Matrix-assisted laser desorption/ionization

In mass spectrometry, matrix-assisted laser desorption/ionization (MALDI) is an ionization technique that uses a laser energy-absorbing matrix to create ions from large molecules with minimal fragmentation.[1] It has been applied to the analysis of biomolecules (biopolymers such as DNA, proteins, peptides and carbohydrates) and various organic molecules (such as polymers, dendrimers and other macromolecules), which tend to be fragile and fragment when ionized by more conventional ionization methods. It is similar in character to electrospray ionization (ESI) in that both techniques are relatively soft (low fragmentation) ways of obtaining ions of large molecules in the gas phase, though MALDI typically produces far fewer multi-charged ions.
MALDI methodology is a three-step process. First, the sample is mixed with a suitable matrix material and applied to a metal plate. Second, a pulsed laser irradiates the sample, triggering ablation and desorption of the sample and matrix material. Finally, the analyte molecules are ionized by being protonated or deprotonated in the hot plume of ablated gases, and then they can be accelerated into whichever mass spectrometer is used to analyse them.[2]
History
The term matrix-assisted laser desorption ionization (MALDI) was coined in 1985 by Franz Hillenkamp, Michael Karas and their colleagues.[3] These researchers found that the amino acid alanine could be ionized more easily if it was mixed with the amino acid tryptophan and irradiated with a pulsed 266 nm laser. The tryptophan was absorbing the laser energy and helping to ionize the non-absorbing alanine. Peptides up to the 2843 Da peptide melittin could be ionized when mixed with this kind of "matrix".[4] The breakthrough for large molecule laser desorption ionization came in 1987 when Koichi Tanaka of Shimadzu Corporation and his co-workers used what they called the "ultra fine metal plus liquid matrix method" that combined 30 nm cobalt particles in glycerol with a 337 nm nitrogen laser for ionization.[5] Using this laser and matrix combination, Tanaka was able to ionize biomolecules as large as the 34,472 Da protein carboxypeptidase-A. Tanaka received one-quarter of the 2002 Nobel Prize in Chemistry for demonstrating that, with the proper combination of laser wavelength and matrix, a protein can be ionized.[6] Karas and Hillenkamp were subsequently able to ionize the 67 kDa protein albumin using a nicotinic acid matrix and a 266 nm laser.[7] Further improvements were realized through the use of a 355 nm laser and the cinnamic acid derivatives ferulic acid, caffeic acid and sinapinic acid as the matrix.[8] The availability of small and relatively inexpensive nitrogen lasers operating at 337 nm wavelength and the first commercial instruments introduced in the early 1990s brought MALDI to an increasing number of researchers.[9] Today, mostly organic matrices are used for MALDI mass spectrometry.
Matrix
| Compound | Other names | Solvent | Wavelength (nm) | Applications |
|---|---|---|---|---|
| 2,5-dihydroxy benzoic acid (gentisic acid)[10] | DHB, gentisic acid | acetonitrile, water, methanol, acetone, chloroform | 337, 355, 266 | peptides, nucleotides, oligonucleotides, oligosaccharides |
| 3,5-dimethoxy-4-hydroxycinnamic acid[8][11] | sinapic acid; sinapinic acid; SA | acetonitrile, water, acetone, chloroform | 337, 355, 266 | peptides, proteins, lipids |
| 4-hydroxy-3-methoxycinnamic acid[8][11] | ferulic acid | acetonitrile, water, propanol | 337, 355, 266 | proteins |
α-cyano-4-hydroxycinnamic acid[12]
|
CHCA | acetonitrile, water, ethanol, acetone | 337, 355 | peptides, lipids, nucleotides |
| Picolinic acid[13] | PA | Ethanol | 266 | oligonucleotides |
3-hydroxy picolinic acid[14]
|
HPA | Ethanol | 337, 355 | oligonucleotides |
The

The identification of suitable matrix compounds is determined to some extent by trial and error, but they are based on some specific molecular design considerations. They are of a fairly low molecular weight (to allow easy vaporization), but are large enough (with a low enough vapor pressure) not to evaporate during sample preparation or while standing in the mass spectrometer. They are often acidic, therefore act as a proton source to encourage ionization of the analyte. Basic matrices have also been reported.
