Methicillin-resistant Staphylococcus aureus
Methicillin-resistant Staphylococcus aureus (MRSA) is a group of gram-positive bacteria that are genetically distinct from other strains of Staphylococcus aureus. MRSA is responsible for several difficult-to-treat infections in humans. It caused more than 100,000 deaths worldwide attributable to antimicrobial resistance in 2019.
MRSA is any strain of S. aureus that has developed (through natural selection) or acquired (through horizontal gene transfer) a multiple drug resistance to beta-lactam antibiotics. Beta-lactam (β-lactam) antibiotics are a broad-spectrum group that include some penams (penicillin derivatives such as methicillin and oxacillin) and cephems such as the cephalosporins.[1] Strains unable to resist these antibiotics are classified as methicillin-susceptible S. aureus, or MSSA.
MRSA infection is common in hospitals, prisons, and nursing homes, where people with open wounds, invasive devices such as catheters, and weakened immune systems are at greater risk of healthcare-associated infection. MRSA began as a hospital-acquired infection but has become community-acquired, as well as livestock-acquired. The terms HA-MRSA (healthcare-associated or hospital-acquired MRSA), CA-MRSA (community-associated MRSA), and LA-MRSA (livestock-associated MRSA) reflect this.[citation needed]
Signs and symptoms

In humans, Staphylococcus aureus is part of the normal microbiota present in the upper respiratory tract,[2] and on skin and in the gut mucosa.[3] However, along with similar bacterial species that can colonize and act symbiotically, they can cause disease if they begin to take over the tissues they have colonized or invade other tissues; the resultant infection has been called a "pathobiont".[2]
After 72 hours, MRSA can take hold in human tissues and eventually become resistant to treatment. The initial presentation of MRSA is small red bumps that resemble pimples, spider bites, or boils; they may be accompanied by fever and, occasionally, rashes. Within a few days, the bumps become larger and more painful; they eventually open into deep, pus-filled boils. About 75 percent of CA-MRSA infections are localized to skin and soft tissue and usually can be treated effectively.[4]
Risk factors
A select few of the populations at risk include:
- People with indwelling implants, prostheses, drains, and catheters[1][5]
- People who are frequently in crowded places, especially with shared equipment and skin-to-skin contact[6]
- People with weak immune systems (; etc.)
- Diabetics[1][7]
- Regular contact with someone who has injected drugs in the past year[11]
- Users of quinolone antibiotics[5][12]
- Elderly people[5][13]
- School children sharing sports and other equipment
- College students living in dormitories[6]
- People staying or working in a health-care facility for an extended period of time[5][6]
- People who spend time in coastal waters where MRSA is present, such as some beaches in Florida and the West Coast of the United States[14][15]
- People who spend time in confined spaces with other people, including occupants of homeless shelters,
- Veterinarians, livestock handlers, and pet owners[18]
- People who ingest unpasteurized milk[19]
- People who are immunocompromised and also colonized[20]: 249
- People with chronic obstructive pulmonary disease[5]
- People who have had thoracic surgery[5]
As many as 22% of people infected with MRSA do not have any discernable risk factors.[21]: 637
Hospitalized people
People who are hospitalized, including the elderly, are often
Both surgical and nonsurgical wounds can be infected with HA-MRSA.[1][5][22] Surgical site infections occur on the skin surface, but can spread to internal organs and blood to cause sepsis.[1] Transmission can occur between healthcare providers and patients because some providers may neglect to perform preventative hand-washing between examinations.[12][25]
People in nursing homes are at risk for all the reasons above, further complicated by their generally weaker immune systems.[13][26]
Prison inmates and military personnel
Prisons and military barracks[19] can be crowded and confined, and poor hygiene practices may proliferate, thus putting inhabitants at increased risk of contracting MRSA.[18] Cases of MRSA in such populations were first reported in the United States and later in Canada. The earliest reports were made by the Centers for Disease Control and Prevention in US state prisons. In the news media, hundreds of reports of MRSA outbreaks in prisons appeared between 2000 and 2008. For example, in February 2008, the Tulsa County jail in Oklahoma started treating an average of 12 S. aureus cases per month.[27]
Animals
Antibiotic use in livestock increases the risk that MRSA will develop among the livestock and other animals that may reside near them; strains MRSA ST 398 and CC398 are transmissible to humans.[19][28] Generally, animals are asymptomatic.[1]
Domestic pets are susceptible to MRSA infection by transmission from their owners; conversely, MRSA-infected pets can also transmit MRSA to humans.[29]
Athletes
Locker rooms, gyms, and related athletic facilities offer potential sites for MRSA contamination and infection.[30] Athletes have been identified as a high-risk group.[19] A study linked MRSA to the abrasions caused by artificial turf.[31] Three studies by the Texas State Department of Health found the infection rate among football players was 16 times the national average. In October 2006, a high-school football player was temporarily paralyzed from MRSA-infected turf burns. His infection returned in January 2007 and required three surgeries to remove infected tissue, and three weeks of hospital stay.[32]
In 2013, Lawrence Tynes, Carl Nicks, and Johnthan Banks of the Tampa Bay Buccaneers were diagnosed with MRSA. Tynes and Nicks apparently did not contract the infection from each other, but whether Banks contracted it from either individual is unknown.[33] In 2015, Los Angeles Dodgers infielder Justin Turner was infected while the team visited the New York Mets.[34] In October 2015, New York Giants tight end Daniel Fells was hospitalized with a serious MRSA infection.[35]
Children
MRSA is becoming a critical problem in children;[36] studies found 4.6% of patients in U.S. health-care facilities, (presumably) including hospital nurseries,[37] were infected or colonized with MRSA.[38] Children and adults who come in contact with day-care centers,[19] playgrounds, locker rooms, camps, dormitories, classrooms and other school settings, and gyms and workout facilities are at higher risk of contracting MRSA. Parents should be especially cautious of children who participate in activities where sports equipment is shared, such as football helmets and uniforms.[39]
Intravenous drug users
Needle-required drugs have caused an increase of MRSA,[40] with injection drug use (IDU) making up 24.1% (1,839 individuals) of Tennessee Hospital's Discharge System. The unsanitary methods of injection causes an access point for the MRSA to enter the blood stream and begin infecting the host. Furthermore, with MRSA's high contagion rate,[11] a common risk factor is individuals who are in constant contact with someone who has injected drugs in the past year.
