Melioidosis
| Melioidosis | |
|---|---|
| Diagnostic method | Growing the bacteria in culture mediums[1] |
| Differential diagnosis | Tuberculosis[2] |
| Prevention | Prevention from exposure to contaminated water, antibiotic prophylaxis[1] |
| Treatment | Ceftazidime, meropenem, trimethoprim/sulfamethoxazole[1] |
| Frequency | 165,000 people per year[1] |
| Deaths | 89,000 people per year[1] |
Melioidosis is an
Humans are infected with B. pseudomallei by contact with contaminated soil or water. The bacteria enter the body through wounds, inhalation, or ingestion. Person-to-person or animal-to-human transmission is extremely rare.
Efforts to prevent melioidosis include: wearing protective gear while handling contaminated water or soil, practising hand hygiene, drinking boiled water, and avoiding direct contact with soil, water, or heavy rain.
Approximately 165,000 people are infected by melioidosis per year, resulting in about 89,000 deaths, based on a mathematical model published in 2016.[5] Diabetes is a major risk factor for melioidosis; over half of melioidosis cases are in people with diabetes.[1] Increased rainfall and severe weather events such as thunderstorms are associated with an increased number of melioidosis cases in endemic areas.[2]
Signs and symptoms
Acute




Most people exposed to B. pseudomallei experience no symptoms.[2] The mean incubation period of acute melioidosis is 9 days (range 1–21 days).[1] Nevertheless, symptoms of melioidosis can appear in 24 hours for those who experienced near drowning in water.[7] Those affected present with symptoms of sepsis (predominantly fever) with or without pneumonia, or localised abscess or other focus of infection. The presence of non-specific signs and symptoms has caused melioidosis to be nicknamed "the great mimicker".[1]
People with only pneumonia may have a prominent cough with sputum and shortness of breath. However, those with septic shock together with pneumonia may have minimal coughing.[2] Results of a chest X-ray can range from diffuse nodular infiltrates in those with septic shock to progressive consolidation located most commonly in the upper lobes for those with pneumonia only. Pleural effusion and empyema are more common for melioidosis affecting lower lobes of the lungs.[2] In 10% of cases, people develop secondary pneumonia caused by other bacteria after the primary infection.[3] In northern Australia, 60% of the infected children presented with only skin lesions, while 20% presented with pneumonia.[3]Depending on the course of infection, other severe manifestations develop. Approximately 1 to 5% of those infected develop
Chronic
Chronic melioidosis is usually defined by symptoms lasting greater than two months and occurs in about 10% of patients.[1] Clinical presentations include fever, weight loss, productive cough with or without bloody sputum which may mimic tuberculosis. Additionally, long-standing abscesses at multiple body sites may also present.[2] Tuberculosis should be considered for lymph nodes enlargement at the root of the lung. Additionally, pneumonia caused by melioidosis rarely causes scarring and calcification of the lungs, unlike tuberculosis.[10]
Latent
The potential for prolonged incubation was recognized in US servicemen involved in the Vietnam War, and was referred to as the "Vietnam time-bomb".[2] Initially, it was thought that the longest period between presumed exposure and clinical presentation is 62 years in a prisoner of war in Burma-Thailand-Malaysia.[11] However, subsequent genotyping of the bacteria isolate from the Vietnam veteran showed that the isolate may not come from Southeast Asia, but from South America.[12] This reinstates another report that put the longest latency period for melioidosis as 29 years.[13] Patients with latent melioidosis may be symptom-free for decades.[11] Less than 5% of all melioidosis cases have activation after a period of latency.[1] Various comorbidities such as diabetes, renal failure, and alcoholism can predispose to reactivation of melioidosis.[2]
Cause
Bacteria

Melioidosis is caused by
The genome of B. pseudomallei consists of two replicons: chromosome 1 encodes housekeeping functions of the bacteria such as cell wall synthesis, mobility, and metabolism; chromosome 2 encodes functions that allow the bacteria to adapt to various environments. Horizontal gene transfer has resulted in highly variable genomes in B. pseudomallei. Australia has been suggested as the origin for B. pseudomallei because of the high genetic variability of the bacteria found in this region. Bacteria that was introduced to Central and South America in the 17th to 19th centuries seem to have a common ancestor from Africa.[16] B. mallei is a clone of B. pseudomallei that has lost substantial portions of its genome as it adapted to live exclusively in mammals.[3] This makes the B. mallei genome much smaller than B. pseudomallei.[17]
