Metoprolol
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /mɛˈtoʊproʊlɑːl/, /mɛtoʊˈproʊlɑːl/ |
| Trade names | Lopressor, Metolar XR, Toprol XL, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682864 |
| License data |
|
| Pregnancy category |
|
intravenous | |
| Drug class | Beta blocker |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 50% (single dose)[2] 70% (repeated administration)[3] |
| Protein binding | 12% |
| Metabolism | Liver via CYP2D6, CYP3A4 |
| Elimination half-life | 3–7 hours |
| Excretion | Kidney |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture |
| Melting point | 120 °C (248 °F) |
| |
| |
| (verify) | |
Metoprolol, sold under the brand name Lopressor among others, is a medication used to treat
Common side effects include trouble sleeping,
Metoprolol was first made in 1969, patented in 1970, and approved for medical use in 1978.
Medical uses
Metoprolol is used for a number of conditions, including
Both oral and intravenous forms of metoprolol are available for administration.[18] The different salt versions of metoprolol – metoprolol tartrate and metoprolol succinate – are approved for different conditions and are not interchangeable.[19][20]Off-label uses include supraventricular tachycardia and thyroid storm.[18]
Adverse effects
Adverse effects, especially with higher doses, include dizziness, drowsiness, fatigue, diarrhea, unusual dreams, trouble sleeping, depression, and vision problems such as blurred vision or dry eyes.
There may also be an impact on blood sugar levels and it can potentially mask signs of low blood sugar.[21]
The safety of metoprolol during pregnancy is not fully established.[27][28]
Precautions
Metoprolol reduces long-term mortality and hospitalisation due to worsening heart failure.[29] A meta-analysis further supports reduced incidence of heart failure worsening in patients treated with beta-blockers compared to placebo.[30] However, in some circumstances, particularly when initiating metoprolol in patients with more symptomatic disease, an increased prevalence of hospitalisation and mortality has been reported within the first two months of starting.[31] Patients should monitor for swelling of extremities, fatigue, and shortness of breath.[32]
This medicine may cause changes in blood sugar levels or cover up signs of low blood sugar, such as a rapid pulse rate.[32] It also may cause some people to become less alert than they are normally, making it dangerous for them to drive or use machines.[32]
Pregnancy and breastfeeding
Risk for the fetus has not been ruled out, per being rated pregnancy category C in Australia, meaning that it may be suspected of causing harmful effects on the human fetus (but no malformations).[7] It appears to be safe in breastfeeding.[8]
Overdose
Excessive doses of metoprolol can cause severe hypotension, bradycardia, metabolic acidosis, seizures, and cardiorespiratory arrest. Blood or plasma concentrations may be measured to confirm a diagnosis of overdose or poisoning in hospitalized patients or to assist in a medicolegal death investigation. Plasma levels are usually less than 200 μg/L during therapeutic administration, but can range from 1–20 mg/L in overdose victims.[33][34][35]
Pharmacology
Mechanism of action
Metoprolol is a
Metoprolol exerts its effects by blocking the action of certain neurotransmitters, specifically adrenaline and noradrenaline. It does this by selectively binding to and antagonizing β-1 adrenergic receptors in the body. When adrenaline (epinephrine) or noradrenaline (norepinephrine) are released from nerve endings or secreted by the adrenal glands, they bind to β-1 adrenergic receptors found primarily in cardiac tissues such as the heart. This binding activates these receptors, leading to various physiological responses, including an increase in heart rate, force of contraction (inotropic effect), conduction speed through electrical pathways in the heart, and release of renin from the kidneys. Metoprolol competes with adrenaline and noradrenaline for binding sites on these β-1 receptors. By occupying these receptor sites without activating them, metoprolol blocks or inhibits their activation by endogenous catecholamines like adrenaline or noradrenaline.[37]
Metoprolol blocks β1-adrenergic receptors in
Through this mechanism of selective blockade at beta-(β)-1 receptors, metoprolol exerts the following effects:
- Heart rate reduction, i.e., decrease of the resting heart rate (negative chronotropic effect) and reduction of excessive elevations resulting from exercise or stress.[37]
- Reduction of the force of contraction, i.e., decrease in contractility (negative inotropic effect), which lessens how hard each heartbeat contracts.[37]
- Decrease in cardiac output, i.e., decrease in both heart rate and contractility within myocardium cells, where beta-(β)-1 is predominantly located, overall blood output per minute lowers called cardiac output/dysfunction, allowing decreased demands placed onto impaired hearts, reducing oxygen demand-supply mismatch.[37]
- Lowering of blood pressure.[37]