Mechanism
SCCmec
Staphylococcal
Different SCCmec genotypes confer different microbiological characteristics, such as different antimicrobial resistance rates.[49] Different genotypes are also associated with different types of infections. Types I–III SCCmec are large elements that typically contain additional resistance genes and are characteristically isolated from HA-MRSA strains.[44][49] Conversely, CA-MRSA is associated with types IV and V, which are smaller and lack resistance genes other than mecA.[44][49]
These distinctions were thoroughly investigated by Collins et al. in 2001, and can be explained by the fitness differences associated with carriage of a large or small SCCmec plasmid. Carriage of large plasmids, such as SCCmecI–III, is costly to the bacteria, resulting in a compensatory decrease in virulence expression.[50] MRSA is able to thrive in hospital settings with increased antibiotic resistance but decreased virulence – HA-MRSA targets immunocompromised, hospitalized hosts, thus a decrease in virulence is not maladaptive.[50] In contrast, CA-MRSA tends to carry lower-fitness cost SCCmec elements to offset the increased virulence and toxicity expression required to infect healthy hosts.[50]
mecA
mecA is under the control of two
Arginine catabolic mobile element
The arginine catabolic mobile element (ACME) is a virulence factor present in many MRSA strains but not prevalent in MSSA.[53] SpeG-positive ACME compensates for the polyamine hypersensitivity of S. aureus and facilitates stable skin colonization, wound infection, and person-to-person transmission.[54]
Strains
Acquisition of SCCmec in methicillin-sensitive S. aureus (MSSA) gives rise to a number of genetically different MRSA lineages. These genetic variations within different MRSA strains possibly explain the variability in virulence and associated MRSA infections.[55] The first MRSA strain, ST250 MRSA-1, originated from SCCmec and ST250-MSSA integration.[55] Historically, major MRSA clones ST2470-MRSA-I, ST239-MRSA-III, ST5-MRSA-II, and ST5-MRSA-IV were responsible for causing hospital-acquired MRSA (HA-MRSA) infections.[55] ST239-MRSA-III, known as the Brazilian clone, was highly transmissible compared to others and distributed in Argentina, Czech Republic, and Portugal.[55]
In the UK, the most common strains of MRSA are EMRSA15 and EMRSA16.
