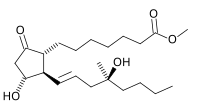
Misoprostol
 | |
| Clinical data | |
|---|---|
| Trade names | Cytotec, Misodel, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a689009 |
| License data |
|
| Pregnancy category |
|
under the tongue | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | extensively absorbed |
| Protein binding | 80–90% (active metabolite, misoprostol acid) |
| Metabolism | Liver (extensive to misoprostic acid) |
| Elimination half-life | 20–40 minutes |
| Excretion | Urine (80%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Misoprostol is a synthetic
Common side effects include diarrhea and abdominal pain.[11] It is in pregnancy category X, meaning that it is known to result in negative outcomes for the fetus if taken during pregnancy.[11] In rare cases, uterine rupture may occur.[11] It is a prostaglandin analogue—specifically, a synthetic prostaglandin E1 (PGE1).[11]
Misoprostol was developed in 1973.
Medical uses
Ulcer prevention
Misoprostol is used for the prevention of
However, even in the treatment of NSAID-induced ulcers, omeprazole proved to be at least as effective as misoprostol,[22] but was significantly better tolerated, so misoprostol should not be considered a first-line treatment. Misoprostol-induced diarrhea and the need for multiple daily doses (typically four) are the main issues impairing compliance with therapy.[medical citation needed]
Labor induction
Misoprostol is commonly used for
Oxytocin has long been used as the standard agent for labor induction, but does not work well when the cervix is not yet ripe. Misoprostol also may be used in conjunction with oxytocin.[24]
Between 2002 and 2012, a misoprostol vaginal insert was studied, and was approved in the EU.[25][26][27] It was not approved for use in the United States, and the US FDA still considers cervical ripening and labor induction to be outside of the approved uses for misoprostol.[28][29]
Myomectomy
When administered prior to
Abortion
Misoprostol is used either alone or in conjunction with another medication (mifepristone or methotrexate) for medical abortions as an alternative to surgical abortion.[31] Medical abortion has the advantage of being less invasive, and more autonomous, self-directed, and discreet. It is preferred by some women because it feels more natural, as the drugs induce a miscarriage.[32] It is also more easily accessible in places where abortion is illegal.[33] The World Health Organization (WHO) provides clear guidelines on the use, benefits and risks of misoprostol for abortions.[34]
Misoprostol is most effective when it is used in combination with methotrexate or mifepristone (RU-486).
Most large studies recommend a protocol for the use of misoprostol in combination with mifepristone.[38][39] Together they are effective in around 95% for early pregnancies.[40] Misoprostol alone may be more effective in earlier gestation.[41]
Misoprostol can also be used to dilate the cervix in preparation for a surgical abortion, particularly in the second trimester (either alone or in combination with laminaria stents). Vaginal misoprostol can also be used to facilitate intrauterine device insertion after previous insertion failure.[42]
Misoprostol by mouth is the least effective treatment for producing complete abortion in a period of 24 hours due to the liver's first-pass effect which reduces the bioavailability of the misoprostol. Vaginal and sublingual routes result in greater efficacy and extended duration of action because these routes of administration allow the drug to be directly absorbed into circulation by bypassing the liver first-pass effect.[43][17][18]
Hematocrit or Hb tests and Rh testing are recommended before use for abortion confirmation of pregnancy.[44] Following use, it is recommended that people attend a follow-up visit 2 weeks after treatment. If used for treatment of complete abortion, a pregnancy test, physical examination of the uterus, and ultrasound should be performed to ensure success of treatment. Surgical management is possible in the case of failed treatment.[43]
Early pregnancy loss
Misoprostol may be used to complete a
Misoprostol is regularly used in some Canadian hospitals for labour induction for fetal deaths early in pregnancy, and for termination of pregnancy for fetal anomalies. A low dose is used initially, then doubled for the remaining doses until delivery. In the case of a previous Caesarian section, however, lower doses are used.
Postpartum bleeding
Misoprostol is also used to prevent and treat
Insertion of intrauterine contraceptive device
In women with prior caesarean section or prior failure of insertion of an intrauterine contraceptive device, pre-procedure administration of misoprostol reduces the rate of failure of insertion of intrauterine contraceptive device. However, due to a higher rate of adverse effects, routine use of misoprostol for this purpose in other women is not supported by the data.[53]
Other
For cervical ripening in advance of endometrial biopsy to reduce the need for use of a tenaculum or cervical dilator.[citation needed]
There is limited evidence supporting the use of misoprostol for the treatment of trigeminal neuralgia in patients with multiple sclerosis.[54][55]
Adverse effects
The most commonly reported adverse effect of taking misoprostol by mouth for the prevention of stomach ulcers is diarrhea. In clinical trials, an average 13% of people reported diarrhea, which was dose-related and usually developed early in the course of therapy (after 13 days) and was usually self-limiting (often resolving within 8 days), but sometimes (in 2% of people) required discontinuation of misoprostol.[56]
The next most commonly reported adverse effects of taking misoprostol by mouth for the prevention of gastric ulcers are:
There are increased side effects with sublingual or oral misoprostol, compared to a low dose (400 μg) vaginal misoprostol. However, low dose vaginal misoprostol was linked with low complete abortion rate.[43] The study concluded that sublingually administered misoprostol dosed at 600 μg or 400 μg had greater instances of fever and diarrhea due to its quicker onset of action, higher peak concentration and bioavailability in comparison to vaginal or oral misoprostol.[43]
For the indication of medical abortion, bleeding and cramping is commonly experienced after administration of misoprostol. Bleeding and cramping is likely to be greater than that experienced with menses, however, emergency care is advised if bleeding is excessive.[44]
Misoprostol should not be taken by pregnant women with wanted pregnancies to reduce the risk of NSAID-induced gastric ulcers because it increases uterine tone and contractions in pregnancy, which may cause partial or complete abortions, and because its use in pregnancy has been associated with birth defects.[56][57]
All cervical ripening and induction agents can cause uterine hyperstimulation, which can negatively affect the blood supply to the fetus and increases the risk of complications such as uterine rupture.[58] Concern has been raised that uterine hyperstimulation that occurs during a misoprostol-induced labor is more difficult to treat than hyperstimulation during labors induced by other drugs.[59] Because the complications are rare, it is difficult to determine if misoprostol causes a higher risk than do other cervical ripening agents. One estimate is that it would require around 61,000 people enrolled in randomized controlled trials to detect a difference in serious fetal complications and about 155,000 people to detect a difference in serious maternal complications.[60]
Contraindications
It is recommended that medical treatment for
Pharmacology
Mechanism of action
Misoprostol, a prostaglandin analogue, binds to myometrial cells to cause strong myometrial contractions leading to expulsion of tissue. This agent also causes cervical ripening with softening and dilation of the cervix. Misoprostol binds to and stimulates prostaglandin EP2 receptors, prostaglandin EP3 receptor and prostaglandin EP4 receptor but not prostaglandin EP1 receptor and therefore is expected to have a more restricted range of physiological and potentially toxic actions than prostaglandin E2 or other analogs which activate all four prostaglandin receptors.[61]
Society and culture
In August 2000, a letter from G.D. Searle, LLC, the inventor of the drug,[62][63] generated controversy by warning against its use by pregnant women because of its ability to induce abortion, citing reports of maternal and fetal deaths when it was used to induce labor.[64] The American College of Obstetricians and Gynecologists holds that substantial evidence supports the use of misoprostol for induction of labor, a position it reaffirmed in response to the Searle letter.[65] It is on the World Health Organization's List of Essential Medicines.[21]
A vaginal form of the medication is sold in the EU under the names Misodel[66] and Mysodelle[67] for use in labor induction.[medical citation needed]
Black market
Misoprostol is used for self-induced abortions in Brazil, where black market prices exceed US$100 per dose. Illegal medically unsupervised misoprostol abortions in Brazil are associated with a lower complication rate than other forms of illegal self-induced abortion, but are still associated with a higher complication rate than legal, medically supervised surgical and medical abortions. Failed misoprostol abortions are associated with birth defects in some cases.[68][69][70][71][72] Low-income and immigrant populations in New York City have also been observed to use self-administered misoprostol to induce abortions, as this method is much cheaper than a surgical abortion (about $2 per dose).[73] The drug is readily available in Mexico.[74] Use of misoprostol has also increased in Texas in response to increased regulation of abortion providers.[75] Following the United States Supreme Court decision of Dobbs v. Jackson Women's Health Organization, many states restricted access to legal abortion services, including medication abortion using misoprostol. As a result of these restrictions, it was reported that there was an increase in self-managed abortions by women in the United States. Many women purchased the pills from overseas online pharmacies or obtained misoprostol from Mexico.[76]
References
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". Archived from the original on 14 April 2023. Retrieved 14 April 2023.
- ^ "TGA eBS - Product and Consumer Medicine Information Licence". Archived from the original on 14 April 2023. Retrieved 14 April 2023.
- FDA. Retrieved 22 October 2023.
- ^ "Angusta (Norgine Pty Ltd)". Therapeutic Goods Administration (TGA). 13 January 2023. Archived from the original on 18 March 2023. Retrieved 9 April 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 15 August 2023.
- ^ "Angusta 25 microgram tablets - Summary of Product Characteristics (SmPC)". (emc). 8 April 2022. Archived from the original on 10 April 2023. Retrieved 9 April 2023.
- ^ "Cytotec- misoprostol tablet". DailyMed. 9 July 2021. Archived from the original on 18 April 2023. Retrieved 13 April 2023.
- ^ "List of nationally authorised medicinal products. Misoprostol (gastrointestinal indication). Procedure no.: PSUSA/00010353/202005" (PDF). European Medicines Agency. 14 January 2021. Archived (PDF) from the original on 29 June 2022. Retrieved 8 August 2021.
- ^ "List of nationally authorised medicinal products. Misoprostol (gastrointestinal indication). Procedure no.: PSUSA/00010291/202006" (PDF). European Medicines Agency. 14 January 2021. Archived (PDF) from the original on 29 June 2022. Retrieved 8 August 2021.
- S2CID 1052260.
- ^ a b c d e f g "Misoprostol". The American Society of Health-System Pharmacists. Archived from the original on 21 February 2015. Retrieved 20 February 2015.
- ^ PMID 35608608.
- S2CID 28486936.
- PMID 30531568.
- PMID 25701235.
- ^ Prager S. "Early Pregnancy Loss" (PDF). ACOG Practice Bulletin (200). ACOG. Archived (PDF) from the original on 2 June 2021. Retrieved 2 June 2021.
- ^ S2CID 13149908.
- ^ a b c "Early Pregnancy Loss". ACOG. 20 January 2015. Archived from the original on 19 January 2023. Retrieved 25 June 2023.
- S2CID 10997666.
- ISBN 9781444358476. Archivedfrom the original on 29 June 2022. Retrieved 18 August 2020.
- ^ hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- PMID 9494149.
- PMID 11136959.
- ^ PMID 9107114.
- ^ "Misodel, Ferring's Removable Misoprostol Vaginal Delivery System, Approved for Labour Induction in European Decentralised Procedure" (Press release). Ferring Pharmaceuticals. 17 October 2013. Retrieved 25 June 2023 – via Business Wire.
- S2CID 31090241.
- PMID 33919898.
- ^ "Safety Information, Cytotec (misoprostol) Tablets". U.S. Food and Drug Administration (FDA). 11 December 2012. Archived from the original on 20 January 2017. Retrieved 16 March 2017.
- ^ "Misoprostol (marketed as Cytotec) Information". U.S. Food and Drug Administration (FDA). 10 July 2015. Archived from the original on 27 May 2020. Retrieved 17 August 2020.
- S2CID 220307235.
- ^ "Medical methods for first trimester abortion". World Health Organization (WHO). Archived from the original on 14 February 2016. Retrieved 4 February 2016.
- S2CID 30491792.
- from the original on 28 April 2016. Retrieved 4 February 2016.
- ^ "Medical methods for first trimester abortion". The WHO Medical Reproductive Library. Archived from the original on 2 August 2014. Retrieved 22 June 2014.
- OCLC 812323067.
- ^ "Mifepristone". The American Society of Health System Pharmacists. 18 January 2023. Archived from the original on 22 December 2015. Retrieved 25 February 2023.
- ^ What is the "Mexican abortion pill" and how safe is it? Archived 30 July 2013 at the Wayback Machine Jen Gunter, 27 July 2013
- ^ "Annotated Bibliography on Misoprostol Alone for Early Abortion" (PDF). Gynuity Health Projects. Archived (PDF) from the original on 29 September 2007. Retrieved 22 August 2006.
- ^ "Annotated Bibliography - Misoprostol for Early Abortion". Gynuity Health Projects. 1 September 2004. Archived from the original on 18 May 2022. Retrieved 25 June 2023.
- ^ Providing medical abortion in low-resource settings (PDF) (2 ed.). Gynuity Health Projects. 2009. p. 4. Archived (PDF) from the original on 22 February 2016. Retrieved 31 August 2015.
- ^ "Instructions for Use: Abortion Induction with Misoprostol in Pregnancies up to 9 Weeks LMP" (PDF). Gynuity Health Projects. 2003. Archived (PDF) from the original on 29 September 2007. Retrieved 24 August 2006.
- S2CID 160014019.
- ^ PMID 28490770..
 Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine
Material was copied from this source, which is available under a Creative Commons Attribution 4.0 International License Archived 16 October 2017 at the Wayback Machine - ^ S2CID 23951273.
- PMID 31206170.
- S2CID 24561459.
- PMID 9699753.
- S2CID 23335277.
- S2CID 23593148.
- S2CID 22275092.
- ^ Sanghvi H, Zulkarnain M, Chanpong G (2009). Blouse A, Lewison D (eds.). Prevention of Postpartum Hemorrhage at Home Birth: A Program Implementation Guide (PDF). United States Agency for International Development. Archived (PDF) from the original on 6 December 2013.[page needed]
- PMID 22879523.
- from the original on 29 June 2022. Retrieved 21 June 2022.
- S2CID 36493785.
- PMID 30178160.
- ^ a b c "Cytotec- misoprostol tablet". DailyMed. 9 July 2021. Archived from the original on 18 April 2023. Retrieved 25 June 2023.
- ^ "Cytotec 200mcg Tablets - Summary of Product Characteristics (SmPC)". (emc). 26 April 2021. Archived from the original on 1 July 2022. Retrieved 25 June 2023.
- PMID 16754739.
- ISBN 0-520-24596-2., which cites:
- Wing DA, Paul RH (July 1996). "A comparison of differing dosing regimens of vaginally administered misoprostol for preinduction cervical ripening and labor induction". American Journal of Obstetrics and Gynecology. 175 (1): 158–64. PMID 8694043.
- Wing DA, Rahall A, Jones MM, Goodwin TM, Paul RH (June 1995). "Misoprostol: an effective agent for cervical ripening and labor induction". American Journal of Obstetrics and Gynecology. 172 (6): 1811–6. PMID 7778637.
- Wing DA, Paul RH (July 1996). "A comparison of differing dosing regimens of vaginally administered misoprostol for preinduction cervical ripening and labor induction". American Journal of Obstetrics and Gynecology. 175 (1): 158–64.
- ^ Goldberg & Wing 2003, which cites:
- Weeks A, Alfirevic Z (September 2006). "Oral misoprostol administration for labor induction". Clinical Obstetrics and Gynecology. 49 (3): 658–71. S2CID 13063131.
- Weeks A, Alfirevic Z (September 2006). "Oral misoprostol administration for labor induction". Clinical Obstetrics and Gynecology. 49 (3): 658–71.
- S2CID 1513449.
- ISBN 9781845698386. Archivedfrom the original on 29 June 2022. Retrieved 10 May 2022.
- ISBN 9781784784638. Archivedfrom the original on 29 June 2022. Retrieved 10 May 2022.
- PMID 12867908.
- ^ Goldberg & Wing 2003, which cites:
- American College of Obstetricians and Gynecologists (November 1999). "Induction of labor with misoprostol". ACOG Committee Opinion No. 228. Washington, DC.
- American College of Obstetricians and Gynecologists (November 1999). "Response to Searle's drug warning on misoprostol". ACOG Committee Opinion No. 248. Washington, DC.
- ^ "List of nationally authorized medicinal products" (PDF). Archived (PDF) from the original on 2 November 2021. Retrieved 29 June 2022.
- ^ "List of nationally authorised medicinal products" (PDF). Archived (PDF) from the original on 3 March 2022. Retrieved 26 June 2023.
- S2CID 30907894.
- PMID 8143449.
- PMID 8212093.
- PMID 7950553.
- PMID 8368254.
- ^ Leland J (2 October 2005). "Abortion Might Outgrow Its Need for Roe v. Wade". The New York Times. Archived from the original on 18 May 2013. Retrieved 6 March 2014.
- ^ Eckholm E (13 July 2013). "In Mexican Pill, a Texas Option for an Abortion". The New York Times. Archived from the original on 14 July 2013. Retrieved 14 July 2013.
- ^ Hellenstein E (27 June 2014). "The Rise of the DIY Abortion in Texas". The Atlantic. Archived from the original on 2 March 2017.
- from the original on 8 February 2023. Retrieved 8 February 2023.
