Ximelagatran
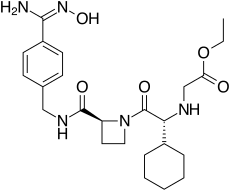 | |
| Clinical data | |
|---|---|
| Trade names | Exanta |
| Pregnancy category |
|
| Routes of administration | Oral (tablets) |
| ATC code | |
| Legal status | |
| Legal status |
|
melagatran | |
| Elimination half-life | 3–5 hours |
| Excretion | Renal (80%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Ximelagatran (Exanta or Exarta, H 376/95) is an anticoagulant that has been investigated extensively as a replacement for warfarin[1] that would overcome the problematic dietary, drug interaction, and monitoring issues associated with warfarin therapy. In 2006, its manufacturer AstraZeneca announced that it would withdraw pending applications for marketing approval after reports of hepatotoxicity (liver damage) during trials, and discontinue its distribution in countries where the drug had been approved (Germany, Portugal, Sweden, Finland, Norway, Iceland, Austria, Denmark, France, Switzerland, Argentina and Brazil).[2]
Method of action
Ximelagatran, a
Uses
Ximelagatran was expected to replace
An advantage, according to early reports by its manufacturer, was that it could be taken orally without any monitoring of its anticoagulant properties. This would have set it apart from
Side effects
Ximelagatran was generally well tolerated in the trial populations, but a small proportion (5–6%) developed elevated
A chemically different but pharmacologically similar substance, AZD-0837, was developed by AstraZeneca for similar indications.[2] It is a prodrug of a potent, competitive, reversible inhibitor of free and fibrin-bound thrombin called ARH0637.[8] The development of AZD-0837 has been discontinued. Due to a limitation identified in long-term stability of the extended-release AZD-0837 drug product, a follow-up study from ASSURE on stroke prevention in patients with non-valvular atrial fibrillation, was prematurely closed in 2010 after 2 years. There was also a numerically higher mortality against warfarin.[9][10][11] In a Phase 2 trial for AF the mean serum creatinine concentration increased by about 10% from baseline in patients treated with AZD-0837, which returned to baseline after cessation of therapy.[12]
References
- PMID 17664384.
- ^ a b "AstraZeneca Decides to Withdraw Exanta" (Press release). AstraZeneca. February 14, 2006. Retrieved 2012-07-16.
- PMID 17319469.
- S2CID 20556829.
- S2CID 26026547.
- PMID 14585939.
- S2CID 36324117.
- PMID 22742650.
- ^ "AZD0837". Astrazenecaclinicaltrials.com. Retrieved 2012-10-16.
- ^ "Long-term treatment with the oral direct thrombin inhibitor AZD0837, compared to Vitamin-K antagonists, as stroke prevention in patients with non-valvular atrial fibrillation and one or more risk factors for stroke and systemic embolic events. A 5-year follow-up study study". Clinical Study Report Synopsis. AstraZeneca. 21 January 2010. Trial D1250C0004221. Archived from the original on 10 November 2013.
- PMID 20368532.
- PMID 19690349.
External links
- "Ximelagatran". Drug Information Portal. U.S. National Library of Medicine.
