McFadyen–Stevens reaction
The McFadyen–Stevens reaction is a chemical reaction best described as a base-catalyzed thermal decomposition of acylsulfonylhydrazides to aldehydes.[1][2]

Dudman et al. have developed an alternative hydrazide reagent.[3]
Reaction mechanism
The mechanism of the McFadyen–Stevens reaction is still under investigation. Two groups have independently proposed a heterolytic fragmentation mechanism.[4][5] The mechanism involves the deprotonation of the acyl sulfonamide followed by a 1,2-hydride migration to give the alkoxide (3). The collapse of the alkoxide results in the fragmentation producing the desired aldehyde (4),
nitrogen gas
, and an aryl sulfinate ion (5).
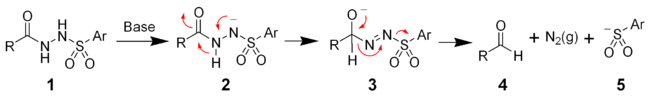
Martin et al. have proposed a different mechanism involving an
See also
References
- .
- ISBN 0471264180.
- .
- .
- PMID 13807198.
- .