Transmission
B. pseudomallei is normally found in soil and surface water, and is most abundant at soil depths of 10 to 90 cm.[1] It has been found in soils, ponds, streams, pools, stagnant water, and rice paddy fields.[2] B. pseudomallei can survive in nutrient-poor conditions such as distilled water, desert soil, and nutrient-depleted soil for more than 16 years.[1] It can also survive in antiseptic and detergent solutions, acidic environments (pH 4.5 for 70 days), and in environments at temperatures ranging from 24 °C (75.2 °F) to 32 °C (89.6 °F). However, the bacteria may be killed by the presence of ultraviolet light.[1]
Bacteria can enter the body through wounds, inhalation, and ingestion of contaminated soil or water.[1] Person-to-person transmission is extremely rare.[2] Melioidosis is a recognised disease in animals including pigs, cats, dogs, goats, sheep, horses and others. Cattle, water buffalo, and crocodiles are considered to be relatively resistant to melioidosis despite their constant exposure to mud. Birds are also considered resistant to melioidosis although several cases had been reported in Australia and aquatic birds.[10][15] Transmission from animals to humans is rare.[1][2]
Inadequate chlorination of water supply has been associated with B. pseudomallei outbreak in Northern and Western Australia.[18][19] There were also several cases of bacteria being found in unchlorinated water supply in rural Thailand.[20] Based on the whole genome sequencing of the bacteria, the variety of the bacteria B. pseudomallei in Papua New Guinea is narrow due to limited movements of the indigenous people. This findings supports the hypothesis that humans play an important role in bacterial dispersal.[21]
Pathogenesis
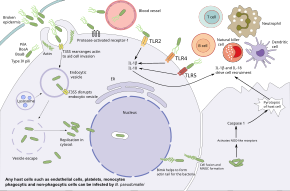
B. pseudomallei has the ability to infect various types of cells and to evade human immune responses. Bacteria first enter at a break in the skin or
Inside the host cell, the bacteria move by inducing the polymerization of the host
While B. pseudomallei can survive in phagocytic cells, these cells can kill B. pseudomallei by several mechanisms. Macrophages activated by
Additional elements of the immune system are activated by the host toll-like receptors such as TLR2, TLR4, and TLR5 that recognize the conserved pieces of the bacteria such as LPS and flagella. This activation results in the production of cytokines such as Interleukin 1 beta (IL-1β) and Interleukin 18 (IL-18). IL-18 increases IFN production through natural killer cells while IL-1beta reduces the IFN production. These immune molecules drive the recruitment of other immune cells such as neutrophils, dendritic cells, B cells, and T cells to the site of infection. T cells seem to be particularly important for controlling B. pseudomallei; T cell numbers are increased in survivors, and low T cell numbers are associated with a high risk of death from melioidosis. Despite this, HIV infection is not a risk factor for melioidosis. Although macrophages show deregulated cytokine responses in individuals with HIV infection, bacterial internalization and intracellular killing are still effective. People infected with B. pseudomallei may develop antibodies against the bacteria, and people that live in endemic areas tend to have antibodies in their blood that recognize B. pseudomallei. However, the effectiveness of these antibodies at preventing melioidosis is unclear.[1][10]
B. pseudomallei can remain latent in the human body for up to 29 years until it is reactivated during human immunosuppression or stress response. However, the site of bacteria during latent infection and the mechanism by which they avoid immune recognition for years are both unclear. Amongst mechanisms suggested are: residing in the nucleus of the cell to prevent being digested, entering a stage of slower growth, antibiotic resistance, and genetic adaption to the host environment. Granulomas (containing neutrophils, macrophages, lymphocytes, and multinucleated giant cells) formed at the infection site in melioidosis have been associated with latent infection in humans.[1]
Diagnosis



Culture
