Muscarine
| |

| |
| Names | |
|---|---|
| IUPAC name
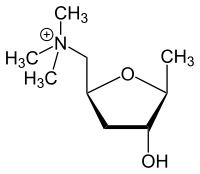
2,5-Anhydro-1,4,6-trideoxy-6-(trimethylazaniumyl)-D-ribo-hexitol
| |
| Systematic IUPAC name
1-[(2S,4R,5S)-4-Hydroxy-5-methyloxolan-2-yl]-N,N,N-trimethylmethanaminium | |
| Other names
L-(+)-muscarine, muscarin, (2S,4R,5S)-(4-hydroxy-5-methyl-tetrahydrofuran-2-ylmethyl)-trimethyl-ammonium
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.005.541 |
IUPHAR/BPS |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C9H20NO2+ | |
| Molar mass | 174.26 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Muscarine, L-(+)-muscarine, or muscarin is a
Muscarine is a selective agonist of the muscarinic acetylcholine receptors.
History
The name muscarine derives from that of
Muscarine was the first
Structure and reactivity
Muscarine mimics the function of the natural neurotransmitter acetylcholine in the muscarinic part of the cholinergic nervous system, despite the less flexible structure due to the five-membered ring in the molecular skeleton. With the exception of the double bonded oxygen, all of the acetylcholine structure is present in the right bottom side of muscarine (see Figure 3 below for comparison of both structures).[6]
There are two mirror forms of muscarine, named: 2S-muscarine and 2R-muscarine.
-
Figure 1. The structural formula of 2S-muscarine.
-
Figure 2. The structural formula of 2R-muscarine.
-
Figure 3. Acetylcholine for comparison.
Efficient synthesis of (+)-muscarine
The scheme below represents a very efficient way of the synthesis of (+)-muscarine according to the scientists Chan and Li in the Canadian journal of Chemistry in 1992.[7] S-(−)-Ethyl lactate (2)(Figure 4) is converted into the 2,6-dichlorobenzyl ether (3). Diisobutylaluminium hydride (DIBAL) reduction of the 2,6-dichlorobenzyl ether gives the aldehyde (4). Treatment of the crude aldehyde with allyl bromide and zinc powder in water with NH4Cl as catalyst resulted in an anti:syn mixture of 5a and 5b. Treatment of 5a with iodine in CH3CN at 0 °C gives the cyclized product 6a. Finally treatment of 6a with excess trimethylamine in ethanol gave (+)-muscarine (2S,4R,5S). A similar reaction sequence with 5b gave (+)-epimuscarine (7).[7]

Other Syntheses
It can be synthesized in various ways from completely different substances,[8][9][10][11][12][13][14][15][16][17] particularly from 2,5-dimethyl-3-carboxymethyl flurane.
Pharmacology
Pharmacodynamics
Muscarine mimics the action of the
Muscarinic receptors also signal via other pathways, for instance via G beta-gamma complex modulation of potassium channels. This allows muscarine to modulate cellular excitability via the membrane potential.
Metabolism
A paucity of research exists on the metabolism of muscarine in the human body, suggesting this compound is not metabolized by humans. Though there has been extensive research in the field of acetylcholine metabolism by acetylcholinesterase, muscarine is not metabolized by this enzyme, partly explaining the compound's potential toxicity. Muscarine is readily soluble in water. The most likely way for muscarine to leave the blood is via renal clearance; it will eventually leave the body in urine.[19]
Medical uses
This section needs more primary sources. (November 2019) |  |
Muscarinic agonists are used as drugs in treating
Efficacy
As muscarine works on the muscarinic acetylcholine receptor, the best comparison can be made with acetylcholine, which normally works on this receptor. Pure muscarine compared to pure acetylcholine is stated in most cases to be more potent, its action is always slower but longer lasting than acetylcholine. A possible explanation for this long-lasting behavior might be that muscarine does not get hydrolyzed by acetylcholinesterase in the synaptic cleft.[20]
Toxicology
Muscarine poisoning is characterized by
Antidote
to muscarine. Atropine is, like muscarine, an alkaloid but unlike muscarine is an antagonist of the muscarinic receptors. Hence, it inhibits the effects of acetylcholine. Muscarinic antagonists dilate the pupil and relax the ciliary muscle, are used in treatment of inflammatory uveitis and is associated with glaucoma. They are also used to treat urinary incontinence and diseases characterized by bowel hypermotility such as irritable bowel syndrome. Muscarinic antagonists are often calledReferences
- ^ S2CID 205902282.
- ^ Schmiedeberg, O.; Koppe, R. (1869). Das Muscarin, das giftige Alkaloid des Fliegenpilzes (Agaricus muscarius L.), seine Darstellung, chemischen Eigenschaften, physiologischen Wirkungen, toxicologische Bedeutung und sein Verhältniss zur Pilzvergiftung im allgemeinen [Muscarine, the poisonous alkaloid of the fly agaric (Agaricus muscarius L.), its preparation, chemical properties, physiological effects, toxicological importance, and its relation to mushroom poisoning in general]. Leipzig: Verlag von F.C.W. Vogel.
- ^ Pappano Achilles J, "Chapter 7. Cholinoceptor-Activating & Cholinesterase-Inhibiting Drugs" (Chapter). Katzung BG: Basic & Clinical Pharmacology, 11e Archived 2009-09-10 at the Wayback Machine
- .
- .
- .
- ^ doi:10.1139/v92-346.
- .
- S2CID 38142806.
- S2CID 28914321.
- .
- .
- .
- doi:10.1139/v72-532.
- .
- .
- .
- ISBN 0815124562.
- ISBN 978-1-143-46767-7,
- PMID 13413151.
- ^ Peter G. Waser; Chemistry and pharmacology of muscarine, muscarone and some related compounds; Pharmacology Department, University of Zurich, Switzerland 1961



