Medical genetics
| Part of a series on |
| Genetics |
|---|
 |
Medical genetics is the branch of
In contrast, the study of typically non-medical phenotypes such as the genetics of eye color would be considered part of human genetics, but not necessarily relevant to medical genetics (except in situations such as albinism). Genetic medicine is a newer term for medical genetics and incorporates areas such as gene therapy, personalized medicine, and the rapidly emerging new medical specialty, predictive medicine.
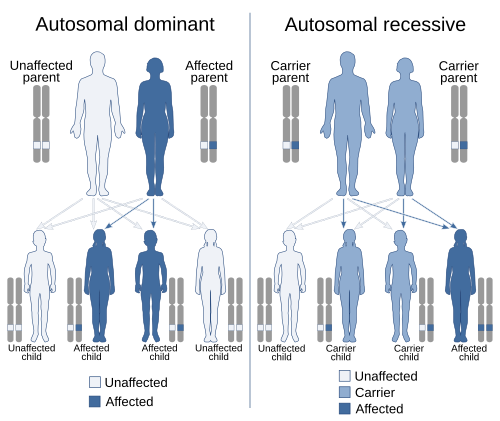
Scope
Medical genetics encompasses many different areas, including clinical practice of physicians, genetic counselors, and nutritionists, clinical
Subspecialties
In some ways, many of the individual fields within medical genetics are hybrids between clinical care and research. This is due in part to recent advances in science and technology (for example, see the
Clinical genetics
Clinical genetics a
- 1. Prenatal genetics
- Couples at risk of having a child with a genetic disorder preconception or while pregnant
- High risk prenatal screeningresults
- Abnormal fetal ultrasound
- 2. Pediatric genetics
- Birth defects
- autism, epilepsy
- skeletal dysplasia
- 3. Adult genetics
- cardiac dysrhythmias
- inherited kidney disease
- neurodegeneration
- connective tissue disease
- 4. Cancer genetics
- breast/ovarian cancer
- bowel cancer
- endocrine tumors
Examples of genetic syndromes that are commonly seen in the genetics clinic include
Training and qualification
In the United States, physicians who practice clinical genetics are accredited by the American Board of Medical Genetics and Genomics (ABMGG).
In Australia and New Zealand, clinical genetics is a three-year advanced training program for those who already have their primary medical qualification (
Metabolic/biochemical genetics
Cytogenetics
Cytogenetics is the study of
Molecular genetics
Molecular genetics involves the discovery of and laboratory testing for
Mitochondrial genetics
There exists some overlap between medical genetic diagnostic laboratories and molecular pathology.
Genetic counseling
Genetic counseling is the process of providing information about genetic conditions, diagnostic testing, and risks in other family members, within the framework of nondirective counseling.
History
Although genetics has its roots back in the 19th century with the work of the Bohemian monk
Medical genetics was a late developer, emerging largely after the close of World War II (1945) when the eugenics movement had fallen into disrepute.[5] The Nazi misuse of eugenics sounded its death knell.[6] Shorn of eugenics, a scientific approach could be used and was applied to human and medical genetics. Medical genetics saw an increasingly rapid rise in the second half of the 20th century and continues in the 21st century.
Current practice
The clinical setting in which patients are evaluated determines the scope of practice, diagnostic, and therapeutic interventions. For the purposes of general discussion, the typical encounters between patients and genetic practitioners may involve:
- Referral to an out-patient genetics clinic (pediatric, adult, or combined) or an in-hospital consultation, most often for diagnostic evaluation.
- Specialty genetics clinics focusing on management of lysosomal storage diseases.
- Referral for counseling in a prenatal genetics clinic to discuss risks to the pregnancy (advanced maternal age, teratogen exposure, family history of a genetic disease), test results (abnormal maternal serum screen, abnormal ultrasound), and/or options for prenatal diagnosis (typically non-invasive prenatal screening, diagnostic amniocentesis or chorionic villus sampling).
