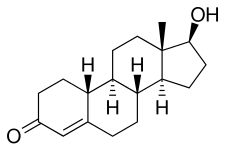
Nandrolone
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /ˈnændrəloʊn/[1] |
| Trade names | • Deca-Durabolin (as ND) • Durabolin (as NPP) • Many others (see here) |
| Other names | • 19-Nortestosterone[2][3] • 10-Nortestosterone • Estr-4-en-17β-ol-3-one • Estrenolone • Oestrenolone • 19-Norandrost-4-en-17β-ol-3-one • Norandrostenolone[2] • Nortestrionate[2] • Nortestonate[2] • Norandroone • SG-4341[2][3] |
| Pregnancy category |
|
| Drug class | Androgen; Anabolic steroid; Progestogen |
| ATC code | |
| Legal status | |
| Legal status |
|
Conjugates[9] | |
| Elimination half-life | • Nandrolone: <4.3 hours[7] • ND (IM): 6–12 days[7][8][10] • NPP: 2.7 days[10] |
| Duration of action | • ND (IM): 2–3 weeks[8][11] • NPP (IM): 5–7 days[8][10] |
| Excretion | Urine[7] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Nandrolone, also known as 19-nortestosterone, is an
Nandrolone esters were first described and introduced for medical use in the late 1950s.[8] They are among the most widely used anabolic steroid worldwide.[8] In addition to their medical use, nandrolone esters are used to improve physique and performance, and are said to be the most widely used anabolic steroid for such purposes.[8][17] The drugs are controlled substances in many countries and so non-medical use is generally illicit.[8]
Medical uses
Nandrolone esters are used clinically, although increasingly rarely, for people in catabolic states with major burns, cancer, and AIDS, and an ophthalmological formulation was available to support cornea healing.[18]: 134
The positive effects of nandrolone esters include muscle growth, appetite stimulation and increased red blood cell production,[medical citation needed] and bone density.[19] Clinical studies have shown them to be effective in treating anemia, osteoporosis, and breast cancer.
Nandrolone sulfate has been used in an eye drop formulation as an ophthalmic medication.[2][12]
Non-medical uses
Nandrolone esters are used for
Side effects
Nandrolone theoretically may produce
Pharmacology
Pharmacodynamics
| Medication | Ratioa |
|---|---|
| Testosterone | ~1:1 |
| Androstanolone (DHT) | ~1:1 |
| Methyltestosterone | ~1:1 |
| Methandriol | ~1:1 |
| Fluoxymesterone | 1:1–1:15 |
| Metandienone | 1:1–1:8 |
| Drostanolone | 1:3–1:4 |
| Metenolone | 1:2–1:30 |
| Oxymetholone | 1:2–1:9 |
| Oxandrolone | 1:3–1:13 |
| Stanozolol | 1:1–1:30 |
| Nandrolone | 1:3–1:16 |
| Ethylestrenol | 1:2–1:19 |
| Norethandrolone | 1:1–1:20 |
| Notes: In rodents. Footnotes: a = Ratio of androgenic to anabolic activity. Sources: See template. | |
Nandrolone is an
In addition to its AR agonistic activity, unlike many other anabolic steroids, nandrolone is also a potent
| Compound | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|
| Nandrolone | 20 | 154–155 | <0.1 | 0.5 | 1.6 | 1–16 | 0.1 |
| Testosterone | 1.0–1.2 | 100 | <0.1 | 0.17 | 0.9 | 19–82 | 3–8 |
| Estradiol | 2.6 | 7.9 | 100 | 0.6 | 0.13 | 8.7–12 | <0.1 |
| Notes: Values are percentages (%). Reference CBG . Sources: See template.