Community-acquired MRSA (CA-MRSA) strains emerged in late 1990 to 2000, infecting healthy people who had not been in contact with healthcare facilities.[59] Researchers suggest that CA-MRSA did not evolve from HA-MRSA.[59] This is further proven by molecular typing of CA-MRSA strains[60] and genome comparison between CA-MRSA and HA-MRSA, which indicate that novel MRSA strains integrated SCCmec into MSSA separately on its own.[59] By mid-2000, CA-MRSA was introduced into healthcare systems and distinguishing CA-MRSA from HA-MRSA became a difficult process.[59] Community-acquired MRSA is more easily treated and more virulent than hospital-acquired MRSA (HA-MRSA).[59] The genetic mechanism for the enhanced virulence in CA-MRSA remains an active area of research. The Panton–Valentine leukocidin (PVL) genes are of particular interest because they are a unique feature of CA-MRSA.[55]
In the United States, most cases of CA-MRSA are caused by a CC8 strain designated ST8:USA300, which carries SCCmec type IV, Panton–Valentine leukocidin, PSM-alpha and enterotoxins Q and K,[57] and ST1:USA400.[61] The ST8:USA300 strain results in skin infections, necrotizing fasciitis, and toxic shock syndrome, whereas the ST1:USA400 strain results in necrotizing pneumonia and pulmonary sepsis.[55] Other community-acquired strains of MRSA are ST8:USA500 and ST59:USA1000. In many nations of the world, MRSA strains with different genetic background types have come to predominate among CA-MRSA strains; USA300 easily tops the list in the U.S. and is becoming more common in Canada after its first appearance there in 2004. For example, in Australia, ST93 strains are common, while in continental Europe ST80 strains, which carry SCCmec type IV, predominate.[62][63] In Taiwan, ST59 strains, some of which are resistant to many non-beta-lactam antibiotics, have arisen as common causes of skin and soft tissue infections in the community. In a remote region of Alaska, unlike most of the continental U.S., USA300 was found only rarely in a study of MRSA strains from outbreaks in 1996 and 2000 as well as in surveillance from 2004 to 2006.[64]
A MRSA strain, CC398, is found in intensively reared production animals (primarily pigs, but also cattle and poultry), where it can be transmitted to humans as LA-MRSA (livestock-associated MRSA).[58][65][66]
Diagnosis



Diagnostic microbiology laboratories and reference laboratories are key for identifying outbreaks of MRSA. Normally, a bacterium must be cultured from blood, urine,
Another common laboratory test is a rapid latex
Microbiology
Like all S. aureus (also abbreviated SA at times), methicillin-resistant S. aureus is a gram-positive, spherical (
Other strains of S. aureus have emerged that are resistant to oxacillin, clindamycin, teicoplanin, and erythromycin. These resistant strains may or may not possess the mecA gene. S. aureus has also developed resistance to vancomycin (VRSA). One strain is only partially susceptible to vancomycin and is called vancomycin-intermediate S. aureus (VISA). GISA, a strain of resistant S. aureus, is glycopeptide-intermediate S. aureus and is less suspectible to vancomycin and teicoplanin. Resistance to antibiotics in S. aureus can be quantified by determining the amount of the antibiotic that must be used to inhibit growth. If S. aureus is inhibited at a concentration of vancomycin less than or equal to 4 μg/ml, it is said to be susceptible. If a concentration greater than 32 μg/ml is necessary to inhibit growth, it is said to be resistant.[21]: 637
Prevention
Screening
In health-care settings, isolating those with MRSA from those without the infection is one method to prevent transmission. Rapid culture and sensitivity testing and molecular testing identifies carriers and reduces infection rates.[71] It is especially important to test patients in these settings since 2% of people are carriers of MRSA, even though in many of these cases the bacteria reside in the nostril and the patient will not present any symptoms.[72]
MRSA can be identified by swabbing the nostrils and isolating the bacteria found there. Combined with extra sanitary measures for those in contact with infected people, swab screening people admitted to hospitals has been found to be effective in minimizing the spread of MRSA in hospitals in the United States, Denmark, Finland, and the Netherlands.[73]
Handwashing
The
Isolation
Excluding
To prevent the spread of MRSA in the home, health departments recommend laundering materials that have come into contact with infected persons separately and with a dilute bleach solution; to reduce the bacterial load in one's nose and skin; and to clean and disinfect those things in the house that people regularly touch, such as sinks, tubs, kitchen counters, cell phones, light switches, doorknobs, phones, toilets, and computer keyboards.[79]
Restricting antibiotic use
Glycopeptides, cephalosporins, and in particular, quinolones are associated with an increased risk of colonisation of MRSA. Reducing use of antibiotic classes that promote MRSA colonisation, especially fluoroquinolones, is recommended in current guidelines.[12][25]
Public health considerations
Mathematical models describe one way in which a loss of infection control can occur after measures for screening and isolation seem to be effective for years, as happened in the UK. In the "search and destroy" strategy that was employed by all UK hospitals until the mid-1990s, all hospitalized people with MRSA were immediately isolated, and all staff were screened for MRSA and were prevented from working until they had completed a course of eradication therapy that was proven to work. Loss of control occurs because colonised people are discharged back into the community and then readmitted; when the number of colonised people in the community reaches a certain threshold, the "search and destroy" strategy is overwhelmed.[80] One of the few countries not to have been overwhelmed by MRSA is the Netherlands: an important part of the success of the Dutch strategy may have been to attempt eradication of carriage upon discharge from hospital.[81]