- Multidisciplinary specialty clinics that include a clinical geneticist or genetic counselor (cancer genetics, cardiovascular genetics, craniofacial or cleft lip/palate, hearing loss clinics, muscular dystrophy/neurodegenerative disorder clinics).
Diagnostic evaluation
Each patient will undergo a diagnostic evaluation tailored to their own particular presenting signs and symptoms. The geneticist will establish a
Chromosome studies
Chromosome studies are used in the general genetics clinic to determine a cause for developmental delay or intellectual disability, birth defects, dysmorphic features, or autism.[citation needed] Chromosome analysis is also performed in the prenatal setting to determine whether a fetus is affected with aneuploidy or other chromosome rearrangements. Finally, chromosome abnormalities are often detected in cancer samples. A large number of different methods have been developed for chromosome analysis:
- Chromosome analysis using a karyotype involves special stains that generate light and dark bands, allowing identification of each chromosome under a microscope.
- Fluorescence in situ hybridization (FISH) involves fluorescent labeling of probes that bind to specific DNA sequences, used for identifying aneuploidy, genomic deletions or duplications, characterizing chromosomal translocations and determining the origin of ring chromosomes.
- Chromosome painting is a technique that uses fluorescent probes specific for each chromosome to differentially label each chromosome. This technique is more often used in cancer cytogenetics, where complex chromosome rearrangements can occur.
- Array comparative genomic hybridization is a newer molecular technique that involves hybridization of an individual DNA sample to a glass slide or microarray chip containing molecular probes (ranging from large ~200kb bacterial artificial chromosomesto small oligonucleotides) that represent unique regions of the genome. This method is particularly sensitive for detection of genomic gains or losses across the genome but does not detect balanced translocations or distinguish the location of duplicated genetic material (for example, a tandem duplication versus an insertional duplication).
Basic metabolic studies
Biochemical studies are performed to screen for imbalances of metabolites in the bodily fluid, usually the blood (plasma/serum) or urine, but also in cerebrospinal fluid (CSF). Specific tests of enzyme function (either in leukocytes, skin fibroblasts, liver, or muscle) are also employed under certain circumstances. In the US, the newborn screen incorporates biochemical tests to screen for treatable conditions such as galactosemia and phenylketonuria (PKU). Patients suspected to have a metabolic condition might undergo the following tests:
- Quantitative amino acid analysis is typically performed using the ninhydrin reaction, followed by urea cycle disorders, maple syrup urine disease, and PKU. Measurement of amino acids in urine can be useful in the diagnosis of cystinuria or renal Fanconi syndrome as can be seen in cystinosis.
- Urine organic acid analysis can be either performed using quantitative or qualitative methods, but in either case the test is used to detect the excretion of abnormal metabolic conditions.
- The acylcarnitine combination profile detects compounds such as organic acids and fatty acids conjugated to carnitine. The test is used for detection of disorders involving fatty acid metabolism, including MCAD.
- Pyruvate and lactate are byproducts of normal metabolism, particularly during anaerobic metabolism. These compounds normally accumulate during exercise or ischemia, but are also elevated in patients with disorders of pyruvate metabolism or mitochondrial disorders.
- urea cycle disorders, as well as other conditions involving liver failure.
- Enzyme testing is performed for a wide range of metabolic disorders to confirm a diagnosis suspected based on screening tests.
Molecular studies
- intronsare analyzed. Therefore, although these tests are highly specific and sensitive, they do not routinely identify all of the mutations that could cause disease.
- epigenetic mechanisms such as genomic imprinting and uniparental disomy.
- trinucleotide repeats.
