomena and techniques.
Related or interdisciplinary fields
- Condensed matter physics, solid-state physics and solid-state chemistry
- Nanotechnology
- Mineralogy
- Supramolecular chemistry
- Biomaterials science
Professional societies
- American Ceramic Society
- ASM International
- Association for Iron and Steel Technology
- Materials Research Society
- The Minerals, Metals & Materials Society
See also
References
Citations
- ^ Eddy, Matthew Daniel (2008). The Language of Mineralogy: John Walker, Chemistry and the Edinburgh Medical School 1750–1800. Ashgate Publishing. Archived from the original on 2015-09-03 – via Academia.edu.
- ISBN 978-0262191913.
- ^ ISBN 978-3-11-066999-2.
- ISBN 978-94-007-7807-8.
- S2CID 117809375.
- ^ ISBN 978-1-351-97740-1.
- ^ "For Authors: Nature Materials" Archived 2010-08-01 at the Wayback Machine
- ^ Callister, Jr., Rethwisch. "Materials Science and Engineering – An Introduction" (8th ed.). John Wiley and Sons, 2009 pp.5–6
- ^ Callister, Jr., Rethwisch. Materials Science and Engineering – An Introduction (8th ed.). John Wiley and Sons, 2009 pp.10–12
- ISSN 0897-4756.
- ISBN 978-0-08-044552-6.
- .
- ISSN 0001-4842.
- from the original on 2012-07-03.
- ISSN 0924-0136.
- S2CID 244023491, retrieved 2023-10-29
- S2CID 225430517.
- ISBN 978-3-527-31024-1.
- doi:10.1063/1.1343489. Archived from the original(PDF) on June 18, 2010.
- PMID 10990641.
- S2CID 199492241.
- ISBN 9780470419977.
- ISSN 0001-6160.
- ISSN 0001-6160.
- S2CID 31152759.
- PMID 25796128.
- S2CID 260266502.
- ISSN 0261-3069.
- ^ "Semiconductor Market Size, Share & COVID-19 Impact Analysis, By Component (Memory Devices, Logic Devices, Analog IC, MPU, Discrete Power Devices, MCU, Sensors, and Others), By Application (Networking & Communications, Data Processing, Industrial, Consumer Electronics, Automotive, and Government), and Regional Forecast, 2022–2029". Fortune Business Insights. 16 July 2023. Archived from the original on 11 June 2023. Retrieved 16 July 2023.
- ^ "Semiconductor Industry Careers". 2013-09-06. Archived from the original on 2016-06-04. Retrieved 2016-05-15.
- ^ "Sto AG, Cabot Create Aerogel Insulation". Construction Digital. 15 November 2011. Archived from the original on 31 December 2011. Retrieved 18 November 2011.
- ^ "Is graphene a miracle material?". BBC Click. 21 May 2011. Retrieved 18 November 2011.
- ^ "Could graphene be the new silicon?". The Guardian. 13 November 2011. Archived from the original on 2 September 2013. Retrieved 18 November 2011.
- ^ "Applications of Graphene under Development". understandingnano.com. Archived from the original on 2014-09-21.
- ^ "The 'new age' of super materials". BBC News. 5 March 2007. Retrieved 27 April 2011.
- ^ "Strides in Materials, but No Invisibility Cloak". The New York Times. 8 November 2010. Archived from the original on 1 July 2017. Retrieved 21 April 2011.
- ^ NAE Website: Frontiers of Engineering Archived 2014-07-28 at the Wayback Machine. Nae.edu. Retrieved 22 February 2011.
- ^ "Carbon nanotubes used to make batteries from fabrics". BBC News. 21 January 2010. Retrieved 27 April 2011.
- ^ "Researchers One Step Closer to Building Synthetic Brain". Daily Tech. 25 April 2011. Archived from the original on 29 April 2011. Retrieved 27 April 2011.
- ^ "Pentagon Developing Shape-Shifting 'Transformers' for Battlefield". Fox News. 10 June 2009. Archived from the original on 5 February 2011. Retrieved 26 April 2011.