Bacterial culture has 60% sensitivity in diagnosing melioidosis.[23] B. pseudomallei is never part of human flora. Therefore, any growth of the bacteria is diagnostic of melioidosis. Other samples such as throat, rectal swabs, pus from abscesses, and sputum can also be used for culture.[1] However, culture from CSF is difficult because in one case series, only 29% of the neuromelioidosis cases are culture positive.[10] When bacteria do not grow from people strongly suspected of having melioidosis, repeated cultures should be taken as subsequent cultures can become positive.[1] B. pseudomallei can be grown on any blood agar, MacConkey agar, and agar containing antibiotics such as Ashdown's medium (containing gentamicin),[10] and Ashdown's broth (containing colistin)[3] for better isolation of B. pseudomallei from other types of bacteria.[10] Agar plates for melioidosis should be incubated at 37 °C (98.6 °F) in air [2] and inspected daily for four days. On the agar plates, B. pseudomallei forms creamy, non-haemolytic, colonies after 2 days of incubation. After 4 days of incubation, colonies appear dry and wrinkled.[1] Colonies of B. pseudomallei that are grown on Francis medium (a modification of Ashdown medium with gentamicin concentration increased to 8 mg/L and neutral red indicator replaced with 0.2% bromocresol purple) are yellow.[24] For laboratories located outside endemic areas, Burkholderia cepacia selective agar can be used if Ashdown's medium is not available.[2] It is important not misinterpret the bacterial growth as Pseudomonas or Bacillus spp. Other biochemical screening tools can also be used for detecting B. pseudomallei, including the API 20NE or 20E biochemical kit combined with Gram stain, oxidase test, typical growth characteristics, and resistance to certain antibiotics of the bacteria.[3] API 20NE biochemical kit is 99% sensitive in identifying B. pseudomallei.[10]
Molecular methods such as 16S rDNA sequencing, multiplex polymerase chain reaction (PCR), and real-time PCR can also be used to identify B. pseudomallei in culture.[1][25][26][27] Other bacterial genes such as fliC genes encoding flagellin, rpsU gene encoding for ribosomal protein, and TTS genes encoding Type III secretion systems has also been employed for detection. Another method of gene detection namely multiple cross displacement amplification for the bacterial TTS1 gene detection produces results within an hour.[27]
Hematological and biochemical tests
General blood tests in people with melioidosis show low white blood cell counts (indicates infection), raised liver enzymes, increased bilirubin levels (indicates liver dysfunction), and raised urea and creatinine levels (indicates kidney dysfunction). Low blood glucose and acidosis predicts a poorer prognosis in those with melioidosis. However, other tests such as C-reactive protein and procalcitonin levels are not reliable in predicting the severity of melioidosis infection.[15]
Serological tests
Antigen detect tests allow rapid detection of melioidosis. Examples of antigen detection tests are: latex agglutination test and
Microscopy
By microscopy, B. pseudomallei is seen as
Imaging
Various imaging modalities can also help with the diagnosis of melioidosis. In acute melioidosis with the spreading of the bacteria through the bloodstream, the chest X-ray shows multifocal nodular lesions. It may also show merging nodules or
Prevention
Melioidosis is a notifiable disease in Australia
Large-scale water chlorination has been successful at reducing B. pseudomallei in the water in Australia.[35][1] In middle to low-income countries, water should be boiled before consumption.[1] In high income countries, water could be treated with ultraviolet light for those at risk of contracting melioidosis.[36][1] Those who are at high risk of contact with the bacteria should wear protective gear (such as boots and gloves) during work.[1] Those staying in endemic areas should avoid direct contact with soil, and outdoor exposure to heavy rain or dust clouds. Bottled water or boiled water are preferred as drinking water.[37][1] A study conducted from 2014 to 2018, however showed no significant differences on whether behavioural changes can reduce the risk of contracting melioidosis. Modification of behavioural changes or more frequent interventions may be needed to ensure a definite reduction in risk of getting melioidosis.[38]
Antibiotic prophylaxis
Administering cotrimoxazole three times a week throughout a wet season for dialysis patients has no obvious benefit of preventing melioidosis. Besides, high cost and side effects of this drug limits its use to only those with high risk of getting melioidosis.