Treatments
Each cell of the body contains the hereditary information (
Management of metabolic disorders
In general, metabolic disorders arise from enzyme deficiencies that disrupt normal metabolic pathways. For instance, in the hypothetical example:
A ---> B ---> C ---> D AAAA ---> BBBBBB ---> CCCCCCCCCC ---> (no D) X Y Z X Y | (no or insufficient Z) EEEEE
Compound "A" is metabolized to "B" by enzyme "X", compound "B" is metabolized to "C" by enzyme "Y", and compound "C" is metabolized to "D" by enzyme "Z". If enzyme "Z" is missing, compound "D" will be missing, while compounds "A", "B", and "C" will build up. The pathogenesis of this particular condition could result from lack of compound "D", if it is critical for some cellular function, or from toxicity due to excess "A", "B", and/or "C", or from toxicity due to the excess of "E" which is normally only present in small amounts and only accumulates when "C" is in excess. Treatment of the metabolic disorder could be achieved through dietary supplementation of compound "D" and dietary restriction of compounds "A", "B", and/or "C" or by treatment with a medication that promoted disposal of excess "A", "B", "C" or "E". Another approach that can be taken is enzyme replacement therapy, in which a patient is given an infusion of the missing enzyme "Z" or cofactor therapy to increase the efficacy of any residual "Z" activity.
- Diet
Dietary restriction and supplementation are key measures taken in several well-known metabolic disorders, including
- Medication
Medical approaches include enhancement of residual enzyme activity (in cases where the enzyme is made but is not functioning properly), inhibition of other enzymes in the biochemical pathway to prevent buildup of a toxic compound, or diversion of a toxic compound to another form that can be excreted. Examples include the use of high doses of
Certain
Other examples
- Angiotensin receptor blockers in Marfan syndrome & Loeys-Dietz
- Bone marrow transplantation
- Gene therapy
Career paths and training
The examples and perspective in this article deal primarily with the United States and do not represent a worldwide view of the subject. (August 2015) |

There are a variety of career paths within the field of medical genetics, and naturally the training required for each area differs considerably. The information included in this section applies to the typical pathways in the United States and there may be differences in other countries. US practitioners in clinical, counseling, or diagnostic subspecialties generally obtain board certification through the American Board of Medical Genetics.
| Career | Degree | Description | Training |
| Clinical geneticist | MD-PhD |
A clinical geneticist is typically a physician who evaluates patients in the office or as a hospital consultation. This process includes a medical history, family history (pedigree), a detailed physical examination, reviewing objective data such as imaging and test results, establishing a differential diagnosis, and recommending appropriate diagnostic tests. | College (4 yrs) → Medical school (4 yrs) → Primary residency (1 yr) → Residency in Clinical genetics (2 yrs). Some Clinical geneticists also obtain a PhD degree (4-7 yrs). A new residency track offers a 4 yr primary residency in Clinical genetics immediately after finishing Medical school.[citation needed] |
| Genetic counselor | MS | A genetic counselor specializes in communication of genetic information to patients and families. Genetic counselors often work closely with Clinical geneticists or other physicians (such as Obstetricians or Oncologists) and often convey the results of the recommended tests. | College (4 yrs) → Graduate program in Genetic counseling (2 yrs). |
| Metabolic nurse and/or nutritionist | BA/BS, MS, RN | One of the critical aspects of the management of patients with metabolic disorders is the appropriate nutritional intervention (either restricting the compound that cannot be metabolized, or supplementing compounds that are deficient as the result of an enzyme deficiency). The metabolic nurse and nutritionist play important roles in coordinating the dietary management. | College (4 yrs) → Nursing school or graduate training in nutrition. |
| Biochemical diagnostics | BS, MS, PhD, MBBS, MD, DO, MD-PhD | Individuals who specialize in organic acids, and enzyme activity. Some Clinical Geneticists are also board certified in Biochemical Genetics. |
College (4 yrs) → Graduate school (PhD, usually 4–7 years) and/or Medical school (4 years) |
