NAPQI
 | |
| Clinical data | |
|---|---|
| Other names | N-Acetyl-p-benzoquinone imine; N-Acetylimidoquinone |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
NAPQI, also known as NAPBQI or N-acetyl-p-benzoquinone imine, is a toxic byproduct produced during the xenobiotic metabolism of the analgesic paracetamol (acetaminophen).[1] It is normally produced only in small amounts, and then almost immediately detoxified in the liver.
However, under some conditions in which NAPQI is not effectively detoxified (usually in the case of
Metabolism
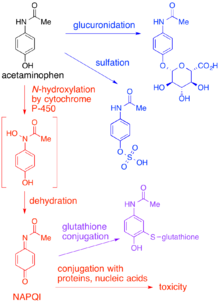
In adults, the primary metabolic pathway for paracetamol is
The minimum dosage at which paracetamol causes toxicity usually is 7.5 to 10g in the average person.[2] The lethal dose is usually between 10 g and 15 g.[citation needed] Concurrent alcohol intake lowers these thresholds significantly. Chronic alcoholics may be more susceptible to adverse effects due to reduced glutathione levels.[3] Other populations may experience effects at lower or higher dosages depending on differences in P-450 enzyme activity and other factors which affect the amount of NAPQI produced. In general, however, the primary concern is accidental or intentional paracetamol overdose.
When a toxic dose of paracetamol is ingested, the normal glucuronide pathway is saturated and large amounts of NAPQI are produced. Liver reserves of glutathione are depleted by conjugation with this excess NAPQI. The mechanism by which toxicity results is complex, but is believed to involve reaction between unconjugated NAPQI and critical proteins as well as increased susceptibility to oxidative stress caused by the depletion of glutathione.[4]
Poisoning
The prognosis is good for paracetamol overdoses if treatment is initiated up to 8 hours after the drug has been taken. Most hospitals stock the antidote (
Mechanism and antidote
NAPQI becomes toxic when GSH is depleted by an overdose of acetaminophen, Glutathione is an essential antidote to overdose. Glutathione conjugates to NAPQI and helps to detoxify it. In this capacity, it protects cellular protein thiol groups, which would otherwise become covalently modified; when all GSH has been spent, NAPQI begins to react with the cellular proteins, killing the cells in the process. The preferred treatment for an overdose of this painkiller is the administration of N-acetyl-L-cysteine (either via oral or IV administration)[5]), which is processed by cells to L-cysteine and used in the de novo synthesis of GSH.
See also
- Cytochrome P450 oxidase
- Liver failure
- Centrilobular necrosis
References
- ^ a b c d Mehta, S. (25 August 2012). "Metabolism of Paracetamol (Acetaminophen), Acetanilide and Phenacetin | Medicinal Chemistry | PharmaXChange.info". pharmaxchange.info. Archived from the original on 11 May 2022. Retrieved 29 August 2012.
- ^ "Acetaminophen Toxicity: Practice Essentials, Background, Pathophysiology". 5 October 2021.
- ^ "NIH Publications" (PDF). pubs.niaaa.nih.gov.
- PMID 20020268.
- ^ "Pharmaceutical Information – MUCOMYST". RxMed. Retrieved 2014-02-13.
External links
- Alsalim W, Fadel M (July 2003). "Towards evidence based emergency medicine: best BETs from the Manchester Royal Infirmary. Oral methionine compared with intravenous N-acetyl cysteine for paracetamol overdose". Emerg Med J. 20 (4): 366–7. PMID 12835357.
- van de Straat R, de Vries J, Debets AJ, Vermeulen NP (July 1987). "The mechanism of prevention of paracetamol-induced hepatotoxicity by 3,5-dialkyl substitution. The roles of glutathione depletion and oxidative stress". Biochem. Pharmacol. 36 (13): 2065–70. PMID 3606627.
- Cytochrome P-450 Inducers, Inhibitors, and Substrates