Decolonization
As of 2013, no randomized clinical trials had been conducted to understand how to treat nonsurgical wounds that had been colonized, but not infected, with MRSA,[22] and insufficient studies had been conducted to understand how to treat surgical wounds that had been colonized with MRSA.[1] As of 2013, whether strategies to eradicate MRSA colonization of people in nursing homes reduced infection rates was not known.[26]
Care should be taken when trying to drain boils, as disruption of surrounding tissue can lead to larger infections, including infection of the blood stream.[82] Mupirocin 2% ointment can be effective at reducing the size of lesions. A secondary covering of clothing is preferred.[79] As shown in an animal study with diabetic mice, the topical application of a mixture of sugar (70%) and 3% povidone-iodine paste is an effective agent for the treatment of diabetic ulcers with MRSA infection.[83]
Community settings
The examples and perspective in this section deal primarily with the United Kingdom and do not represent a worldwide view of the subject. (August 2020) |
Maintaining the necessary cleanliness may be difficult for people if they do not have access to facilities such as public toilets with handwashing facilities. In the United Kingdom, the
Agriculture
The World Health Organization advocates regulations on the use of antibiotics in animal feed to prevent the emergence of drug-resistant strains of MRSA.[28] MRSA is established in animals and birds.[19]
Treatment
Antibiotics
Treatment of MRSA infection is urgent and delays can be fatal.[20]: 328 The location and history related to the infection determines the treatment. The route of administration of an antibiotic varies. Antibiotics effective against MRSA can be given by IV, oral, or a combination of both, and depend on the specific circumstances and patient characteristics.[4] The use of concurrent treatment with vancomycin or other beta-lactam agents may have a synergistic effect.[21]: 637
Both CA-MRSA and HA-MRSA are resistant to traditional anti-staphylococcal
Vancomycin and
Several newly discovered strains of MRSA show antibiotic resistance even to vancomycin and teicoplanin. Strains with intermediate (4–8 μg/ml) levels of resistance, termed glycopeptide-intermediate S. aureus (GISA) or
Skin and soft-tissue infections
In skin abscesses, the primary treatment recommended is removal of dead tissue, incision, and drainage. More information is needed to determine the effectiveness of specific antibiotics therapy in surgical site infections (SSIs).[4] Examples of soft-tissue infections from MRSA include ulcers, impetigo, abscesses, and SSIs.[89] In surgical wounds, evidence is weak (high risk of bias) that linezolid may be better than vancomycin to eradicate MRSA SSIs.[1]
MRSA colonization is also found in nonsurgical wounds such as traumatic wounds,
Children
In skin infections and secondary infection sites, topical mupirocin is used successfully. For bacteremia and endocarditis, vancomycin or daptomycin is considered. For children with MRSA-infected bone or joints, treatment is individualized and long-term. Neonates can develop neonatal pustulosis as a result of topical infection with MRSA.[4] Clindamycin is not approved for the treatment of MRSA infection, but it is still used in children for soft-tissue infections.[4]
Endocarditis and bacteremia
Evaluation for the replacement of a prosthetic valve is considered. Appropriate antibiotic therapy may be administered for up to six weeks. Four to six weeks of antibiotic treatment is often recommended, and is dependent upon the extent of MRSA infection.[4]
Respiratory infections
CA-MRSA in hospitalized patients pneumonia treatment begins before culture results. After the susceptibility to antibiotics is performed, the infection may be treated with vancomycin or linezolid for up to 21 days. If the pneumonia is complicated by the accumulation of pus in the pleural cavity surrounding the lungs, drainage may be done along with antibiotic therapy.[4] People with cystic fibrosis may develop respiratory complications related to MRSA infection. The incidence of MRSA in those with cystic fibrosis increased during 2000 to 2015 by five times. Most of these infections were HA-MRSA. MRSA accounts for 26% of lung infections in those with cystic fibrosis.[103]
There is insufficient evidence to support the use of topical or systematic antibiotics for nasal or extra-nasal MRSA infection.[104]
Bone and joint infections
Cleaning the wound of dead tissue and draining abscesses is the first action to treat the MRSA infection. Administration of antibiotics is not standardized and is adapted by a case-by-case basis. Antibiotic therapy can last up to 3 months and sometimes even longer.[4]
Infected implants
MRSA infection can occur associated with implants and joint replacements. Recommendations on treatment are based upon the length of time the implant has been in place. In cases of a recent placement of a surgical implant or artificial joint, the device may be retained while antibiotic therapy continues. If the placement of the device has occurred over 3 weeks ago, the device may be removed. Antibiotic therapy is used in each instance sometimes long-term.[4]
Central nervous system