The matrix solution is mixed with the analyte (e.g.
In analysis of biological systems, inorganic salts, which are also part of protein extracts, interfere with the ionization process. The salts can be removed by solid phase extraction or by washing the dried-droplet MALDI spots with cold water.[18] Both methods can also remove other substances from the sample. The matrix-protein mixture is not homogeneous because the polarity difference leads to a separation of the two substances during co-crystallization. The spot diameter of the target is much larger than that of the laser, which makes it necessary to make many laser shots at different places of the target, to get the statistical average of the substance concentration within the target spot.
The matrix can be used to tune the instrument to ionize the sample in different ways. As mentioned above, acid-base like reactions are often utilized to ionize the sample, however, molecules with conjugated pi systems, such as naphthalene like compounds, can also serve as an electron acceptor and thus a matrix for MALDI/TOF.[19] This is particularly useful in studying molecules that also possess conjugated pi systems.[20] The most widely used application for these matrices is studying porphyrin-like compounds such as chlorophyll. These matrices have been shown to have better ionization patterns that do not result in odd fragmentation patterns or complete loss of side chains.[21] It has also been suggested that conjugated porphyrin like molecules can serve as a matrix and cleave themselves eliminating the need for a separate matrix compound.[22]
Instrumentation
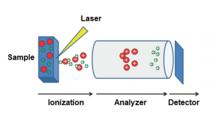
There are several variations of the MALDI technology and comparable instruments are today produced for very different purposes, from more academic and analytical, to more industrial and high throughput. The mass spectrometry field has expanded into requiring ultrahigh resolution mass spectrometry such as the FT-ICR instruments
Laser
MALDI techniques typically employ the use of UV
Infrared laser wavelengths used for infrared MALDI include the 2.94 μm
Time of flight

The type of a mass spectrometer most widely used with MALDI is the time-of-flight mass spectrometer (TOF), mainly due to its large mass range. The TOF measurement procedure is also ideally suited to the MALDI ionization process since the pulsed laser takes individual 'shots' rather than working in continuous operation. MALDI-TOF instruments are often equipped with a reflectron (an "ion mirror") that reflects ions using an electric field. This increases the ion flight path, thereby increasing time of flight between ions of different m/z and increasing resolution. Modern commercial reflectron TOF instruments reach a resolving power m/Δm of 50,000 FWHM (full-width half-maximum, Δm defined as the peak width at 50% of peak height) or more.[28]
MALDI has been coupled with
MALDI-
Atmospheric pressure
Atmospheric pressure (AP) matrix-assisted laser desorption/ionization (MALDI) is an ionization technique (ion source) that in contrast to vacuum MALDI operates at normal atmospheric environment.[32] The main difference between vacuum MALDI and AP-MALDI is the pressure in which the ions are created. In vacuum MALDI, ions are typically produced at 10 mTorr or less while in AP-MALDI ions are formed in atmospheric pressure. In the past, the main disadvantage of the AP-MALDI technique compared to the conventional vacuum MALDI has been its limited sensitivity; however, ions can be transferred into the mass spectrometer with high efficiency and attomole detection limits have been reported.[33] AP-MALDI is used in mass spectrometry (MS) in a variety of applications ranging from proteomics to drug discovery. Popular topics that are addressed by AP-MALDI mass spectrometry include: proteomics; mass analysis of DNA, RNA, PNA, lipids, oligosaccharides, phosphopeptides, bacteria, small molecules and synthetic polymers, similar applications as available also for vacuum MALDI instruments. The AP-MALDI ion source is easily coupled to an ion trap mass spectrometer[34] or any other MS system equipped with electrospray ionization (ESI) or nanoESI source.
