Melatonin

| |
| |
| Names | |
|---|---|
| IUPAC name
N-[2-(5-methoxy-1H-indol-3-yl)ethyl]acetamide
| |
| Other names
5-Methoxy-N-acetyltryptamine; N-Acetyl-5-methoxytryptamine; NSC-113928
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.000.725 |
| EC Number |
|
| KEGG | |
| MeSH | Melatonin |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C13H16N2O2 | |
| Molar mass | 232.281 g/mol |
| Melting point | 117 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Melatonin, an
In vertebrates, melatonin's functions extend to
In addition to its
Biological activity
In humans, melatonin primarily acts as a potent
Furthermore, melatonin functions as a high-capacity
Biological functions
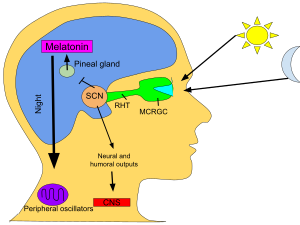
Circadian rhythm
In mammals, melatonin is critical for the regulation of sleep–wake cycles, or circadian rhythms.
The antioxidant properties of melatonin were first recognized in 1993.
Melatonin's concentration in the mitochondrial matrix is significantly higher than that found in the blood plasma,[15][16][17] emphasizing its role not only in direct free radical scavenging but also in modulating the expression of antioxidant enzymes and maintaining mitochondrial integrity. This multifaceted role shows the physiological significance of melatonin as a mitochondrial antioxidant, a notion supported by numerous scholars.[14][15][16][17][18]
Furthermore, the interaction of melatonin with reactive oxygen and nitrogen species results in the formation of metabolites capable of reducing free radicals.
Immune system
Melatonin's interaction with the
Weight regulation
Melatonin's potential to regulate weight gain is posited to involve its inhibitory effect on
Biochemistry
Biosynthesis

The
Serotonin, an essential
In
The hypothesis that melatonin synthesis occurs within mitochondria and
Mechanism

The mechanism of melatonin biosynthesis initiates with the hydroxylation of L-tryptophan, a process that requires the cofactor tetrahydrobiopterin (THB) to react with oxygen and the active site iron of tryptophan hydroxylase. Although the complete mechanism is not entirely understood, two main mechanisms have been proposed:
The first mechanism involves a slow transfer of one electron from THB to molecular oxygen (O2), potentially producing a superoxide (O−2). This superoxide could then recombine with the THB radical to form 4a-peroxypterin. 4a-peroxypterin may either react with the active site iron (II) to create an iron-peroxypterin intermediate or directly transfer an oxygen atom to the iron, facilitating the hydroxylation of L-tryptophan.
Alternatively, the second mechanism proposes that oxygen interacts with the active site iron (II) first, forming iron (III) superoxide. This molecule could then react with THB to form an iron-peroxypterin intermediate.
Following the formation of iron (IV) oxide from the iron-peroxypterin intermediate, this oxide selectively
The decarboxylation of 5-hydroxy-L-tryptophan to produce 5-hydroxytryptamine is then facilitated by a decarboxylase enzyme with
Serotonin N-acetyltransferase, with
The final step in the biosynthesis of melatonin involves the methylation of N-acetylserotonin at the hydroxyl position by SAM, resulting in the production of
Regulation
In vertebrates, the secretion of melatonin is regulated through the activation of the
Blue light, especially within the 460–480
Metabolism
Melatonin is
Measurement
For both research and clinical purposes, melatonin levels in humans can be determined through saliva or blood plasma analysis.[54]
Use as a medication and supplement
Melatonin is used both as a
A study published by the
History
Discovery
Melatonin's discovery is linked to the study of skin color changes in some amphibians and reptiles, a phenomenon initially observed through the administration of pineal gland extracts.
The hormone melatonin was isolated in 1958 by
The first patent for the therapeutic use of melatonin as a low-dose sleep aid was awarded to Richard Wurtman at the Massachusetts Institute of Technology in 1995.[65]
Etymology
The etymology of melatonin stems from its skin-lightening properties. As detailed in their publication in the
Occurrence
Animals
In vertebrates, melatonin is produced in darkness, thus usually at night, by the pineal gland, a small endocrine gland[71] located in the center of the brain but outside the
Many animals use the variation in duration of melatonin production each day as a seasonal clock.
During the night, melatonin regulates leptin, lowering its levels.