MRSA can infect the central nervous system and form brain abscess, subdural empyema, and spinal epidural abscess. Excision and drainage can be done along with antibiotic treatment. Septic thrombosis of cavernous or
Other infections
Treatment is not standardized for other instances of MRSA infection in a wide range of tissues. Treatment varies for MRSA infections related to: subperiosteal abscesses, necrotizing pneumonia, cellulitis, pyomyositis, necrotizing fasciitis, mediastinitis, myocardial, perinephric, hepatic, and splenic abscesses, septic thrombophlebitis, and severe ocular infections, including endophthalmitis.[4] Pets can be reservoirs and pass on MRSA to people. In some cases, the infection can be symptomatic and the pet can develop a MRSA infection. Health departments recommend that the pet be taken to the veterinarian if MRSA infections keep occurring in the people who have contact with the pet.[79]
Epidemiology
Worldwide, an estimated 2 billion people carry some form of S. aureus; of these, up to 53 million (2.7% of carriers) are thought to carry MRSA.[105] S. aureus was identified as one of the six leading pathogens for deaths associated with resistance in 2019 and 100,000 deaths caused by MRSA were attributable to antimicrobial resistance.[106]
HA-MRSA
In a US cohort study of 1,300 healthy children, 2.4% carried MRSA in their nose.[107] Bacterial sepsis occurs with most (75%) of cases of invasive MRSA infection.[4] In 2009, there were an estimated 463,017 hospitalizations due to MRSA, or a rate of 11.74 per 1,000 hospitalizations.[108] Many of these infections are less serious, but the Centers for Disease Control and Prevention (CDC) estimate that there are 80,461 invasive MRSA infections and 11,285 deaths due to MRSA annually.[109] In 2003, the cost for a hospitalization due to MRSA infection was US$92,363; a hospital stay for MSSA was $52,791.[89]
Infection after surgery is relatively uncommon, but occurs as much as 33% in specific types of surgeries. Infections of surgical sites range from 1% to 33%. MRSA sepsis that occurs within 30 days following a surgical infection has a 15–38% mortality rate; MRSA sepsis that occurs within one year has a mortality rate of around 55%. There may be increased mortality associated with cardiac surgery. There is a rate of 12.9% in those infected with MRSA while only 3% infected with other organisms. SSIs infected with MRSA had longer hospital stays than those who did not.[1]
Globally, MRSA infection rates are dynamic and vary year to year.[110] According to the 2006 SENTRY Antimicrobial Surveillance Program report, the incidence of MRSA bloodstream infections was 35.9% in North America. MRSA blood infections in Latin America was 29%. European incidence was 22.8%. The rate of all MRSA infections in Europe ranged from 50% in Portugal down to 0.8% in Sweden. Overall MRSA infection rates varied in Latin America: Colombia and Venezuela combined had 3%, Mexico had 50%, Chile 38%, Brazil 29%, and Argentina 28%.[89]
The Centers for Disease Control and Prevention (CDC) estimated that about 1.7 million nosocomial infections occurred in the United States in 2002, with 99,000 associated deaths.[111] The estimated incidence is 4.5 nosocomial infections per 100 admissions, with direct costs (at 2004 prices) ranging from $10,500 (£5300, €8000 at 2006 rates) per case (for bloodstream, urinary tract, or respiratory infections in immunocompetent people) to $111,000 (£57,000, €85,000) per case for antibiotic-resistant infections in the bloodstream in people with transplants. With these numbers, conservative estimates of the total direct costs of nosocomial infections are above $17 billion. The reduction of such infections forms an important component of efforts to improve healthcare safety. (BMJ 2007)[citation needed] MRSA alone was associated with 8% of nosocomial infections reported to the CDC National Healthcare Safety Network from January 2006 to October 2007.[112]
The
In the United States, an estimated 95 million people carry S. aureus in their noses; of these, 2.5 million (2.6% of carriers) carry MRSA.[114] A population review conducted in three U.S. communities showed the annual incidence of CA-MRSA during 2001–2002 to be 18–25.7/100,000; most CA-MRSA isolates were associated with clinically relevant infections, and 23% of people required hospitalization.[115]
CA-MRSA
In a US cohort study of 1,300 healthy children, 2.4% carried MRSA in their noses.[107] There are concerns that the presence of MRSA in the environment may allow resistance to be transferred to other bacteria through phages (viruses that infect bacteria). The source of MRSA could come from hospital waste, farm sewage, or other waste water.[4]
LA-MRSA
In 2004, MRSA was first isolated on a Dutch pig farm leading to further investigations of livestock associated MRSA (LA-MRSA).[116] Livestock associated MRSA (LA-MRSA) has been observed in Korea, Brazil, Switzerland, Malaysia, India, Great Britain, Denmark, and China.[19]
History
In 1961, the first known MRSA isolates were reported in a British study, and from 1961 to 1967, infrequent hospital outbreaks occurred in Western Europe and Australia,[17] with methicillin then being licensed in England to treat resistant infections. Other reports of MRSA began to be described in the 1970s.[1] Resistance to other antibiotics was documented in some strains of S. aureus. In 1996, vancomycin resistance was reported in Japan.[21]: 637 In many countries, outbreaks of MRSA infection were reported to be transmitted between hospitals.[70]: 402 The rate had increased to 22% by 1995, and by 1997 the level of hospital S. aureus infections attributable to MRSA had reached 50%.
