Meclizine
 | |
| Clinical data | |
|---|---|
| Trade names | Bonine, Antivert, others |
| Other names | Meclozine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682548 |
| License data | |
under the tongue, in the cheek | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Metabolism | Liver |
| Elimination half-life | 6 hours |
| Identifiers | |
| |
JSmol) | |
| Boiling point | 230 °C (446 °F) |
| |
| |
| | |
Meclizine, sold under the brand name Bonine, among others, is an antihistamine used to treat motion sickness and dizziness (vertigo).[1] It is taken by mouth.[1] Effects generally begin in an hour and last for up to a day.[1]
Common side effects include sleepiness and dry mouth.
Meclizine was patented in 1951 and came into medical use in 1953.
Medical uses
Meclizine is used to treat symptoms of
Motion sickness
Meclizine is effective in inhibiting nausea, vomiting, and dizziness caused by motion sickness.[8]
The drug is safe for treating nausea in pregnancy and is a first-line therapy for this use.[9][10] Doxylamine is similarly safe. Meclizine may not be strong enough for especially sickening motion stimuli, and second-line defenses should be tried in those cases.[11]
Vertigo
Meclizine may be used to treat vertigo, such as in those with Ménière's disease.[12][13]
Side effects
Some common side effects such as drowsiness, dry mouth, and tiredness may occur. Meclizine has been shown to have fewer dry mouth side effects than the traditional treatment for motion sickness, transdermal
Drowsiness
Drowsiness may result as a side effect of taking meclizine. Users are advised not to operate heavy machinery while under the influence. The consumption of alcohol while under the influence of meclizine may result in additional drowsiness.[1]
Elderly
As with any
Mechanism of action
Meclizine is an antagonist at H1 receptors. It possesses
Chemistry
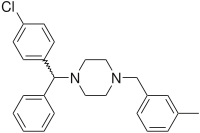
Meclizine is a
Synthesis
(4-Chlorophenyl)-phenylmethanol is halogenated with thionyl chloride before adding acetylpiperazine. The acetyl group is cleaved with diluted sulfuric acid. An N-alkylation of the piperazine ring with 3-methylbenzylchloride completes the synthesis.[19]
Alternatively, the last step can be replaced by a reductive N-alkylation with 3-methylbenzaldehyde. The reductive agent is
Meclizine is obtained and used as a
Names
Meclizine is an international nonproprietary name.[22]
It is sold under the brand names Bonine, Bonamine, Antivert, Postafen, Sea Legs, and Dramamine II (Less Drowsy Formulation). Emesafene is a combination of meclizine (1/3) and
Notes
References
- ^ a b c d e f g h "Meclizine Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 22 March 2019.
- ^ "Meclizine Use During Pregnancy". Drugs.com. Retrieved 3 March 2019.
- ISBN 9783527607495.
- ISBN 9783318056372.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Meclizine - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ PMID 32809480.
- ^ "Drugs & Medications". www.webmd.com. Retrieved 28 December 2018.
- ^ "Antiemetische Therapie bei Schwangerschaftserbrechen" [Antiemetic therapy in pregnancy]. Arznei-Telegramm (in German). 40: 87–89. 2009.
- ^ "Meclozin" [Federal Ministry of Health]. Embryotox (in German). Bundesministerium für Gesundheit.
- ^ Lawson BD, McGee HA, Castaneda MA, Golding JF, Kass SJ, McGrath CM (2 December 2009). "Evaluation of several common antimotion sickness medications and recommendations concerning their potential usefulness during special operations". Pensacola, Florida: Naval Aerospace Research Lab. Archived from the original on 27 April 2016. Retrieved 7 February 2016.
- S2CID 3987838.
- PMID 31643231.
- S2CID 40691502.
- ^ "Meclizine - oral, Antivert, D-vert, Dramamine II". MedicineNet. Retrieved 7 November 2010.
- ^ "Meclizine". Drug Information Provided by Lexi-Comp. January 2010. Archived from the original on 22 August 2009. Retrieved 7 November 2010 – via Merck Manuals, Online Medical Library.
- PMID 32077140.
- ^ PMID 9193868. Archived from the originalon 28 August 2021. Retrieved 12 June 2014.
- ISBN 978-3-527-30272-7.
- ^ US 2709169, Morren H, issued 24 May 1955, assigned to Union Chimique Belge Société Anonyme
- ISBN 3-13-115134-X.
- ^ "Guidelines on the Use of INNs for Pharmaceutical Substances" (PDF). WHO. 1997. Retrieved 1 November 2013.
- ^ Wishard D. "Drug card for Meclizine". DrugBank. Canada: University of Alberta. Retrieved 7 November 2010.


