Naringenin
| |

| |
| Names | |
|---|---|
| IUPAC name
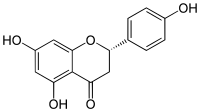
(2S)-4′,5,7-Trihydroxyflavan-4-one
| |
| Systematic IUPAC name
(2S)-5,7-Dihydroxy-2-(4-hydroxyphenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Naringetol; Salipurol; Salipurpol
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.006.865 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H12O5 | |
| Molar mass | 272.256 g·mol−1 |
| Melting point | 251 °C (484 °F; 524 K)[1] |
| 475 mg/L[citation needed] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Naringenin is a flavanone from the flavonoid group of polyphenols. It is commonly found in a variety of citrus fruits and is the predominant flavonone in grapefruit.[2] Naringenin has demonstrated numerous biological activities, including anti-inflammatory properties, antioxidant activity and skin healing.[3][4][5][6] It is used as a cosmetic ingredient and dietary supplement.[7] Naringenin (along with furanocoumarins presented in citrus fruits) is thought to be responsible for CYP3A4 suppression in the intestinal wall, that may result in serious changes of pharmacokinetics in drugs related with this enzyme.[8][9][10]
Structure
Naringenin has the skeleton structure of a flavanone with three hydroxy groups at the 4′, 5, and 7 carbons. It may be found both in the aglycol form, naringenin, or in its glycosidic form, naringin, which has the addition of the disaccharide neohesperidose attached via a glycosidic linkage at carbon 7.
Like the majority of flavanones, naringenin has a single chiral center at carbon 2, although the optical purity is variable.[11][12] Racemization of (S)-(−)-naringenin has been shown to occur fairly quickly.[13]
Sources and bioavailability
Naringenin and its glycoside has been found in a variety of
The
Grapefruit juice can provide much higher plasma concentrations of naringenin than orange juice.[24] Also found in grapefruit is the related compound kaempferol, which has a hydroxyl group next to the ketone group.
Naringenin can be absorbed from cooked tomato paste. There are 3.8 mg of naringenin in 150 grams of tomato paste.[25]
Biosynthesis and metabolism
Naringenin can be produced from naringin by the hydrolytic action of the liver enzyme naringinase.[26]
Naringenin is derived from malonyl-CoA and 4-coumaroyl-CoA. The latter is derived from phenylalanine. The resulting tetraketide is acted on by chalcone synthase to give the chalcone that then undergoes ring-closure to naringenin.[27]
The enzyme
Potential biological effects
This section needs more primary sources. (June 2017) |  |
Studies show naringenin has numerous biological activities, including anti-inflammatory, antioxidant, antibacterial, antiviral and anticancer. It is deemed safe for both topical and ingestible use in healthy adults.[7]
Anti-inflammatory
Naringenin’s anti-inflammatory benefits have been well established with multiple in vitro and in vivo studies, revealing it effectively suppresses proinflammatory factors, cytokine and chemokine expressions in inflammation.[3] When taken orally, it has also been shown to reduce inflammatory pain.[29]
Antioxidant
Naringenin has been shown to have significant antioxidant properties.[30][31] It has been shown to reduce oxidative damage to DNA in vitro and in animal studies.[32][33] When consumed it has been shown to increase antioxidant markers superoxide dismutase and glutathione.[6]
Endothelium protection
Some human studies and many animal studies have shown the ability of both naringenin and naringin to protect and improve the health of the vascular endothelium.[26] Naringenin reportedly stimulates Nrf2 to protect blood vessels.[34]
Antibacterial, antifungal, and antiviral
Naringenin has an antimicrobial effect on
Naringenin has also been shown to reduce hepatitis C virus production by infected hepatocytes (liver cells) in cell culture. This seems to be secondary to naringenin's ability to inhibit the secretion of very-low-density lipoprotein by the cells.[40] The antiviral effects of naringenin are currently under clinical investigation.[41] Reports of antiviral effects on polioviruses, HSV-1 and HSV-2 have also been made, though replication of the viruses has not been inhibited.[42][43] In in vitro experiments naringenin also showed a strong antiviral activity against SARS-CoV-2. [44]
Skin Healing
When used in topical formulations, naringenin has been shown to be an anti-inflammatory with skin barrier restoration and antioxidant activities.