MALDI with ionization at reduced pressure is known to produce mainly singly-charged ions (see "Ionization mechanism" below). In contrast, ionization at atmopsheric pressure can generate highly-charged analytes as was first shown for infrared [35] and later also for nitrogen lasers.[36] Multiple charging of analytes is of great importance, because it allows to measure high-molecular-weight compounds like proteins in instruments, which provide only smaller m/z detection ranges such as quadrupoles. Besides the pressure, the composition of the matrix is important to achieve this effect.
Aerosol
In aerosol mass spectrometry, one of the ionization techniques consists in firing a laser to individual droplets. These systems are called single particle mass spectrometers (SPMS).[37] The sample may optionally be mixed with a MALDI matrix prior to aerosolization.
Ionization mechanism
The
The gas phase proton transfer model,[2] implemented as the coupled physical and chemical dynamics (CPCD) model,[39] of UV laser MALDI postulates primary and secondary processes leading to ionization.[40] Primary processes involve initial charge separation through absorption of photons by the matrix and pooling of the energy to form matrix ion pairs. Primary ion formation occurs through absorption of a UV photon to create excited state molecules by
- S0 + hν → S1
- S1 + S1 → S0 + Sn
- S1 + Sn → M+ + M−
where S0 is the ground electronic state, S1 the first electronic excited state, and Sn is a higher electronic excited state.[39] The product ions can be proton transfer or electron transfer ion pairs, indicated by M+ and M− above. Secondary processes involve ion-molecule reactions to form analyte ions.

The lucky survivor model (cluster ionization mechanism
The thermal model postulates that the high temperature facilitates the proton transfer between matrix and analyte in melted matrix liquid.[43] Ion-to-neutral ratio is an important parameter to justify the theoretical model, and the mistaken citation of ion-to-neutral ratio could result in an erroneous determination of the ionization mechanism.[44] The model quantitatively predicts the increase in total ion intensity as a function of the concentration and proton affinity of the analytes, and the ion-to-neutral ratio as a function of the laser fluences.[45][46] This model also suggests that metal ion adducts (e.g., [M+Na]+ or [M+K]+) are mainly generated from the thermally induced dissolution of salt.[47]
The matrix-assisted ionization (MAI) method uses matrix preparation similar to MALDI but does not require laser ablation to produce analyte ions of volatile or nonvolatile compounds.[48] Simply exposing the matrix with analyte to the vacuum of the mass spectrometer creates ions with nearly identical charge states to electrospray ionization.[49] It is suggested that there are likely mechanistic commonality between this process and MALDI.[42]
Ion yield is typically estimated to range from 10−4 to 10−7,[50] with some experiments hinting to even lower yields of 10−9.[51] The issue of low ion yields had been addressed, already shortly after introduction of MALDI by various attempts, including post-ionization utilizing a second laser.[52] Most of these attempts showed only limited success, with low signal increases. This might be attributed to the fact that axial time-of-flight instruments were used, which operate at pressures in the source region of 10−5 to 10−6, which results in rapid plume expansion with particle velocities of up to 1000 m/s.[53] In 2015, successful laser post-ionization was reported, using a modified MALDI source operated at an elevated pressure of ~3 mbar coupled to an orthogonal time-of-flight mass analyzer, and employing a wavelength-tunable post-ionization laser, operated at wavelength from 260 nm to 280 nm, below the two-photon ionization threshold of the matrices used, which elevated ion yields of several lipids and small molecules by up to three orders of magnitude.[54] This approach, called MALDI-2, due to the second laser, and the second MALDI-like ionization process, was afterwards adopted for other mass spectrometers, all equipped with sources operating in the low mbar range.[55][56]
Applications
Biochemistry
In
MALDI-TOF have been used to characterise
Besides proteins, MALDI-TOF has also been applied to study
Organic chemistry
Some synthetic macromolecules, such as catenanes and rotaxanes, dendrimers and hyperbranched polymers, and other assemblies, have molecular weights extending into the thousands or tens of thousands, where most ionization techniques have difficulty producing molecular ions. MALDI is a simple and fast analytical method that can allow chemists to rapidly analyze the results of such syntheses and verify their results.[citation needed]
Polymers
In polymer chemistry, MALDI can be used to determine the
A good matrix for polymers is dithranol[74] or AgTFA.[75] The sample must first be mixed with dithranol and the AgTFA added afterwards; otherwise the sample will precipitate out of solution.