| Cytogenetic diagnostics | BS, MS, PhD, MBBS, MD, DO, MD-PhD | Individuals who specialize in FISH, and comparative genomic hybridization tests. Some Clinical Geneticists are also board certified in Cytogenetics. |
College (4 yrs) → Graduate school (PhD, usually 4–7 years) and/or Medical school (4 years) |
| Molecular genetics | BS, MS, PhD, MBBS, MD, DO, MD-PhD | Individuals who specialize in Southern blotting . |
College (4 yrs) → Graduate school (PhD, usually 4–7 years) and/or Medical school (4 years) |
| Research geneticist | BS, MS, PhD, MBBS, MD, DO, MD-PhD | Any researcher who studies the genetic basis of human disease or uses model organisms to study disease mechanisms could be considered a Research Geneticist. Many of the clinical career paths also include basic or translational research, and thus individuals in the field of medical genetics often participate in some form of research. | College (4 yrs) → Graduate school (PhD, usually 4–7 years) and/or Medical school (4 years) → Post-doctoral research training (usually 3+ years) |
| Laboratory technician | AS, BS, MS | Technicians in the diagnostic or research labs handle samples and run the assays at the bench. | College (4 yrs), may have higher degree (MS, 2+ years) |
Ethical, legal and social implications
Genetic information provides a unique type of knowledge about an individual and his/her family, fundamentally different from a typically laboratory test that provides a "snapshot" of an individual's health status. The unique status of genetic information and inherited disease has a number of ramifications with regard to ethical, legal, and societal concerns.
On 19 March 2015, scientists urged a worldwide ban on clinical use of methods, particularly the use of
Societies
The more empirical approach to human and medical genetics was formalized by the founding in 1948 of the American Society of Human Genetics. The Society first began annual meetings that year (1948) and its international counterpart, the International Congress of Human Genetics, has met every 5 years since its inception in 1956. The Society publishes the American Journal of Human Genetics on a monthly basis.
Medical genetics is recognized as a distinct medical specialty. In the U.S., medical genetics has its own approved board (the American Board of Medical Genetics) and clinical specialty college (the
In Australia and New Zealand, medical geneticists are trained and certified under the auspices of the Royal Australasian College of Physicians, but professionally belong to the Human Genetics Society of Australasia and its special interest group, the Australasian Association of Clinical Geneticists, for ongoing education, networking and advocacy.
bioethics
Research
This section needs expansion with: more details and additional citations. You can help by adding to it. (August 2008) |
The broad range of research in medical genetics reflects the overall scope of this field, including basic research on genetic inheritance and the human genome, mechanisms of genetic and metabolic disorders, translational research on new treatment modalities, and the impact of genetic testing
Basic genetics research
Basic research geneticists usually undertake research in universities, biotechnology firms and research institutes.
Allelic architecture of disease
Sometimes the link between a disease and an unusual gene variant is more subtle. The genetic architecture of common diseases is an important factor in determining the extent to which patterns of genetic variation influence group differences in health outcomes.[17][18][19] According to the common disease/common variant hypothesis, common variants present in the ancestral population before the dispersal of modern humans from Africa play an important role in human diseases.[20] Genetic variants associated with Alzheimer disease, deep venous thrombosis, Crohn disease, and type 2 diabetes appear to adhere to this model.[21] However, the generality of the model has not yet been established and, in some cases, is in doubt.[18][22][23] Some diseases, such as many common cancers, appear not to be well described by the common disease/common variant model.[24]
Another possibility is that common diseases arise in part through the action of combinations of variants that are individually rare.[25][26] Most of the disease-associated alleles discovered to date have been rare, and rare variants are more likely than common variants to be differentially distributed among groups distinguished by ancestry.[24][27] However, groups could harbor different, though perhaps overlapping, sets of rare variants, which would reduce contrasts between groups in the incidence of the disease.