- ^ "Intel: Programmable matter takes shape". ZD Net. 22 August 2008. Retrieved 2 January 2012.
- ^ "'Quantum dots' to boost performance of mobile cameras". BBC News. 22 March 2010. Retrieved 16 April 2011.
Bibliography
- Ashby, Michael; Hugh Shercliff; David Cebon (2007). Materials: engineering, science, processing and design (1st ed.). Butterworth-Heinemann. ISBN 978-0-7506-8391-3.
- Askeland, Donald R.; Pradeep P. Phulé (2005). The Science & Engineering of Materials (5th ed.). Thomson-Engineering. ISBN 978-0-534-55396-8.
- Callister, Jr., William D. (2000). Materials Science and Engineering – An Introduction (5th ed.). John Wiley and Sons. ISBN 978-0-471-32013-5.
- ISBN 978-1-4000-4760-4.
- Gaskell, David R. (1995). Introduction to the Thermodynamics of Materials (4th ed.). Taylor and Francis Publishing. ISBN 978-1-56032-992-3.
- González-Viñas, W. & Mancini, H.L. (2004). An Introduction to Materials Science. Princeton University Press. ISBN 978-0-691-07097-1.
- ISBN 978-0-691-02380-9.
- Mathews, F.L. & Rawlings, R.D. (1999). Composite Materials: Engineering and Science. Boca Raton: CRC Press. ISBN 978-0-8493-0621-1.
- Lewis, P.R.; Reynolds, K. & Gagg, C. (2003). Forensic Materials Engineering: Case Studies. Boca Raton: CRC Press. ISBN 9780849311826.
- Wachtman, John B. (1996). Mechanical Properties of Ceramics. New York: Wiley-Interscience, John Wiley & Son's. ISBN 978-0-471-13316-2.
- Walker, P., ed. (1993). Chambers Dictionary of Materials Science and Technology. Chambers Publishing. ISBN 978-0-550-13249-9.
- Mahajan, S. (2015). "The role of materials science in the evolution of microelectronics". MRS Bulletin. 12 (40): 1079–1088. .
Further reading
- Timeline of Materials Science at The Minerals, Metals & Materials Society (TMS) – accessed March 2007
- Burns, G.; Glazer, A.M. (1990). Space Groups for Scientists and Engineers (2nd ed.). Boston: Academic Press, Inc. ISBN 978-0-12-145761-7.
- Cullity, B.D. (1978). Elements of X-Ray Diffraction (2nd ed.). Reading, Massachusetts: Addison-Wesley Publishing Company. ISBN 978-0-534-55396-8.
- Giacovazzo, C; Monaco HL; Viterbo D; Scordari F; Gilli G; Zanotti G; Catti M (1992). Fundamentals of Crystallography. Oxford: ISBN 978-0-19-855578-0.
- Green, D.J.; Hannink, R.; Swain, M.V. (1989). Transformation Toughening of Ceramics. Boca Raton: CRC Press. ISBN 978-0-8493-6594-2.
- Lovesey, S. W. (1984). Theory of Neutron Scattering from Condensed Matter; Volume 1: Neutron Scattering. Oxford: Clarendon Press. ISBN 978-0-19-852015-3.
- Lovesey, S. W. (1984). Theory of Neutron Scattering from Condensed Matter; Volume 2: Condensed Matter. Oxford: Clarendon Press. ISBN 978-0-19-852017-7.
- O'Keeffe, M.; Hyde, B.G. (1996). "Crystal Structures; I. Patterns and Symmetry". ISBN 978-0-939950-40-9.
- Squires, G.L. (1996). Introduction to the Theory of Thermal Neutron Scattering (2nd ed.). Mineola, New York: Dover Publications Inc. ISBN 978-0-486-69447-4.
- Young, R.A., ed. (1993). The Rietveld Method. Oxford: Oxford University Press & International Union of Crystallography. ISBN 978-0-19-855577-3.