| |||||||
Anabolic and androgenic activity
Nandrolone has a very high ratio of anabolic to androgenic activity.[15] In fact, many nandrolone-like anabolic steroids and even nandrolone itself are said to have among the highest ratio of anabolic to androgenic effect of all anabolic steroids.[25] This is attributed to the fact that whereas testosterone is potentiated via conversion into dihydrotestosterone (DHT) in androgenic tissues, the opposite is true with nandrolone and similar anabolic steroids (i.e., other 19-nortestosterone derivatives).[15] As such, nandrolone-like anabolic steroids, namely nandrolone esters, are the most frequently used anabolic steroids in clinical settings in which anabolic effects are desired; for instance, in the treatment of AIDS-associated cachexia, severe burns, and chronic obstructive pulmonary disease.[25] However, anabolic steroids with a very high ratio of anabolic to androgenic action like nandrolone still have significant androgenic effects and can produce symptoms of masculinization like hirsutism and voice deepening in women and children with extended use.[15]
| Compound | rAR (%) | hAR (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| Testosterone | 38 | 38 | ||||||
| 5α-Dihydrotestosterone | 77 | 100 | ||||||
| Nandrolone | 75 | 92 | ||||||
| 5α-Dihydronandrolone | 35 | 50 | ||||||
| Ethylestrenol | ND | 2 | ||||||
| Norethandrolone | ND | 22 | ||||||
| 5α-Dihydronorethandrolone | ND | 14 | ||||||
| Metribolone | 100 | 110 | ||||||
| Sources: See template. | ||||||||
Pharmacokinetics
The
Nandrolone has very low
Single intramuscular injections of 100 mg nandrolone phenylpropionate or nandrolone decanoate have been found to produce an anabolic effect for 10 to 14 days and 20 to 25 days, respectively.[37] Conversely, unesterified nandrolone has been used by intramuscular injection once daily.[20][33]
-
Nandrolone levels after a single 50, 100, or 150 mg intramuscular injection of nandrolone decanoate in oil solution in men.[38]
-
Nandrolone levels after a single 100 mg intramuscular injection of
-
Nandrolone levels with a single 50 mg intramuscular injection of nandrolone decanoate or nandrolone hexyloxyphenylpropionate in oil solution in men.[40]
-
Dose-normalized nandrolone exposure (serum level divided by dose administered) with nandrolone decanoate in oil solution by intramuscular or subcutaneous injection in men.[41][42]
Chemistry

Nandrolone, also known as 19-nortestosterone (19-NT) or as estrenolone, as well as estra-4-en-17β-ol-3-one or 19-norandrost-4-en-17β-ol-3-one,
Derivatives
Esters
A variety of esters of nandrolone have been marketed and used medically.[2][12] The most commonly used esters are nandrolone decanoate and to a lesser extent nandrolone phenylpropionate. Examples of other nandrolone esters that have been marketed and used medically include nandrolone cyclohexylpropionate, nandrolone cypionate, nandrolone hexyloxyphenylpropionate, nandrolone laurate, nandrolone sulfate, and nandrolone undecanoate.[2][12][8]
Anabolic steroids
Nandrolone is the parent compound of a large group of anabolic steroids. Notable examples include the non-17α-alkylated
|
|
Progestins
Nandrolone, together with
Synthesis
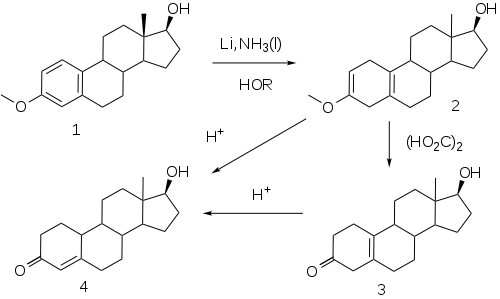
The elaboration of a method for the reduction of aromatic rings to the corresponding dihydrobenzenes under controlled conditions by A. J. Birch opened a convenient route to compounds related to the putative
This reaction, now known as the
Esters
- Treatment of 4 with decanoic anhydride and pyridine affords nandrolone decanoate.[52]
- Acylation of 4 with phenylpropionyl chloride yields nandrolone phenpropionate.[53]
Detection in body fluids
Nandrolone use is directly detectable in hair or indirectly detectable in urine by testing for the presence of
A number of nandrolone cases in
Heavy consumption of the essential amino acid
Mass spectrometry is also used to detect small amounts of nandrolone in urine samples.[63]
History

Nandrolone was first synthesized in 1950.[2][43][18]: 130 [64] It was first introduced, as nandrolone phenylpropionate, in 1959, and then as nandrolone decanoate in 1962, followed by additional esters.[65]
Society and culture
Generic names
Nandrolone is the
Doping in sports
Nandrolone was probably among the first anabolic steroids to be used as a doping agent in sports in the 1960s.[
Research
Nandrolone esters have been studied in several indications. They were intensively studied for
: 134References
- ^ "Nandrolone Meaning in the Cambridge English Dictionary".
- ^ OCLC 1079003025.
- ^ ISBN 978-94-011-4439-1.