Vaccination
Several vaccine candidates have been tested in animal models. Nevertheless, no vaccine candidates have been tried in humans. Major hurdles of the vaccines are limited efficacy in animal models, establishing the best method of vaccine administration in humans and logistical and financial issues in establishing human trials in endemic areas.[10]
Treatment
The treatment of melioidosis is divided into two stages: an intravenous intensive phase and an eradication phase to prevent recurrence. The choice of antibiotics depends upon the susceptibility of the bacteria to various antibiotics. B. pesudomallei are generally susceptible to ceftazidime, meropenem, imipenem, and co-amoxiclav. These drugs generally kill bacteria. B. pseudomallei is also susceptible to doyxcycline, chloramphenicol, and co-trimoxazole. These drugs generally inhibit the growth of the bacteria. However, the bacteria are resistant to penicillin, ampicillin, 1st and 2nd generation cephalosporin, gentamicin, streptomycin, tobramycin, macrolides, and polymyxins.[1] On the other hand, 86% of the B. pseudomallei isolates from the region of Sarawak, Malaysia are susceptible to gentamicin and this has not been found elsewhere in other parts of the world.[41]
Prior to 1989, the standard treatment for acute melioidosis was a three-drug combination of
Intensive phase
Meropenem is the preferred antibiotic therapy for neurological melioidosis and those with septic shock admitted into intensive care units. Co-trimoxazole is recommended in addition to ceftazidime for neurological melioidosis, osteomyelitis, septic arthritis, skin and gastrointestinal infection, and deeply seated abscess. For deep-seated infections such as abscesses of internal organs, osteomyelitis, septic arthritis, and neurological melioidosis, the duration of antibiotics given should be longer (up to 4 to 8 weeks). The time taken for the fever to be resolved can be more than 10 days in those with deep-seated infection. According to the 2020 Revised Royal Darwin Hospital Guideline, the dosage for intravenous ceftazidime is 2g 6-hourly in adults (50 mg/kg up to 2g in children less than 15 years old). The dosage for meropenem is 1g 8-hourly in adults (25 mg/kg up to 1g in children).[46] Acquired resistance to ceftazidime, carbapenems, and co-amoxiclav is rare in the intensive phase but resistance to cotrimoxazole during eradication therapy is technically difficult to assess.[47] There are no differences between using cefoperazone/sulbactam or ceftazidime to treat melioidosis as both shows similar death rates and disease progression following treatment. However, data are lacking to recommend cefoperazone/sulbactam usage.[47][48] For those with kidney impairment, the dosage of ceftazidime, meropenem, and co-trimoxazole should be lowered.[3] Once the clinical condition improved, meropenem can be switched back to ceftazidime.[1]
Eradication phase
Following the treatment of the acute disease, eradication treatment with
In Australia, co-trimoxazole is used with children and pregnant mothers after the first 12 weeks of pregnancy. Meanwhile, in Thailand, co-amoxiclav is the drug of choice for children and pregnant women.
Surgery
Surgical drainage is indicated for single, large abscesses in the liver, muscle, and prostate. However, for multiple abscesses in the liver, spleen, and kidney, surgical drainage may not be possible or necessary. For septic arthritis, arthrotomy washout and drainage are required. Surgical debridement may be necessary.[1] For those with mycotic aneurysm, urgent surgery is required for prosthetic vascular grafts. Lifelong therapy with co-trimoxazole may be needed for those with prosthetic vascular grafts according to a review of case reports in 2005.[53] Other abscesses rarely need to be drained because most resolve with antibiotic treatment.[1] Prostate abscess may require routine imaging. Antibiotics treatment for prostatic abscess may be enough except for abscesses more than 10 to 15 mm where surgical drainage is required.[54][55][56]
Others
Several immunomodulating therapies are suggested to boost the human body immune function against the bacteria because the pathogenesis of melioidosis is thought to be contributed by defects in
Anti-PDI (programmed cell death) agents could be useful in melioidosis treatment especially for those with septic shock. This is because Burkholderia pseudomallei bacteria increases the expression of PDI-1 that regulates and inhibits the formation of T-cells that are essential for fighting against melioidosis.[57]
Prognosis
In well-resourced settings, where the disease can be detected and treated early, the risk of death is 10%. In resource-poor settings, the risk of death from the disease is more than 40%.[1]