Microbiology
MALDI-TOF spectra are often used for the identification of microorganisms such as bacteria or fungi. A portion of a colony of the microbe in question is placed onto the sample target and overlaid with matrix. The mass spectra of expressed proteins generated are analyzed by dedicated software and compared with stored profiles for species determination in what is known as biotyping. It offers benefits to other immunological or biochemical procedures and has become a common method for species identification in clinical microbiological laboratories.[76][77] Benefits of high resolution MALDI-MS performed on a Fourier transform ion cyclotron resonance mass spectrometry (also known as FT-MS) have been demonstrated for typing and subtyping viruses though single ion detection known as proteotyping, with a particular focus on influenza viruses.[78]
One main advantage over other microbiological identification methods is its ability to rapidly and reliably identify, at low cost, a wide variety of microorganisms directly from the selective medium used to isolate them. The absence of the need to purify the suspect or "presumptive" colony[79] allows for a much faster turn-around times. For example, it has been demonstrated that MALDI-TOF can be used to detect bacteria directly from blood cultures.[80]
Another advantage is the potential to predict
Parasitology
MALDI-TOF spectra have been used for the detection and identification of various parasites such as
Medicine
MALDI-TOF spectra are often utilized in tandem with other analysis and spectroscopy techniques in the diagnosis of diseases. MALDI/TOF is a diagnostic tool with much potential because it allows for the rapid identification of proteins and changes to proteins without the cost or computing power of
One example of this is necrotizing enterocolitis (NEC), which is a devastating disease that affects the bowels of premature infants. The symptoms of NEC are very similar to those of sepsis, and many infants die awaiting diagnosis and treatment. MALDI/TOF was used to identify bacteria present in the fecal matter of NEC positive infants. This study focused on characterization of the fecal microbiota associated with NEC and did not address the mechanism of disease. There is hope that a similar technique could be used as a quick, diagnostic tool that would not require sequencing.[91]
Another example of the diagnostic power of MALDI/TOF is in the area of
MALDI/TOF can also potentially be used to dictate treatment as well as diagnosis. MALDI/TOF serves as a method for determining the
Detection of protein complexes
Following initial observations that some peptide-peptide complexes could survive MALDI deposition and ionization,[96] studies of large protein complexes using MALDI-MS have been reported.[97][98]
Small molecules
While MALDI is a common technique for large macro-molecules, it is often possible to also analyze small molecules with mass below 1000 Da. The problem with small molecules is that of matrix effects, where signal interference, detector saturation, or suppression of the analyte signal is possible since the matrices often consists of small molecules themselves. The choice of matrix is highly dependent on what molecules are to be analyzed.[99][100]
See also
References
- PMID 1789447.
- ^ PMID 12580637.
- .
- .
- .
- The Royal Swedish Academy of Sciences. pp. 1–13. Retrieved 2013-08-28.
- PMID 3239801.
- ^ PMID 2520224.
- .
- .
- ^ PMID 2520223.
- .
- PMID 7949335.
- PMID 8457722.
- ISBN 9781420092219.
- PMID 8291672.
- .
- PMID 14528495.
- PMID 22899508.
- S2CID 2634483.
- S2CID 43158125.
- .
- ^ "Talking About a Revolution: FT-ICR Mass Spectrometry Offers High Resolution and Mass Accuracy for Pr".