- ^ Anvisa (2023-03-31). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-04-04). Archived from the original on 2023-08-03. Retrieved 2023-08-15.
- ^ PMID 10435281.
- ISBN 978-0-7817-1750-2.
- ^ a b c d e f g "Deca-Durabolin" (PDF). Schering-Plough. Archived from the original (PDF) on 18 December 2010.
- ^ ISBN 978-0-9828280-1-4.
- ^ ISBN 978-1-4684-5499-4.
- ^ PMID 9103484.
- ^ "Deca-Durabolin" (PDF). Merck Sharp & Dohme (Australia).
- ^ ISBN 978-3-88763-075-1.
- ^ ISBN 978-0-471-89979-2.
- PMID 24684468.
- ^ PMID 18500378.
- ISBN 978-3-642-66353-6.
- ISBN 978-0-323-32195-2.
- ^ ISBN 978-3-540-79088-4.
- PMID 25905160– via NCBI Bookshelf.
Both testosterone and its non-aromatizable derivative nandrolone, produce increased bone density in men with glucocorticoid-induced osteoporosis with minimal short-term side-effects....
- ^ )
- ^ PMID 27141449.
- ^ PMID 4021486.
- ISBN 978-3527600908.
- ^ S2CID 24616324.
- ^ S2CID 29035729.
- PMID 8954018.
- ^ Mauvais-Jarvis, P. "Progesterone and progestins: a general overview." (1983): 1-16.
- ^ PMID 817322.
- ISSN 0021-972X.
- ^ Furman RH, Howard RP, Smith CW, Norcia LN (January 1956). "Comparison of the effects of oral methyltestosterone, 19-nortestosterone, and 17-methyl-19-nortestosterone on serum lipids and lipoproteins". Journal of Laboratory and Clinical Medicine. 48 (5): 808–809.
- PMID 13498038.
- ^ Arnold A, Potts GO (January 1964). "Oral anabolic and androgenic effects of 19-nortestosterone and 17α-methyl-19-nortestosterone". Federation Proceedings. 23 (2): 412.
- ^ ISSN 0021-972X.
- PMID 6539197.
- ^ PMID 3865479.
- ISBN 978-1-135-25825-2.
- ISBN 978-1-4832-7300-6.
- PMID 15713722.
- PMID 9103484.
- PMID 4010287.
- PMID 26690043.
- ^ Kalicharan, Raween Wikesh (2017). New Insights into Drug Absorption from Oil Depots (PhD). Utrecht University.
- ^ ISBN 978-0-323-14611-1.
- S2CID 6472015.
- S2CID 3862299.
- ^ PMID 14670641.
- ISBN 978-1-59259-375-0.
Estranes. Estrane and gonane progestogens are derived from 19-nortestosterone, the progestogenic parent compound used in oral contraceptives in the United States. Estranes are characterized by the presence of an ethinyl group at position 17 and by the absence of a methyl group between the A and B rings (see Fig. 10). The estrane progestogens that are related structurally to norethindrone (norethynodrel, lynestrenol, norethindrone acetate, ethynodiol diacetate) are converted to this parent compound. Norethindrone is the second most commonly used progestogen in the United States for HRT. Gonanes. The gonanes share the structural modifications found in the estranes and also possess an ethinyl group at position 13 and a keto group at position 3 (see Fig. 11). Norgestrel was synthesized in 1963 and is a racemic mixture of dextro and levorotatory forms. The levorotatory form, levonorgestrel, provides the biologic activity. Third-generation gonanes (desogestrel, gestodene, and norgestimate) have been developed to reduce unwanted side effects of progestogens, [...]
- .
- .
- .
- .
- ^ US 2998423, DeWytt ED, Overbeek O, Overbeek GA, issued 1961, assigned to Organon.
- ^ CH 206119, issued 1939, assigned to Gesellschaft für Chemische Industrie Basel
- ^ "Clarification about Nandrolone Testing". World Anti-Doping Agency. 2005-05-30. Archived from the original on 2012-09-15. Retrieved 2012-01-31.
- PMID 16597518.
- ^ Ueki M, Ikekita A, Takao Y (2000). "[Nandrolone metabolite in urine of Nagano Olympic athlete]". Jap. J. For. Tox. (in Japanese). 18: 198–199.
- PMID 11086369.
- ISBN 978-0-9626523-7-0. Archived from the originalon 2020-12-04. Retrieved 2010-08-19.