Recurrent melioidosis can occur either due to re-infection or relapse after the completion of eradication therapy. Re-infection is due to a new strain of B. pseudomallei bacteria. Meanwhile, relapse is due to failure to clear infections after the eradication therapy. Recurrent melioidosis is rare since 2014 due to improved antibiotic therapy and prolongation of the intensive phase of therapy as evident in Darwin Prospective Melioidosis Study.[58] On the other hand, recrudescence are those who present with symptoms during the eradication therapy. Recrudescence rates may be improved by ensuring adherence to a full course of eradication therapy e.g. by reducing self-discharge against medical advice.[59]
Underlying medical conditions such as diabetes mellitus, chronic kidney disease, and cancer can worsen the long-term survival and disability of those who recover from infection. One of the complications of melioidosis is encephalomyelitis. It can cause quadriparesis (muscle weakness in all the limbs), partial flaccid paraparesis (muscle weakness of both legs), or foot drop. For those with previous melioidosis-associated bone and joint infections, complications such as sinus tract infection, bone and joint deformities with limited range of motion can occur.[1]
Epidemiology

Melioidosis is an understudied disease that remains endemic in developing countries. In 2015, the International Melioidosis Society was formed to raise awareness of the disease.[1] In 2016, a statistical model was developed which predicted that the number is 165,000 cases per year with 138,000 of those occurring in East and South Asia and the Pacific.[60] In approximately half of those cases (54% or 89,000), people will die.[1] Under-reporting is a common problem as only 1,300 cases were reported worldwide since 2010, which is less than 1% of the projected incidence based on the modelling.[1] Lack of laboratory diagnostic capabilities and lack of disease awareness amongst health care providers also causes under diagnosis. Even if bacterial cultures show positive result for B. pesudomallei, they can be discarded as contaminants especially in laboratories in non-endemic areas.[1] In 2015, it was estimated that the yearly disability-adjusted life year (DALY) was 84.3 per 100,000 people. As of 2022, melioidosis is not included in the WHO list of neglected tropical diseases.[61][62]
Geography
Melioidosis is endemic in parts of southeast Asia (including Thailand,[63] Laos,[64] Singapore,[65] Brunei,[66] Malaysia,[67] Myanmar[68] and Vietnam[69]), southern China,[70] Taiwan[71] northern Australia.[72] India,[73] and South America.[74] Since 1991, a total of 583 cases were reported in India. Most Indian cases are located in Karnataka and Tamil Nadu.[73] Fifty-one cases of melioidosis were reported in Bangladesh from 1961–2017. Nonetheless, lack of awareness and resources gives rise to under diagnosis of the disease in the country.[75] The true burden of melioidosis in Africa and Middle East remain unknown due to low amount of data. Several melioidosis cases were reported over the years. Although 24 African countries and three Middle Eastern countries predicted to be endemic with melioidosis, however not a single case was reported from these specific countries.[76] In the United States, two historical cases (1950 and 1971) and four recent cases (2010, 2011, 2013, 2020) have been reported amongst people that did not travel overseas.[3][77] Despite extensive investigations, the source of melioidosis was never confirmed. One possible explanation is that importation of medicinal plant products or exotic reptiles could have resulted in the introduction of melioidosis in the United States.[3] In 2021, there was a melioidosis outbreak in several states in the United States due to usage of contaminated aromatherapy spray imported from India.[78] There are also cases of infection through imported tropical fishes in home aquariums.[79] In Europe, more than half of the melioidosis cases are imported from Thailand.[80]
Age, risk factors
Melioidosis is found in all age groups.
History
Pathologist
The first human case of melioidosis in South Asia was reported in Sri Lanka in 1927.
In 1989, several studies conducted in Thailand demonstrated ceftazidime as an effective antibiotic against melioidosis.
Synonyms
- Pseudoglanders[100]
- Whitmore's disease (after Captain Alfred Whitmore, who first described the disease)[83]
- Nightcliff gardener's disease (Nightcliff is a suburb of Darwin, Australia where melioidosis is endemic)[101]
- Paddy-field disease[102]
- Morphia injector's septicaemia[103]
Biological warfare
Interest in melioidosis has been expressed because it has the potential to be developed as a
References
![]() This article was adapted from the following source under a [ ] license (2022) (reviewer reports):
Siang Ching Raymond Chieng (14 August 2022). "Melioidosis" (PDF). WikiJournal of Medicine. 9 (1): 4.