- PMC 2129108.
- PMID 21699925.
- S2CID 5467069.
- ISBN 9780080438016. Retrieved 2017-04-05.
- PMID 22263633.
- PMID 15538797.(subscription required)
- PMID 12645885.(subscription required)
- .(subscription required)
- PMID 10701247.
- Thermo Scientific. Retrieved 17 June 2011.
- PMID 11080870.
- PMID 17569505.
- PMID 19904915.
- .
- PMID 17047796.
- ^ PMID 27070182.
- PMID 17047796.
- PMID 10633229.
- ^ ISSN 1387-3806.
- PMID 26132345.
- PMID 23592197.
- PMID 24707818.
- S2CID 24085468.
- S2CID 2139197.
- PMID 26486514.
- S2CID 29978978.
- S2CID 24085468.
- S2CID 96435099.
- ISSN 1073-9149.
- PMID 12580636.
- S2CID 206632790.
- S2CID 218874538.
- PMID 28573274.
- PMID 25778392.
- S2CID 4021309.
- PMID 8250262.
- PMID 16053310.
- PMID 18275166.
- PMID 8797382.
- PMID 10639030.
- PMID 17465012.
- PMID 19627133.
- PMID 24797946.
- PMID 20886850.
- S2CID 231584395.
- S2CID 18280663.
- ISBN 978-3-540-44259-2.
- .
- PMID 9666717.
- .
- S2CID 5305444.
- ISSN 0003-2700.
- PMID 19583519.
- PMID 22996584.
- PMID 23632861.
- ISBN 978-0-12-416999-9.)
{{cite book}}: CS1 maint: multiple names: authors list (link - PMID 30383194.
- PMID 27568365.
- PMID 24930405.
- S2CID 20480725.
- PMID 28274205.
- S2CID 43633405.
- PMID 27659938.
- PMID 28724559.
- PMID 28049524.
- PMID 32351208.

- PMID 30838972.

- ^ Sim, K.; Shaw, A. G.; Randell, P.; Cox, M. J.; McClure, Z. E.; Li, M. S.; Haddad, M.; Langford, P. R.; Cookson, W. O.; Moffatt, M. F.; Kroll, J. S. Dysbiosis anticipating necrotizing enterocolitis in very premature infants. Clin. Infect. Dis. 2014.
- S2CID 29118712.
- S2CID 17464318.
- ^ Zhong, N.; Cui, Y.; Zhou, X.; Li, T.; Han, J. Identification of prohibitin 1 as a potential prognostic biomarker in human pancreatic carcinoma using modified aqueous two-phase partition system combined with 2D-MALDI-TOF-TOF-MS/MS. Tumour Biol. 2014.
- ^ Hrabák, Jaroslav (2015). "Detection of Carbapenemases Using Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF MS) Meropenem Hydrolysis Assay". Methods in Molecular Biology (1064-3745), 1237, p. 91.
- .
- S2CID 33248551.
- ISBN 9780470146330.
- ISSN 1618-2642.
- ISSN 1618-2642.
Bibliography
- Ragoussis, J.; Elvidge, G. P.; Kaur, K.; Colella, S. (2006). "Matrix-Assisted Laser Desorption/Ionisation, Time-of-Flight Mass Spectrometry in Genomics Research". PMID 16895448.
- Hardouin, J. (2007). "Protein sequence information by matrix-assisted laser desorption/ionization in-source decay mass spectrometry". PMID 17492750.
- Peter-Katalinic, J.; Hillenkamp, F. (2007). MALDI MS: A Practical Guide to Instrumentation, Methods and Applications. ISBN 978-3-527-31440-9.
- Schrepp, W.; Pasch, H. (2003). MALDI-TOF Mass Spectrometry of Synthetic Polymers. ISBN 978-3-540-44259-2.
External links
- Primer on Matrix-Assisted Laser Desorption Ionization (MALDI) National High Magnetic Field Laboratory