The number of variants contributing to a disease and the interactions among those variants also could influence the distribution of diseases among groups. The difficulty that has been encountered in finding contributory alleles for complex diseases and in replicating positive associations suggests that many complex diseases involve numerous variants rather than a moderate number of alleles, and the influence of any given variant may depend in critical ways on the genetic and environmental background.[22][28][29][30] If many alleles are required to increase susceptibility to a disease, the odds are low that the necessary combination of alleles would become concentrated in a particular group purely through drift.[31]
Population substructure in genetics research
One area in which population categories can be important considerations in genetics research is in controlling for confounding between
Population substructure also can be used to advantage in genetic association studies.[38] For example, populations that represent recent mixtures of geographically separated ancestral groups can exhibit longer-range linkage disequilibrium between susceptibility alleles and genetic markers than is the case for other populations.[39][40][41][42] Genetic studies can use this admixture linkage disequilibrium to search for disease alleles with fewer markers than would be needed otherwise. Association studies also can take advantage of the contrasting experiences of racial or ethnic groups, including migrant groups, to search for interactions between particular alleles and environmental factors that might influence health.[43][44]
See also
- Full genome sequencing
- Inborn error of metabolism
- Predictive medicine
- DNA Valley
References
- ^ "American Board of Medical Genetics and Genomics". abmgg.org.
- ^ "Training Options - ABMGG". abmgg.org.
- ^ RACP. "Clinical Genetics". Royal Australasian College of Physicians.
- S2CID 25809385.
- OCLC 995257497.
- PMID 21435294.
- New York Times. Retrieved 20 March 2015.
- New York Times. Retrieved 20 March 2015.
- PMID 25791083.
- PMID 25810189.
- New York Times. Retrieved 24 April 2015.
- PMID 25894090.
- ^ Regalado, Antonio (2016-05-08). "Chinese Researchers Experiment with Making HIV-Proof Embryos". MIT Technology Review. Retrieved 2016-06-10.
- ^ Gallagher, James (1 February 2016). "Scientists get 'gene editing' go-ahead". BBC News. BBC. Retrieved 10 June 2016.
- ^ Amjad, Anneesa (2016-06-06). "Dutch government seeks to allow creation of human embryos for research". BioNews. Retrieved 2016-06-10.
- ^ D. Bonneau, S. Marlin, D. Sanlaville, J.-M. Dupont, H. Sobol, M. Gonzales, M. Le Merrer, P. Malzac, F. Razavi, S. Manouvrier, S. Odent, D. Stoppa-Lyonnet, Les tests génétiques à l’heure de la deuxième révision des lois de bioéthique, Pathologie Biologie, Volume 58, Issue 5, 2010, Pages 396-401
- PMID 11525833.
- ^ PMID 12351577.
- PMID 12351581.
- PMID 12142358.
- S2CID 6850292.
- ^ S2CID 685795.
- PMID 12615007.
- ^ PMID 14527296.
- PMID 11404818.
- S2CID 39429794.
- ^ Risch N, Burchard E, Ziv E, Tang H, "Categorization of humans in biomedical research: genes, race and disease", Genome Biol (2002) 3 (http://genomebiology.com/2002/3/7/comment/2007 Archived 2006-06-24 at the Wayback Machine) (electronically published July 1, 2002; accessed August 25, 2005)
- S2CID 4392356.
- PMID 11565063.
- PMID 11882781.
- ^ Cooper RS, "Genetic factors in ethnic disparities in health", in Anderson NB, Bulatao RA, Cohen B, eds., Critical perspectives on racial and ethnic differences in health in later life, (Washington DC: National Academy Press, 2004), 267–309.
- PMID 15052271.
- PMID 12050090.
- PMID 12050091.
- PMID 9736746.
- PMID 12817591.
- PMID 15052270.
- PMID 18852203.
- PMID 15088268.
- PMID 15088269.
- PMID 15088270.
- PMID 15540159.
- PMID 11689494.
- S2CID 205209730.
Further reading
- Wikidata Q29581774.
External links
- Genetics home reference
- The National Human Genome Research Institute hosts an information center
- The Phenomizer – A tool for clinical diagnostics in medical genetics. Phenomizer