UVB radiation is very high energy frequency and wreak havoc on the top layers of the skin by damaging the skin cells and causing DNA mutations that can lead to melanoma and other skin cancers.[45] Naringenin has been proven to reduce UVB-induced skin damage, as well as showing efficacy against oxidative stress and improvement in wound healing.[4][46][6]
Naringenin has also been studied on inflammatory skin conditions such as atopic dermatitis and psoriasis.[3][47][48] For atopic dermatitis, naringenin was found to sharply suppress inflammatory levels and alleviate symptoms and may suppress the development of atopic dermatitis like skin lesions.[3][47] In psoriasis, naringenin has been studied to reduce inflammation in psoriatic plaques.[48]
Anticancer
Fatty Liver Disease
Naringenin may have some benefits for non-alcoholic fatty liver disease. It was proven to reduce hepatic lipid accumulation and inflammation in the livers of mice with non-alcoholic fatty liver disease.[55]
Alzheimer's disease
Naringenin is being researched as a potential treatment for Alzheimer's disease. Naringenin has been demonstrated to improve memory and reduce amyloid and tau proteins in a study using a mouse model of Alzheimer's disease.[56][57] The effect is believed to be due to a protein present in neurons known as CRMP2 that naringenin binds to.[58]
Safety
Naringenin has been deemed safe to apply topically and can also be ingested safely by healthy adults in doses of 150 to 900 mg, with 300 mg of naringenin twice a day likely to elicit physiological effect.[7]
References
- ^ Naringenin at the Human Metabolome Database
- S2CID 27540043.
- ^ PMID 26836240.
- ^ PMID 26741806.
- ^ PMID 10552377.
- ^ PMID 28415935.
- ^ PMID 31468636.
- PMID 12611197.
- PMID 10511919.
- PMID 9565737.
- PMID 17113835.
- ^ S2CID 24051610.
- S2CID 95215634.
- PMID 10812937.
- PMID 17960080.
- S2CID 40261445.
- ^ S2CID 26333030.
- PMID 22957841.
- PMID 12526004.
- PMID 14616013.
- PMID 18372132.
- PMID 1421508.
- PMID 10558881.
- PMID 11160539.
- PMID 12421849.
- ^ PMID 37484296.
- PMID 28168876.
- PMID 10680173.
- S2CID 35925963.
- PMID 15826081.
- PMID 15769128.
- PMID 26881453.
- PMID 15313158.
- PMID 35024180.
- PMID 10857921.
- S2CID 2043029.
- PMID 17475517.
- PMID 15881836.
- S2CID 260284591.
- PMID 18393287.
- ^ "A Pilot Study of the Grapefruit Flavonoid Naringenin for HCV Infection". clinicaltrials.gov. Archived from the original on 2010-10-01.
- S2CID 6174141.
- S2CID 34495208.
- PMID 33096221.
- ^ Alexander H. "What's the difference between UVA and UVB rays?". MD Anderson Cancer Center. Retrieved 2024-01-02.
- S2CID 211234647.
- ^ PMID 23933131.
- ^ S2CID 214694347.
- PMID 15744083.
- PMID 33603536.
- PMID 8875554.
- PMID 17482226.
- S2CID 24436665.
- PMID 29570361.
- .
- PMID 26148826.
- S2CID 73449033.
- PMID 28674493.
Further reading
- inhibitory effect on the human PMID 10619367.
- Wistuba D, Trapp O, Gel-Moreto N, Galensa R, Schurig V (May 2006). "Stereoisomeric separation of flavanones and flavanone-7-O-glycosides by capillary electrophoresis and determination of interconversion barriers". Analytical Chemistry. 78 (10): 3424–3433. PMID 16689546.
- Krause M, Galensa R (1991). "High-performance liquid chromatography of diastereomeric flavanone glycosides in Citrus on a β-cyclodextrin-bonded stationary phase (Cyclobond I)". Journal of Chromatography A. 588 (1–2): 41–45. .
- Gaggeri R, Rossi D, Collina S, Mannucci B, Baierl M, Juza M (August 2011). "Quick development of an analytical enantioselective high performance liquid chromatography separation and preparative scale-up for the flavonoid Naringenin". Journal of Chromatography A. 1218 (32): 5414–5422. PMID 21397238.
- Wan L, Sun X, Wang X, Li Y, Yu Q, Guo C (April 2011). "A stereospecific HPLC method and its application in determination of pharmacokinetics profile of two enantiomers of naringenin in rats". Journal of Chromatographic Science. 49 (4): 316–320. PMID 21439124.
- Naringenin also produces S2CID 24620048.
- Gao K, Henning SM, Niu Y, Youssefian AA, Seeram NP, Xu A, Heber D (February 2006). "The citrus flavonoid naringenin stimulates DNA repair in prostate cancer cells". The Journal of Nutritional Biochemistry. 17 (2): 89–95. PMID 16111881.
- Katavic PL, Lamb K, Navarro H, Prisinzano TE (August 2007). "Flavonoids as opioid receptor ligands: identification and preliminary structure-activity relationships". Journal of Natural Products. 70 (8): 1278–1282. PMID 17685652.
- Naringenin has been reported to induce apoptosis in PMID 16756346.
- Naringenin seems to protect PMID 19592617.
- Naringenin lowers the plasma and hepatic S2CID 5685548.
- Naringenin has been demonstrated to improve memory and reduce amyloid and tau proteins in a study using a mouse model of Alzheimer's disease.Ghofrani S, Joghataei MT, Mohseni S, Baluchnejadmojarad T, Bagheri M, Khamse S, Roghani M (October 2015). "Naringenin improves learning and memory in an Alzheimer's disease rat model: Insights into the underlying mechanisms". European Journal of Pharmacology. 764: 195–201. PMID 28674493.