- Antiarrhythmic effects, such as supraventricular tachycardia prevention. Metoprolol also prevents electrical wave propagation.[37]
Pharmacokinetics
Metoprolol is mostly absorbed from the intestine with an absorption fraction of 0.95. The systemic bioavailability after oral administration is approximately 50%.[37] Less than 5% of an orally administered dose of metoprolol is excreted unchanged in urine; most of it is eliminated in metabolized form through feces via bile secretion into the intestines.[37]
Metoprolol undergoes extensive metabolism in the liver, mainly α-hydroxylation and O-demethylation through various cytochrome P450 enzymes such as CYP2D6 (primary), CYP3A4, CYP2B6, and CYP2C9. The primary metabolites formed are α-hydroxymetoprolol and O-demethylmetoprolol.[37][39][10]
Metoprolol is classified as a moderately
Chemistry
Metoprolol was synthesized and its activity discovered in 1969.[12] The specific agent in on-market formulations of metoprolol is either metoprolol tartrate or metoprolol succinate, where tartrate is an immediate-release formulation and the succinate is an extended-release formulation (with 100 mg metoprolol tartrate corresponding to 95 mg metoprolol succinate).[40]
Stereochemistry
This section needs expansion with: a statement from secondary sources of the importance/relevance of this information. You can help by adding to it. (November 2022) |
Metoprolol contains a stereocenter and consists of two
| Enantiomers of metoprolol | |
|---|---|
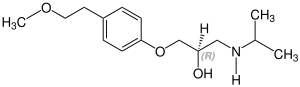 CAS-Nummer: 81024-43-3 |
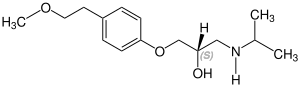 CAS-Nummer: 81024-42-2 |
Society and culture
Legal status
Metoprolol was approved for medical use in the United States in August 1978.[11]
Economics
In the 2000s, a lawsuit was brought against the manufacturers of Toprol XL (a time-release formula version of metoprolol) and its generic equivalent (metoprolol succinate) claiming that to increase profits, lower cost generic versions of Toprol XL were intentionally kept off the market. It alleged that the pharmaceutical companies AstraZeneca AB, AstraZeneca LP, AstraZeneca Pharmaceuticals LP, and Aktiebolaget Hassle violated antitrust and
Sport
Because beta blockers can be used to reduce heart rate and minimize tremors, which can enhance performance in sports such as archery,[43][44] metoprolol is banned by the world anti-doping agency in some sports.[44]
References
- ^ "Lopressor Product information". Health Canada. 22 October 2009. Retrieved 20 June 2023.
- ^ "Metolar 25/50 (metoprolol tartrate) tablet" (PDF). U.S. Food and Drug Administration (FDA). Archived (PDF) from the original on 3 March 2016. Retrieved 5 May 2015.
- ISBN 978-3852001814.
- ^ a b c d e f g h "Metoprolol". The American Society of Health-System Pharmacists. Archived from the original on 12 March 2014. Retrieved 21 April 2014.
- ISBN 978-9350259658. Archivedfrom the original on 7 July 2017.
- ISBN 978-1455706051.
- ^ a b "Prescribing medicines in pregnancy database". Australian Government. 3 March 2014. Archived from the original on 8 April 2014. Retrieved 22 April 2014.
- ^ ISBN 978-0781728454. Archivedfrom the original on 7 July 2017.
- S2CID 1042204.
- ^ PMID 23665868.
- ^ a b "Lopressor: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Retrieved 20 June 2023.
- ^ ISBN 978-0792399728. Archivedfrom the original on 3 March 2017.
- ISBN 978-3527607495.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Metoprolol – Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- PMID 1352658.
- ^ PMID 30422518.
- ^ "Metoprolol vs Toprol-XL Comparison". Drugs.com. 1 August 2019. Retrieved 24 September 2019.
- ^ Eske J (25 September 2019). "Metoprolol tartrate vs. succinate: Differences in uses and effects". Medical News Today. Archived from the original on 25 September 2019. Retrieved 24 September 2019.
- ^ a b c "Metoprolol (Oral Route)". Mayo Clinic.
- ISBN 978-1416023203.
- PMID 27040294.
- ^ "Metoprolol". Drugs.com. Archived from the original on 21 January 2010.
- PMID 21180298.
- ^ "Common questions about metoprolol". National Health Service (NHS). 15 March 2022.
- PMID 30000215.
- PMID 35952115.
- PMID 10714728.
- PMID 23796325.
- S2CID 24119608.
- ^ a b c "Metoprolol (Oral Route) Precautions". Drug Information. Mayo Clinic. Archived from the original on 16 April 2009.
- PMID 19655287.
- PMID 15484221.
- ^ Baselt R (2008). Disposition of Toxic Drugs and Chemicals in Man (8th ed.). Foster City, CA: Biomedical Publications. pp. 1023–1025.
- ^ PMID 33572109.
- ^ S2CID 250094483.
- ^ PMID 26203906.
- S2CID 1570605.
- ^ Cupp M (2009). "Alternatives for Metoprolol Succinate" (PDF). Pharmacist's Letter / Prescriber's Letter. 25 (250302). Retrieved 6 July 2012.
- ISBN 978-3946057109, S. 200.
- ^ "$11 Million Settlement Reached in Lawsuit Involving the Heart Medication, Toprol XL, and its generic equivalent, metoprolol succinate". www.prnewswire.com (Press release).
- PMID 26648655.
- ^ a b "The Prohibited List". World Anti Doping Agency. 3 January 2023. Retrieved 20 June 2023.
Further reading
- Dean L (2017). "Metoprolol Therapy and CYP2D6 Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. PMID 28520381. Bookshelf ID: NBK425389.