The first report of community-associated MRSA (CA-MRSA) occurred in 1981, and in 1982, a large outbreak of CA-MRSA occurred among intravenous drug users in Detroit, Michigan.[17] Additional outbreaks of CA-MRSA were reported through the 1980s and 1990s, including outbreaks among Australian Aboriginal populations that had never been exposed to hospitals. In the mid-1990s, scattered reports of CA-MRSA outbreaks among US children were made. While HA-MRSA rates stabilized between 1998 and 2008, CA-MRSA rates continued to rise. A report released by the University of Chicago Children's Hospital comparing two periods (1993–1995 and 1995–1997) found a 25-fold increase in the rate of hospitalizations due to MRSA among children in the United States.[117] In 1999, the University of Chicago reported the first deaths from invasive MRSA among otherwise healthy children in the United States.[17] By 2004, the genome for various strains of MRSA were described.[118]
The observed increased mortality among MRSA-infected people arguably may be the result of the increased underlying
A population-based study of the incidence of MRSA infections in San Francisco during 2004–05 demonstrated that nearly one in 300 residents had such an infection in the course of a year and that greater than 85% of these infections occurred outside of the healthcare setting.[120] A 2004 study showed that people in the United States with S. aureus infection had, on average, three times the length of hospital stay (14.3 vs. 4.5 days), incurred three times the total cost ($48,824 vs. $14,141), and experienced five times the risk of in-hospital death (11.2% vs 2.3%) than people without this infection.[121] In a meta-analysis of 31 studies, Cosgrove et al.,[122] concluded that MRSA bacteremia is associated with increased mortality as compared with MSSA bacteremia (odds ratio= 1.93; 95% CI = 1.93 ± 0.39).[123] In addition, Wyllie et al. report a death rate of 34% within 30 days among people infected with MRSA, a rate similar to the death rate of 27% seen among MSSA-infected people.[124]
In the US, the CDC issued guidelines on October 19, 2006, citing the need for additional research, but declined to recommend such screening.[125] According to the CDC, the most recent estimates of the incidence of healthcare-associated infections that are attributable to MRSA in the United States indicate a decline in such infection rates. Incidence of MRSA central line-associated blood-stream infections as reported by hundreds of intensive care units decreased 50–70% from 2001 to 2007.[126] A separate system tracking all hospital MRSA bloodstream infections found an overall 34% decrease between 2005 and 2008.[126] In 2010, vancomycin was the drug of choice.[4]
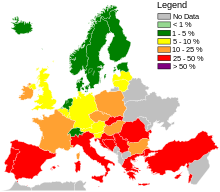
Across Europe, based mostly on data from 2013, seven countries (Iceland, Norway, Sweden, the Netherlands, Denmark, Finland, and Estonia, from lowest to highest) had low levels of hospital-acquired MRSA infections compared to the others,[127]: 92–93 and among countries with higher levels, significant improvements had been made only in Bulgaria, Poland, and the British Isles.[127]: 40
A 1,000-year-old eye salve recipe found in the medieval Bald's Leechbook at the British Library, one of the earliest known medical textbooks, was found to have activity against MRSA in vitro and in skin wounds in mice.[128][129][130]
In the media
MRSA is frequently a media topic, especially if well-known personalities have announced that they have or have had the infection.[131][132][133] Word of outbreaks of infection appears regularly in newspapers and television news programs. A report on skin and soft-tissue infections in the Cook County jail in Chicago in 2004–05 demonstrated MRSA was the most common cause of these infections among those incarcerated there.[134] Lawsuits filed against those who are accused of infecting others with MRSA are also popular stories in the media.[135][136]
MRSA is the topic of radio programs,[137] television shows,[138][139][140] books,[141] and movies.[142]
Research
Various antibacterial chemical extracts from various species of the sweetgum tree (genus Liquidambar) have been investigated for their activity in inhibiting MRSA. Specifically, these are: cinnamic acid, cinnamyl cinnamate, ethyl cinnamate, benzyl cinnamate, styrene, vanillin, cinnamyl alcohol, 2-phenylpropyl alcohol, and 3-phenylpropyl cinnamate.[143]
The delivery of inhaled antibiotics along with systematic administration to treat MRSA are being developed. This may improve the outcomes of those with cystic fibrosis and other respiratory infections.[103] Phage therapy has been used for years in MRSA in eastern countries, and studies are ongoing in western countries.[144][145] Host-directed therapeutics, including host kinase inhibitors, as well as antimicrobial peptides are under study as adjunctive or alternative treatment for MRSA.[146][147][148]
A 2015 Cochrane systematic review aimed to assess the effectiveness of wearing gloves, gowns and masks to help stop the spread of MRSA in hospitals, however no eligible studies were identified for inclusion. The review authors concluded that there is a need for randomized controlled trials to be conducted to help determine if the use of gloves, gowns, and masks reduces the transmission of MRSA in hospitals.[149]
See also
References
- ^ PMID 23963687.
- ^ PMID 27730630.
- PMID 28260936.
- ^ PMID 21208910.
- ^ PMID 27307786.
- ^ a b c "General Information About MRSA in the Community". Centers for Disease Control and Prevention. 10 September 2013. Retrieved 9 October 2014.
- S2CID 5660826.
- PMID 21741111.
- PMID 16445719.
- PMID 22225643.
- ^ PMID 28701438.
- ^ PMID 17986491.
- ^ PMID 28382547.
- ^ "Study: Beachgoers More Likely to Catch MRSA". FoxNews.com. Reuters. 2009-02-16.
- ^ Marchione M (2009-09-12). "Dangerous staph germs found at West Coast beaches". Associated Press.
- PMID 15200838.
- ^ a b c d "MRSA History Timeline: The First Half-Century, 1959–2009". The University of Chicago Medical Center. 2010. Archived from the original on 2020-02-18. Retrieved 2012-04-24.
- ^ PMID 20610826.
- ^ PMID 28435193.
- ^ ISBN 978-0-19-994894-9.
- ^ ISBN 978-0-7817-3014-3.
