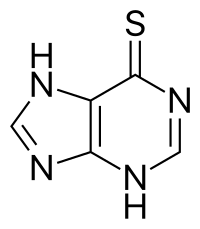
Mercaptopurine
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Purinethol, Purixan, others |
| Other names | 6-mercaptopurine (6-MP) |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682653 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 5 to 37% |
| Metabolism | xanthine oxidase |
| Elimination half-life | 60 to 120 min., longer for its active metabolites |
| Excretion | kidney |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Mercaptopurine (6-MP), sold under the brand name Purinethol among others, is a medication used for
Common side effects include
Mercaptopurine was approved for medical use in the United States in 1953.[1] It is on the World Health Organization's List of Essential Medicines.[4]
Medical uses
It is used to treat
Side effects
Some of the adverse reactions of taking mercaptopurine may include diarrhea, nausea, vomiting, loss of appetite, fatigue, stomach/abdominal pain, weakness, skin rash, darkening of the skin, and hair loss. Serious adverse reactions include mouth sores, fever, sore throat, easy bruising or bleeding, pinpoint red spots on the skin, yellowing of eyes or skin, dark urine, and painful or difficult urination. Other more serious side effects include black or tarry stools (melena), bloody stools, and bloody urine. Treatment is discontinued in up to 30% of patients due these effects but therapeutic drug monitoring of the biologically active metabolites, i.e. thiopurine nucleotides can help to optimize the efficacy and safety. Clinically, most hospitals resort to on-exchange LC-MS (liquid chromatography - mass spectrometry) but the newly developed approach of porous graphitic carbon based chromatography hyphenated with mass spectrometry appears superior with respect to patient care in this respect.[6]
Symptoms of allergic reaction to mercaptopurine include
.In some cases, mercaptopurine may
Toxicity of mercaptopurine can be linked to genetic polymorphisms in thiopurine S-methyltransferase (TPMT), nudix hydrolase 15 (NUDT15),[7][8] and inosine triphosphate pyrophosphatase (ITPA). People with specific allele variants will require dose adjustments, especially for those with homozygous variant genotypes. Large differences of TPMT and NUDT15 among ethnicities in terms of variant allele frequency should be taken into consideration in clinical practice.[9] Caucasian people with a variant allele of the ITPA gene, experience higher rates of febrile neuropenia than people of other ethnic groups, due to differences in allelic frequencies among ethnicities.[10]
Precautions
Mercaptopurine can lower the body's ability to fight off infection. Those taking it should get permission from a doctor to receive immunizations and vaccinations. It is also recommended that, while on the drug, one should avoid those having recently received oral polio vaccine.
This drug was formerly not recommended during pregnancy and early evidence indicated pregnant women on the drug (or the related
Mercaptopurine causes changes to
Drug interactions
Allopurinol inhibits xanthine oxidase, the enzyme that breaks down mercaptopurine. Those taking allopurinol (often used to prevent gout) are at risk for mercaptopurine toxicity. The dose should be reduced or allopurinol should be discontinued. Several published studies have demonstrated that the use of allopurinol in combination with low dose 6-MP helps reduce 6-MP levels, which are toxic to liver tissue, whilst increasing the therapeutic levels of 6-MP for some inflammatory conditions.
Mechanisms of action
This section may be too technical for most readers to understand. (December 2014) |
Official information from the package insert for purinethol:[17]
- Mercaptopurine is an antimetabolite antineoplastic, as such it interferes with normal metabolic processes within cells, typically by combining with enzymes, to disrupt DNA and RNA synthesis (cell-cycle S phase-specific) leading to death of rapidly proliferating cells, especially malignant ones. Specifically, Mercaptopurine is a purine antimetabolite or purine antagonist as such inhibits DNA synthesis by inhibiting the production of the purine containing nucleotides, adenine and guanine thus halting DNA synthesis.[18] Mercaptopurine also acts as an immunomodulator by inhibiting of several pathways in nucleic acid biosynthesis preventing proliferation of cells involved in the determination and amplification of the immune response.[19]
- Mercaptopurine (6-MP) competes with the purine derivatives hypoxanthine and guanine for the enzyme HGPRT and is itself converted to thio inosine monophosphate (TIMP).
- TIMP inhibits several chemical reactions involving adenylic acid(AMP) via adenylosuccinate (SAMP).
- In addition, 6-methylthioinosinate (MTIMP) is formed by the methylation of TIMP.
- Both TIMP and MTIMP have been reported to inhibit glutamine-5-phosphoribosylpyrophosphate amidotransferase, the first enzyme unique to the de novo pathway for purine ribonucleotide synthesis. Experiments indicate that radiolabeled mercaptopurine may be recovered from the DNA in the form of deoxythioguanosine.
- TIMP inhibits several chemical reactions involving
- Some mercaptopurine is converted to nucleotide derivatives of 6-thioguanine (6-TG) by the sequential actions of inosinate (IMP) dehydrogenase and xanthylate (XMP) aminase, converting TIMP to thioguanylic acid (TGMP).
- Animal tumors that are resistant to mercaptopurine often have lost the ability to convert mercaptopurine to TIMP. However, it is clear that resistance to mercaptopurine may be acquired by other means as well, particularly in human leukemias.
- It is not known exactly which of any one or more of the biochemical effects of mercaptopurine and its metabolites are directly or predominantly responsible for cell death.
6-MP ribonucleotide inhibits purine nucleotide synthesis and metabolism by inhibiting an enzyme called
Pharmacogenetics
The enzyme
History
6-MP was discovered by Nobel Prize–winning scientists
See also
References
- ^ a b c d e f g h i "Mercaptopurine". The American Society of Health-System Pharmacists. Archived from the original on 20 December 2016. Retrieved 8 December 2016.
- ^ ISBN 9780857111562.
- S2CID 27475772.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ "Mercaptopurine". The American Society of Health-System Pharmacists. Archived from the original on 2015-09-06. Retrieved Aug 28, 2015.
- S2CID 225287631.
- PMID 25624441.
- PMID 26878724.
- PMID 28088792.
- PMID 6404543.
- S2CID 25314258.
- from the original on 2006-06-19.
- S2CID 25614617.
- PMID 21122490.
- S2CID 21675626.
- S2CID 45441636.
- ^ "PURINETHOL (mercaptopurine) tablet [Gate Pharmaceuticals]" (PDF). DailyMed. Gate Pharmaceuticals. August 2012. Archived from the original on 1 January 2014. Retrieved 31 December 2013.
- ^ "Chemotherapy".
- ^ Nielsen OH, Vainer B, Rask-Madsen J. Review article: the treatment of inflammatory bowel disease with 6-mercaptopurine or azathioprine. Aliment Pharmacol Ther. 2001 Nov;15(11):1699-708. doi: 10.1046/j.1365-2036.2001.01102.x. PMID 11683683.
- ^ Hansen, Barbara. "Purine and Pyrimidine Metabolism." USMLE STEP 1 Biochemistry and Medical Genetics Lecture Notes. 2010 ed. N.p.: Kaplan, 2010. 288-90. Print.
- PMID 19952870.
- PMID 24707136.
- PMID 17691917. Archived from the originalon 2013-01-12.
- ^ PMID 21270794.
- ISBN 978-3-8047-1763-3.
- PMID 17255139.
- ^ "Label: Mercaptopurine – mercaptopurine tablet". Archived from the original on 6 October 2015. Retrieved 11 March 2015.
- ^ "Purixan suspension". DailyMed. 9 April 2019. Retrieved 9 April 2020.
- PMID 20154640.
- ^ Bouton K (29 January 1989). "The Nobel Pair". The New York Times Magazine. Archived from the original on 2016-10-07.
- ^ ISBN 978-1439170915.
Further reading
- Dean L (2012). "Mercaptopurine Therapy and TPMT Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. PMID 28520348. Bookshelf ID: NBK100660.
External links
- "Mercaptopurine". Drug Information Portal. U.S. National Library of Medicine.
