McMurry reaction
| McMurry reaction | |
|---|---|
| Named after | John E. McMurry |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | mcmurry-reaction |

The McMurry reaction is an organic reaction in which two ketone or aldehyde groups are coupled to form an alkene using a titanium chloride compound such as titanium(III) chloride and a reducing agent. The reaction is named after its co-discoverer, John E. McMurry. The McMurry reaction originally involved the use of a mixture TiCl3 and LiAlH4, which produces the active reagents. Related species have been developed involving the combination of TiCl3 or TiCl4 with various other reducing agents, including potassium, zinc, and magnesium.[1][2] This reaction is related to the Pinacol coupling reaction which also proceeds by reductive coupling of carbonyl compounds.
Reaction mechanism
This reductive coupling can be viewed as involving two steps. First is the formation of a pinacolate (1,2-diolate) complex, a step which is equivalent to the pinacol coupling reaction. The second step is the deoxygenation of the pinacolate, which yields the alkene, this second step exploits the oxophilicity of titanium.
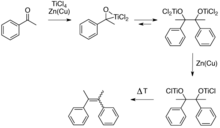
Several mechanisms have been discussed for this reaction.
Background and scope
The original publication by Mukaiyama demonstrated reductive coupling of ketones using reduced titanium reagents.

In another example, the
Further reading
- John E. McMurry; Michael P. Fleming (1974). "New Method for the Reductive Coupling of Carbonyls to Olefins. Synthesis of β-Carotene". PMID 4850242.
References
- ISBN 0471936235.
- ISBN 0471936235.
- ^ doi:10.1039/a804394i.
- ^ .
- .
- PMID 14653742.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .

