Molecular clock
| Part of a series on |
| Evolutionary biology |
|---|
 |
The molecular clock is a figurative term for a technique that uses the
.Early discovery and genetic equidistance
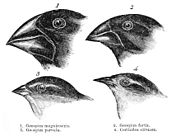
The notion of the existence of a so-called "molecular clock" was first attributed to Émile Zuckerkandl and Linus Pauling who, in 1962, noticed that the number of amino acid differences in hemoglobin between different lineages changes roughly linearly with time, as estimated from fossil evidence.[1] They generalized this observation to assert that the rate of evolutionary change of any specified protein was approximately constant over time and over different lineages (known as the molecular clock hypothesis).
The genetic equidistance phenomenon was first noted in 1963 by Emanuel Margoliash, who wrote: "It appears that the number of residue differences between cytochrome c of any two species is mostly conditioned by the time elapsed since the lines of evolution leading to these two species originally diverged. If this is correct, the cytochrome c of all mammals should be equally different from the cytochrome c of all birds. Since fish diverges from the main stem of vertebrate evolution earlier than either birds or mammals, the cytochrome c of both mammals and birds should be equally different from the cytochrome c of fish. Similarly, all vertebrate cytochrome c should be equally different from the yeast protein."[2] For example, the difference between the cytochrome c of a carp and a frog, turtle, chicken, rabbit, and horse is a very constant 13% to 14%. Similarly, the difference between the cytochrome c of a bacterium and yeast, wheat, moth, tuna, pigeon, and horse ranges from 64% to 69%. Together with the work of Emile Zuckerkandl and Linus Pauling, the genetic equidistance result led directly to the formal postulation of the molecular clock hypothesis in the early 1960s.[3]
Similarly,
Relationship with neutral theory
The observation of a clock-like rate of molecular change was originally purely phenomenological. Later, the work of Motoo Kimura[6] developed the neutral theory of molecular evolution, which predicted a molecular clock. Let there be N individuals, and to keep this calculation simple, let the individuals be haploid (i.e. have one copy of each gene). Let the rate of neutral mutations (i.e. mutations with no effect on fitness) in a new individual be . The probability that this new mutation will become fixed in the population is then 1/N, since each copy of the gene is as good as any other. Every generation, each individual can have new mutations, so there are N new neutral mutations in the population as a whole. That means that each generation, new neutral mutations will become fixed. If most changes seen during molecular evolution are neutral, then fixations in a population will accumulate at a clock-rate that is equal to the rate of neutral mutations in an individual.
Nevertheless, there is some controversy regarding this view. Recently,
Calibration
To use molecular clocks to estimate divergence times, molecular clocks need to be "calibrated". This is because molecular data alone does not contain any information on absolute times. For viral phylogenetics and ancient DNA studies—two areas of evolutionary biology where it is possible to sample sequences over an evolutionary timescale—the dates of the intermediate samples can be used to calibrate the molecular clock. However, most phylogenies require that the molecular clock be calibrated using independent evidence about dates, such as the fossil record.[9] There are two general methods for calibrating the molecular clock using fossils: node calibration and tip calibration.[10]
Node calibration
Sometimes referred to as node dating, node calibration is a method for time-scaling
Tip calibration
Sometimes referred to as tip dating, tip calibration is a method of molecular clock calibration in which fossils are treated as taxa and placed on the tips of the tree. This is achieved by creating a matrix that includes a molecular dataset for the extant taxa along with a morphological dataset for both the extinct and the extant taxa.[14] Unlike node calibration, this method reconstructs the tree topology and places the fossils simultaneously. Molecular and morphological models work together simultaneously, allowing morphology to inform the placement of fossils.[10] Tip calibration makes use of all relevant fossil taxa during clock calibration, rather than relying on only the oldest fossil of each clade. This method does not rely on the interpretation of negative evidence to infer maximum clade ages.[14]
Expansion calibration
Demographic changes in populations can be detected as fluctuations in historical coalescent effective population size from a sample of extant genetic variation in the population using coalescent theory.[17][18][19] Ancient population expansions that are well documented and dated in the geological record can be used to calibrate a rate of molecular evolution in a manner similar to node calibration. However, instead of calibrating from the known age of a node, expansion calibration uses a two-epoch model of constant population size followed by population growth, with the time of transition between epochs being the parameter of interest for calibration.[20][21] Expansion calibration works at shorter, intraspecific timescales in comparison to node calibration, because expansions can only be detected after the most recent common ancestor of the species in question. Expansion dating has been used to show that molecular clock rates can be inflated at short timescales[20] (< 1 MY) due to incomplete fixation of alleles, as discussed below[22][23]
Total evidence dating
This approach to tip calibration goes a step further by simultaneously estimating fossil placement, topology, and the evolutionary timescale. In this method, the age of a fossil can inform its phylogenetic position in addition to morphology. By allowing all aspects of tree reconstruction to occur simultaneously, the risk of biased results is decreased.[10] This approach has been improved upon by pairing it with different models. One current method of molecular clock calibration is total evidence dating paired with the fossilized birth-death (FBD) model and a model of morphological evolution.[24] The FBD model is novel in that it allows for "sampled ancestors", which are fossil taxa that are the direct ancestor of a living taxon or lineage. This allows fossils to be placed on a branch above an extant organism, rather than being confined to the tips.[25]
Methods
Bayesian methods can provide more appropriate estimates of divergence times, especially if large datasets—such as those yielded by phylogenomics—are employed.[26]
Non-constant rate of molecular clock
Sometimes only a single divergence date can be estimated from fossils, with all other dates inferred from that. Other sets of species have abundant fossils available, allowing the hypothesis of constant divergence rates to be tested. DNA sequences experiencing low levels of
In addition to such variation in rate with genomic position, since the early 1990s variation among taxa has proven fertile ground for research too,[28] even over comparatively short periods of evolutionary time (for example mockingbirds[29]). Tube-nosed seabirds have molecular clocks that on average run at half speed of many other birds,[30] possibly due to long generation times, and many turtles have a molecular clock running at one-eighth the speed it does in small mammals, or even slower.[31] Effects of small population size are also likely to confound molecular clock analyses. Researchers such as Francisco J. Ayala have more fundamentally challenged the molecular clock hypothesis.[32][33][34] According to Ayala's 1999 study, five factors combine to limit the application of molecular clock models:
- Changing generation times (If the rate of new mutations depends at least partly on the number of generations rather than the number of years)
- Population size (Genetic drift is stronger in small populations, and so more mutations are effectively neutral)
- Species-specific differences (due to differing metabolism, ecology, evolutionary history, ...)
- Change in function of the protein studied (can be avoided in closely related species by utilizing non-coding DNA sequences or emphasizing silent mutations)
- Changes in the intensity of natural selection.
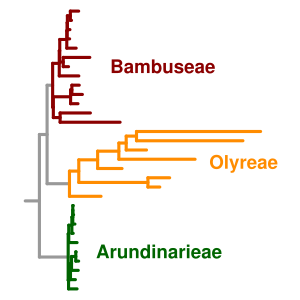
Molecular clock users have developed workaround solutions using a number of statistical approaches including
The molecular clock runs into particular challenges at very short and very long timescales. At long timescales, the problem is
At very short time scales, many differences between samples do not represent
Uses
The molecular clock technique is an important tool in
See also
- Charles Darwin
- Gene orders
- Human mitochondrial molecular clock
- Mitochondrial Eve and Y-chromosomal Adam
- Models of DNA evolution
- Molecular evolution
- Neutral theory of molecular evolution
- Glottochronology
References
- ^ Zuckerkandl E, Pauling (1962). "Molecular disease, evolution, and genic heterogeneity". In Kasha, M., Pullman, B (eds.). Horizons in Biochemistry. Academic Press, New York. pp. 189–225.
- PMID 14077496.
- S2CID 14261833.
- PMID 4962458.
- S2CID 7349579.
- S2CID 4161261.
- ISSN 1471-0064.
- PMID 2068073.
- PMID 17047029.
- ^ PMID 27325838.
- S2CID 17647010.
- ^ PMID 22367748.
- PMID 12538260.
- ^ PMID 26439502.
- S2CID 252353611.
- S2CID 3895351.
- PMID 1316531.
- S2CID 27134675.
- PMID 21753753.
- ^ PMID 21926069.
- PMID 26683588.
- PMID 26333662.
- ^ PMID 15814826.
- PMID 25009181.
- PMID 28173531.
- PMID 22628470.
- S2CID 8260277.
- S2CID 23887665.
- S2CID 51797284.
- S2CID 20390465.
- PMID 1584014.
- PMID 10070256. Archived from the originalon 16 December 2012.
- S2CID 28166727.
- "No Missing Link? Evolutionary Changes Occur Suddenly, Professor Says". ScienceDaily (Press release). 12 February 2007.
- PMID 31111152.
- PMID 16683862.
- S2CID 9791493.
- ^ Marshall, D. C., et al. 2016. Inflation of molecular clock rates and dates: molecular phylogenetics, biogeography, and diversification of a global cicada radiation from Australasia (Hemiptera: Cicadidae: Cicadettini). Systematic Biology 65(1):16–34.
- PMID 19661199.
Further reading
- Ho, S.Y.W., ed. (2020). The Molecular Evolutionary Clock: Theory and Practice. Springer, Cham. S2CID 231672167.
- Kumar S (August 2005). "Molecular clocks: four decades of evolution". Nature Reviews. Genetics. 6 (8): 654–662. S2CID 14261833.
- Morgan GJ (1998). "Emile Zuckerkandl, Linus Pauling, and the molecular evolutionary clock, 1959-1965". Journal of the History of Biology. 31 (2): 155–178. S2CID 5660841.
- Zuckerkandl E, Pauling LB (1965). "Evolutionary divergence and convergence in proteins". In Bryson V, Vogel HJ (eds.). Evolving Genes and Proteins. Academic Press, New York. pp. 97–166.