- ^ PMID 24242704.
- PMID 25002642.
- ^ Davis C. "Is MRSA Contagious?". Medicinenet.com. Retrieved 24 October 2017.
- ^ S2CID 19544705.
- ^ PMID 24254890.
- ^ "PURE Bioscience". purebio.com. Archived from the original on 2009-02-24.
- ^ PMID 25366200.
- PMID 17354344..
 This article incorporates public domain material from websites or documents of the Centers for Disease Control and Prevention
This article incorporates public domain material from websites or documents of the Centers for Disease Control and Prevention - PMID 12522744.
- PMID 15689585.
- ^ Epstein V (21 December 2007). "Texas Football Succumbs to Virulent Staph Infection From Turf". Bloomberg. Retrieved 10 June 2010.
- ^ Yasinskas P (11 October 2013). "Third Tampa Bay Buccaneers player tests positive for MRSA staph infection". ESPN. ESPN Internet Ventures. Retrieved 11 October 2013.
- ^ Hernandez D (August 12, 2015). "Dodgers' Justin Turner nears return from MRSA infection". Los Angeles Times. Retrieved August 13, 2015.
- NFL.com. Retrieved October 12, 2015.
- PMID 15033832.
- PMID 15963273.
- ^ "National Prevalence Study of Methicillin-Resistant Staphylococcus aureus (MRSA) in U.S. Healthcare Facilities". Association for Professionals in Infection Control & Epidemiology. June 25, 2007. Archived from the original on September 7, 2007. Retrieved 2007-07-14.
- ^ "Staph Infections and MRSA in Children: Prevention, Symptoms, and Treatment". webmd.com.
- S2CID 211098414.
- ^ PMID 19492967.
- PMID 19667557.)
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link - ^ PMID 12727914.
- ^ PMID 17661706.
- PMID 21304931.
- PMID 24877726.
- PMID 12032344.
- PMID 11274099.
- ^ PMID 21982834.
- ^ S2CID 38231565.
- ^ Sahebnasagh R, Saderi H, Owlia P. Detection of methicillin-resistant Staphylococcus aureus strains from clinical samples in Tehran by detection of the mecA and nuc genes. The First Iranian International Congress of Medical Bacteriology; 4–7 September; Tabriz, Iran. 2011. 195 pp.
- ^ S2CID 40019841. Archived from the originalon 2013-02-12.
- PMID 17409207.
- PMID 21902734.
- ^ PMID 18462090.
- PMID 11418528.
- ^ PMID 16652276.
- ^ PMID 22230333.
- ^ S2CID 11490852.
- PMID 17652653.
- S2CID 8465052.
- PMID 17553275.
- PMID 22196394.[permanent dead link]
- PMID 18976551.
- .
- S2CID 381926.
- ISBN 978-1-904455-29-5.
- ISBN 978-1-904455-18-9.
- ^ Seiken D. "MRSA latex test for PBP2". Archived from the original on 2009-03-10. Retrieved 2009-01-24.
- ^ ISBN 978-1-55581-371-0.
- PMID 19695491.
- ^ Healthcare Settings. (2019, February 28). Retrieved March 19, 2021, from https://www.cdc.gov/mrsa/healthcare/index.html
- ^ McCaughey B. "Unnecessary Deaths: The Human and Financial Costs of Hospital Infections" (PDF) (2nd ed.). Archived from the original (PDF) on July 11, 2007. Retrieved 2007-08-05.
- ^ "Personal Prevention of MRSA Skin Infections". CDC. 9 August 2010. Retrieved 25 May 2017.
 This article incorporates public domain material from websites or documents of the Centers for Disease Control and Prevention.
This article incorporates public domain material from websites or documents of the Centers for Disease Control and Prevention.
- ^ a b c "NIOSH MRSA and the Workplace". United States National Institute for Occupational Safety and Health. Retrieved 2017-05-25.
- ^ a b Strategies to Prevent Hospital-onset Staphylococcus aureus Bloodstream Infections in Acute Care Facilities. (2019, December 16). Retrieved March 19, 2021, from https://www.cdc.gov/hai/prevent/staph-prevention-strategies.html#anchor_1565005744
- ^ CDC (1998). "Guidelines for Infection Control in Health Care Personnel, 1998". Centers for Disease Control and Prevention. Retrieved December 18, 2007.
- PMID 29321078.
- ^ a b c "Living With MRSA" (PDF). Group Health Cooperative/Tacoma-Pierce County Health Dept./Washington State Dept. of Health. Archived from the original (PDF) on 27 October 2011. Retrieved 20 November 2011.
- PMID 15220470.
- PMID 16565219.
- US National Institutes of Health. Archived from the originalon 2011-02-04. Retrieved 20 November 2011.
- S2CID 28807435.
- Her Majesty's Stationery Office. Retrieved January 12, 2018.
- ^ "[ARCHIVED CONTENT] UK Government Web Archive – The National Archives". Archived from the original on 2012-09-19 – via Internet Memory Foundation.
