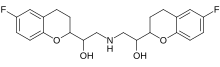
Nebivolol
 | ||
 | ||
| Clinical data | ||
|---|---|---|
| Trade names | Nebilet, Bystolic, others | |
| Other names | Narbivolol, Nebivolol, Nebivololum[1] | |
| AHFS/Drugs.com | Monograph | |
| MedlinePlus | a608029 | |
| License data | ||
| Pregnancy category |
| |
| Routes of administration | By mouth | |
| ATC code | ||
| