Mercuric amidochloride
| |
| Names | |
|---|---|
| IUPAC name
Mercuric azanide chloride
| |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.030.292 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Hg(NH2)Cl | |
| Molar mass | 252.065 g/mol |
| Appearance | White crystalline solid or white amorphous powder[1] |
| Odor | Odorless[1] |
| Density | 5.56 g/cm3 |
| Boiling point | Sublimes[1] |
| 0.14 g in 100 mL of cold water 100 g in 100 mL of hot water (decomposes)[1] | |
| Solubility | Soluble in warm hydrochloric acid, nitric acid and acetic acid, insoluble in ethanol[1] |
| Pharmacology | |
| D08AK01 (WHO) | |
| Hazards | |
| GHS labelling:[1] | |
  
| |
| Danger | |
| H300, H310, H330, H373, H410 | |
| P260, P262, P264, P270, P271, P273, P280, P284, P301+P316, P302+P352, P304+P340, P316, P319, P320, P321, P330, P361+P364, P391, P403+P233, P405, P501 | |
| Flash point | Non-combustible |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
|
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Mercuric amidochloride is an inorganic compound with the formula Hg(NH2)Cl.
Preparation and properties
It arises from the reaction of mercury(II) chloride and ammonia (Calomel reaction), where the resulting mercuric amidochloride is highly insoluble.
- HgCl2 + 2 NH3 → HgCl(NH2) + [NH4]Cl
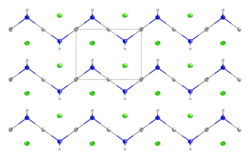
It forms white crystals in the shape of small prisms, which have earthy metallic taste.[1]It consists of a zig-zag 1-dimensional polymer (HgNH2)n with chloride counterions.[2][3]
It is stable in
This substance is a deadly poison. If improperly handled, may cause dangerous environmental pollution, in
Addition of base converts it into "Millon's base" (named after Eugène Millon), which has the formula Hg2(OH)N·xH2O. A variety of related amido and nitrido materials with chloride, bromide, and hydroxide are known.[4]
Uses
Before the toxicity of mercury was revealed, mercuric amidochloride, then known as "ammoniated mercury" or "

See also
- Merbromin, also known as "Mercurochrome", another antiseptic mercury compound
- Thiomersal, another antiseptic mercury compound
References
- ^ a b c d e f g h i j k "Ammoniated mercury".
- ISBN 0-19-855370-6.[page needed]
- .
- ISBN 0-12-352651-5.[page needed]
- S2CID 20467204.
- ^ "Mercury ammonium chloride".