{{cite web}}: CS1 maint: bot: original URL status unknown (link) - ^ "Government Response to the Communities and Local Government Committee Report on the Provision of Public Toilets". Archived from the original on 2012-09-19.
- ^ PMID 24242704.
- PMID 28032484.
- ^ PMID 26758498.
- ^ "FDA Approves Teflaro for Bacterial Infections". Food and Drug Administration.
- PMID 9597254.
- PMID 1829880.
- S2CID 10413339.
- S2CID 15917805.
- S2CID 24390948.
- PMID 9098053.
- PMID 16445718.
- PMID 14563898.
- PMID 21823662.
- PMID 14523769.
- S2CID 27426801.
- PMID 18819981.
- ^ PMID 28509852.
- PMID 14583969.
- ^ "MRSA Infections". Keep Kids Healthy. Archived from the original on December 9, 2007.
- PMID 35065702.
- ^ S2CID 23112235.
- PMID 23449778.
- ^ "Antibiotic Resistance Threats in the United States, 2013 – Antibiotic/Antimicrobial Resistance – CDC". 2019-05-07.
 This article incorporates public domain material from websites or documents of the Centers for Disease Control and Prevention.
This article incorporates public domain material from websites or documents of the Centers for Disease Control and Prevention.
- ^ "ResistanceMap – Antibiotic Resistance". resistancemap.cddep.org. Center for Disease Dynamics, Economics & Policy. 2017. Retrieved 27 May 2017. note: a search must be performed on the website; it is interactive and the statistics are based upon the most current information,
- PMID 17357358.
- S2CID 205988392.
- PMID 16046464.
- S2CID 38816447.
- ^ Jernigan JA, Arnold K, Heilpern K, Kainer M, Woods C, Hughes JM (2006-05-12). "Methicillin-resistant Staphylococcus aureus as community pathogen". Symposium on Community-Associated Methicillin-resistant Staphylococcus aureus (Atlanta, Georgia, U.S.). Cited in Emerg Infect Dis. Centers for Disease Control and Prevention. Retrieved 2007-01-27.
- PMID 16485492.
- ^ "Community-acquired MRSA in Children with no predisposing risk" (PDF). Archived from the original (PDF) on 2017-12-09. Retrieved 2012-04-24.
- PMID 15213324.
- PMID 12390067.
- PMID 18433335.
- PMID 16087824.
- S2CID 22553908.
- PMID 14665563.
- PMID 16798756.
- ^ "Healthcare-associated infections—HAI—CDC" (PDF). cdc.gov. 2019-06-03.
- ^ a b "MRSA Surveillance". Centers for Disease Control and Prevention. April 8, 2011.
- ^ a b "Outcomes in EHCI 2015" (PDF). Health Consumer Powerhouse. 26 January 2016. Archived from the original (PDF) on 6 June 2017. Retrieved 27 January 2016.
- PMID 26265721.
- PMID 33060666.
- ^ Thompson N, Smith-Spark L (31 March 2015). "Thousand-year-old Anglo-Saxon potion kills MRSA superbug". CNN News. CNN/Time Warner. Retrieved 1 April 2015.
- ^ "Bucs' Nicks, Tynes have MRSA infections". Tampa Bay Times. Retrieved 3 June 2017.
- ^ Cohen E (12 October 2015). "MRSA: The tiny bacteria that can tackle giants". CNN. Retrieved 3 June 2017.
- ^ Biddle S, Cush A. "This Australian Says He and His Dead Friend Invented Bitcoin". Gizmodo. Retrieved 3 June 2017.
- PMID 27747300.
- ^ "Hospital Infection Lawsuits are on the Rise – AboutLawsuits.com". www.aboutlawsuits.com. 2 December 2008. Retrieved 3 June 2017.
- ^ "Doc who treated superbug vic was sued before". New York Daily News. November 2007. Retrieved 3 June 2017.
- ^ "MRSA: The Drug-Resistant 'Superbug' That Won't Die". NPR.org. Retrieved 3 June 2017.
- ^ ""Should I Worry About...?" MRSA (TV Episode 2004)". IMDb. 23 September 2004. Retrieved 3 June 2017.
- ^ McKenna M. "Resistance: The Movie That Will Make You Care About Antibiotic Misuse". Wired. Retrieved 3 June 2017.
- ^ "Line of Duty series 4: Everything you need to know". Digital Spy. 24 April 2017. Retrieved 3 June 2017.
- ISBN 978-1-4165-5728-9.
- ^ "Facing the Future of Antibiotic Resistance – The Change Starts Here". HuffPost UK. 18 July 2016. Retrieved 3 June 2017.
- PMID 26009686.
- S2CID 205081324.
- ^ Robson D. "The viruses that may save humanity". Retrieved 2018-09-25.
- PMID 38590439.
- PMID 30890742.
- PMID 33584628.
- PMID 26184396.
Further reading
- The Centers for Disease Control and Prevention information, prevention, statistics, at risk groups, causes, educational resources, and environmental factors.
- National Institute for Occupational Safety and Health information on the bacteria, exposure in the workplace, and reducing risks of being infected.