This article was adapted from the following source under a [ ] license (2022) (reviewer reports):
Siang Ching Raymond Chieng (14 August 2022). "Melioidosis" (PDF). WikiJournal of Medicine. 9 (1): 4. {{cite journal}}: CS1 maint: unflagged free DOI (link
- ^ PMID 29388572.
- ^ PMID 25359677.
- ^ PMID 25643275.
- PMID 29340327.
- ^ PMID 26877885.
- ^ S2CID 251702278.
- ^ ISBN 978-1-4557-4801-3.
- S2CID 52056443.
- S2CID 236431874.
- ^ PMID 32161067.
- ^ PMID 15695721.
- PMID 28628442.
- PMID 9153149.
- ^ S2CID 167205734.
- ^ PMID 15831829.
- ^ PMID 28112723.
- PMID 15377793.
- S2CID 30669924.
- PMID 10694154.
- PMID 24447771.
- PMID 21483841.
- PMID 35137005.
- PMID 20830194.
- PMID 16626918.
- PMID 22705921.
- PMID 14532197.
- ^ PMID 35272632.
- PMID 23532805.
- PMID 17804660.
- ^ PMID 25908634.
- ^ PMID 32083145.
- S2CID 225119300.
- ISBN 978-0-16-085042-4.
- PMID 28232503.
- PMID 14511713.
- PMID 23751401.
- PMID 23437412.
- PMID 34170931.
- PMID 30014827.
- PMID 23171644.
- PMID 24145517.
- S2CID 28919574.
- PMID 2681117.
- PMID 18256414.
- PMID 7893868.
- ^ PMID 32986699.
- ^ PMID 24613038.
- PMID 11823963.
- PMID 32725199.
- PMID 25245211.
- PMID 25758020.
- S2CID 232066583.
- S2CID 10117525.
- PMID 29141724.
- PMID 19421682.
- PMID 19524969.
- PMID 25801435.
- PMID 24478504.
- PMID 25811783.
- PMID 26877885.
- PMID 31285144.
- S2CID 245484437.
- ^ PMID 29725623.
- PMID 30274419.
- PMID 30274428.
- PMID 30274418.
- ^ PMID 30274422.
- PMID 30274425.
- PMID 30274435.
- PMID 30823573.
- PMID 30274423.
- PMID 30274424.
- ^ PMID 30274447.
- PMID 30274456.
- PMID 30274436.
- PMID 30274458.
- PMID 32442394.
- S2CID 247221649.
- PMID 34570693.
- S2CID 96435767.
- PMID 10476750.
- ^ "Melioidosis Locally Endemic in Areas of the Mississippi Gulf Coast after Burkholderia pseudomallei Isolated in Soil and Water and Linked to Two Cases – Mississippi, 2020 and 2022" (PDF). CDC. Retrieved 2022-08-05.
- ^ PMID 29005374.
- ^ S2CID 222196789.
- PMID 11823558.
- ^ Stanton AT; Fletcher W (1921). "Melioidosis, a new disease of the tropics". Far Eastern Association of Tropical Medicine: Transactions of the Fourth Congress. Batavia, Dutch East Indies: Javasche Boekhandel en Drukkerij.
- PMID 12734250.
- ^ PMID 1283774.
- PMID 13638596.
- ^ ISBN 978-0-444-53479-8.
- PMID 25272365.
- PMID 23556010.
- PMID 10674639.
- PMID 13983001.
- .
- S2CID 28919574.
- PMID 11897082.
- PMID 25631791.
- PMID 24284287.
- PMID 30082045.
- ^ Barker A (19 June 2005). "Rise in melioidosis rates in NT". Australian Broadcasting Corporation. Retrieved 2007-06-24.
- PMID 15534940.
- .
- ^ PMID 28846298. NBK448110. Retrieved 15 February 2019.
- ISBN 978-0-16-093126-0.
- PMID 17710778.
External links
- Burkholderia pseudomallei genomes and related information at PATRIC, a Bioinformatics Resource Center funded by NIAID