Plants
Until its identification in plants in 1987, melatonin was for decades thought to be primarily an animal neurohormone. When melatonin was identified in coffee extracts in the 1970s, it was believed to be a byproduct of the extraction process. Subsequently, however, melatonin has been found in all plants that have been investigated. It is present in all the different parts of plants, including leaves, stems, roots, fruits, and seeds, in varying proportions.[8][80] Melatonin concentrations differ not only among plant species, but also between varieties of the same species depending on the agronomic growing conditions, varying from picograms to several micrograms per gram.[36][81] Notably high melatonin concentrations have been measured in popular beverages such as coffee, tea, wine, and beer, and crops including corn, rice, wheat, barley, and oats.[8] In some common foods and beverages, including coffee[8] and walnuts,[82] the concentration of melatonin has been estimated or measured to be sufficiently high to raise the blood level of melatonin above daytime baseline values.
Although a role for melatonin as a plant hormone has not been clearly established, its involvement in processes such as growth and photosynthesis is well established. Only limited evidence of endogenous circadian rhythms in melatonin levels has been demonstrated in some plant species and no membrane-bound receptors analogous to those known in animals have been described. Rather, melatonin performs important roles in plants as a growth regulator, as well as environmental stress protector. It is synthesized in plants when they are exposed to both biological stresses, for example, fungal infection, and nonbiological stresses such as extremes of temperature, toxins, increased soil salinity, drought, etc.[36][83][84]
Herbicide-induced oxidative stress has been experimentally mitigated in vivo in a high-melatonin transgenic rice.[85][86]
Fungal disease resistance is another role. Added melatonin increases
Fungi
Melatonin has been observed to reduce stress tolerance in Phytophthora infestans in plant-pathogen systems.[88] Danish pharmaceutical company Novo Nordisk have used genetically modified yeast (Saccharomyces cerevisiae) to produce melatonin.[89]
Bacteria
Melatonin is produced by α-proteobacteria and photosynthetic cyanobacteria. There is no report of its occurrence in archaea which indicates that melatonin originated in bacteria[11] most likely to prevent the first cells from the damaging effects of oxygen in the primitive Earth's atmosphere.[10]
Novo Nordisk have used genetically modified Escherichia coli to produce melatonin.[90][91]
Food products
Naturally-occurring melatonin has been reported in foods including tart cherries to about 0.17–13.46 ng/g,[92] bananas, plums, grapes, rice, cereals, herbs,[93] olive oil, wine,[94] and beer.[95] The consumption of milk and sour cherries may improve sleep quality.[96] When birds ingest melatonin-rich plant feed, such as rice, the melatonin binds to melatonin receptors in their brains.[97] When humans consume foods rich in melatonin, such as banana, pineapple, and orange, the blood levels of melatonin increase significantly.[98]
References
- S2CID 52954755.
- ^ PMID 28648359.
- ISBN 978-0-323-32602-5.
- S2CID 18050554.
- PMID 15992934.
- S2CID 46984486.
- S2CID 20404509.
- ^ PMID 22016420.
- PMID 29495303.
- ^ S2CID 24373303.
- ^ PMID 31057485.
- ^ PMID 27314810.
Hence, one melatonin molecule and its associated metabolites could scavenge a large number of reactive species, and thus, the overall antioxidant capacity of melatonin is believed to be greater than that of other well-known antioxidants, such as vitamin C and vitamin E, under in vitro or in vivo conditions (Gitto et al., 2001; Sharma and Haldar, 2006; Ortiz et al., 2013).
- ^ "Melatonin receptors | G protein-coupled receptors | IUPHAR/BPS Guide to Pharmacology". www.guidetopharmacology.org. Retrieved 7 April 2017.
- ^ PMID 28400824.
- ^ S2CID 23820389.
melatonin is specifically targeted to the mitochondria where it seems to function as an apex antioxidant ... The measurement of the subcellular distribution of melatonin has shown that the concentration of this indole in the mitochondria greatly exceeds that in the blood.
- ^ S2CID 35435683.
There is credible evidence to suggest that melatonin should be classified as a mitochondria-targeted antioxidant.
- ^ S2CID 24373303.