- ^ Baron P (19 September 2000). "The Drugs Issue: Baumann to fight all the way". The Telegraph. Retrieved 2010-11-13.
- ^ Richardson M (2004-02-19). "Athletics: Mark Richardson explains how he felt at his disciplinary hearing". The Guardian. Retrieved 2010-11-13.
- ^ "Track Star Marion Jones to Admit Steroid Use". National Public Radio. 2007-10-05. Retrieved 2009-11-09.
- ^ UK Sport Anti-doping Directorate (January 2000). Nandrolone Review (PDF) (Report). UK Sport. Archived from the original (PDF) on 2005-04-04. Retrieved 2013-02-02.
- PMID 20533318.
- ISSN 0368-1769.
- ]
- ^ a b "Nandrolone".
Further reading
- Geusens P (September 1995). "Nandrolone decanoate: pharmacological properties and therapeutic use in osteoporosis". Clinical Rheumatology. 14 (Suppl 3): 32–9. S2CID 24365057.
- Kicman AT (June 2008). "Pharmacology of anabolic steroids". British Journal of Pharmacology. 154 (3): 502–21. PMID 18500378.
- Hemmersbach P, Grosse J (2010). "Nandrolone: A Multi-Faceted Doping Agent". Doping in Sports. Handbook of Experimental Pharmacology. Vol. 195. pp. 127–54. PMID 20020363.
- Velema MS, Kwa BH, de Ronde W (March 2012). "Should androgenic anabolic steroids be considered in the treatment regime of selected chronic obstructive pulmonary disease patients?". Current Opinion in Pulmonary Medicine. 18 (2): 118–24. S2CID 6155275.
- Busardò FP, Frati P, Sanzo MD, Napoletano S, Pinchi E, Zaami S, Fineschi V (January 2015). "The impact of nandrolone decanoate on the central nervous system". Current Neuropharmacology. 13 (1): 122–31. PMID 26074747.
- Wu C, Kovac JR (October 2016). "Novel Uses for the Anabolic Androgenic Steroids Nandrolone and Oxandrolone in the Management of Male Health". Current Urology Reports. 17 (10): 72. S2CID 43199715.
- Pan MM, Kovac JR (April 2016). "Beyond testosterone cypionate: evidence behind the use of nandrolone in male health and wellness". Translational Andrology and Urology. 5 (2): 213–9. PMID 27141449.

![Nandrolone levels after a single 50, 100, or 150 mg intramuscular injection of nandrolone decanoate in oil solution in men.[38]](http://upload.wikimedia.org/wikipedia/commons/thumb/0/03/Nandrolone_levels_after_a_single_intramuscular_injection_of_different_doses_of_nandrolone_decanoate.png/298px-Nandrolone_levels_after_a_single_intramuscular_injection_of_different_doses_of_nandrolone_decanoate.png)
![Nandrolone levels after a single 100 mg intramuscular injection of nandrolone decanoate or nandrolone phenylpropionate in 4 mL or 1 mL arachis oil solution into gluteal or deltoid muscle in men.[39]](http://upload.wikimedia.org/wikipedia/commons/thumb/d/da/Nandrolone_levels_after_a_single_100_mg_intramuscular_injection_of_nandrolone_esters.png/300px-Nandrolone_levels_after_a_single_100_mg_intramuscular_injection_of_nandrolone_esters.png)
![Nandrolone levels with a single 50 mg intramuscular injection of nandrolone decanoate or nandrolone hexyloxyphenylpropionate in oil solution in men.[40]](http://upload.wikimedia.org/wikipedia/commons/thumb/e/e8/Nandrolone_levels_with_a_single_dose_of_nandrolone_decanoate_or_nandrolone_hexyloxyphenylpropionate_by_intramuscular_injection_in_men.png/300px-Nandrolone_levels_with_a_single_dose_of_nandrolone_decanoate_or_nandrolone_hexyloxyphenylpropionate_by_intramuscular_injection_in_men.png)
![Dose-normalized nandrolone exposure (serum level divided by dose administered) with nandrolone decanoate in oil solution by intramuscular or subcutaneous injection in men.[41][42]](http://upload.wikimedia.org/wikipedia/commons/thumb/5/50/Dose-normalized_nandrolone_exposure_with_nandrolone_decanoate_by_intramuscular_or_subcutaneous_injection_in_men.png/300px-Dose-normalized_nandrolone_exposure_with_nandrolone_decanoate_by_intramuscular_or_subcutaneous_injection_in_men.png)