While originally thought to be produced exclusively in and secreted from the vertebrate pineal gland [53], it is now known that the indole is present in many, perhaps all, vertebrate organs [54] and in organs of all plants that have been investigated [48, 55, 56]. That melatonin is not relegated solely to the pineal gland is also emphasized by the reports that it is present in invertebrates [57–59], which lack a pineal gland and some of which consist of only a single cell.
- ^ S2CID 10920415.
- PMID 27551178.
- S2CID 41937922.
- S2CID 33664568.
- PMID 19546564.
- ^ Tan DX, Chen LD, Poeggeler B, L Manchester C, Reiter RJ (1993). "Melatonin: a potent, endogenous hydroxyl radical scavenger". Endocr. J. 1: 57–60.
- S2CID 36383425.
- ^ PMID 19521488.
- PMID 7934611.
- S2CID 21133107.
- PMID 12596522.
- PMID 16729718.
- S2CID 6822594.
- S2CID 237603177.
- PMID 20083828.
- ^ "MetaCyc serotonin and melatonin biosynthesis".
- ^ PMID 28503116.
- PMID 22826715.
- ^ PMID 25240067.
- S2CID 206140413.
- PMID 23441081.
- PMID 1540578.
- ^ ISBN 978-0-471-49640-3.
- PMID 10024876.
- PMID 8397829.
- S2CID 33548757.
- S2CID 20517556.
- PMID 11487664.
- PMID 20061218.
- ^ "Recent News – Program of Computer Graphics". www.graphics.cornell.edu.
- PMID 15713707.
- ^ "University of Houston study shows blue light glasses at night increase melatonin by 58%". designeroptics.com. 25 August 2021. Retrieved 26 August 2021.
- S2CID 145296760.
- ^ "Melatonin". www.drugbank.ca. Retrieved 29 January 2019.
- S2CID 38857779.
- S2CID 14555783. Retrieved 25 January 2023.
- PMID 30919486.
- S2CID 52096729.
- PMID 27500861.
- S2CID 3578916.
- ^ Cohen PA, Avula B, Wang Y, Katragunta K, Khan I. (April 2023) "Quantity of Melatonin and CBD in Melatonin Gummies Sold in the US" JAMA. 329 (16): 1401–1402. doi:10.1001/jama.2023.2296. PMID 37097362
- S2CID 41959214.
- PMID 15357831.
- ISBN 978-0-8247-5504-1.
- .
- PMID 14415935.
- PMID 1167425.
- ^ US patent 5449683, Wurtman RJ, "Methods of inducing sleep using melatonin", issued 12 September 1995, assigned to Massachusetts Institute of Technology
- ^ ISSN 0002-7863.
- .
- PMID 9825724.
- PMID 9066504.
- PMID 18100415.
- S2CID 3219721.
- PMID 16336035.
- S2CID 42725506.
- PMID 32382406.
- PMID 14529560.
- ^ PMID 15649736.
Exogenous melatonin has acute sleepiness-inducing and temperature-lowering effects during 'biological daytime', and when suitably timed (it is most effective around dusk and dawn), it will shift the phase of the human circadian clock (sleep, endogenous melatonin, core body temperature, cortisol) to earlier (advance phase shift) or later (delay phase shift) times.
- .
- PMID 7254478.
- ^ PMID 31579821.
- S2CID 15738948.
- PMID 20417677.
- PMID 15979282.
- PMID 25911967.
- PMID 26094813.
- S2CID 6291664.
- ^ S2CID 38637203.
- ^ S2CID 19887642.
- S2CID 220657247.
- PMID 26710256.
- S2CID 219331624.
- PMID 36676997.
- PMID 11600041.
- ^ González-Flores D, Velardo B, Garrido M, González-Gómez D, Lozano M, Ayuso MC, Barriga C, Paredes SD, Rodríguez AB (2011). "Ingestion of Japanese plums (Prunus salicina Lindl. cv. Crimson Globe) increases the urinary 6-sulfatoxymelatonin and total antioxidant capacity levels in young, middle-aged and elderly humans: Nutritional and functional characterization of their content". Journal of Food and Nutrition Research. 50 (4): 229–36.
- S2CID 8034935.
- ^ Salehi B (5 July 2019). "Melatonin in Medicinal and Food Plants" (PDF). Cells. 681. Archived from the original (PDF) on 29 November 2021. Retrieved 2 July 2021.
- PMID 31856339.
- PMID 7773197.
- S2CID 979886.
External links
- "Melatonin". Chemwatch.
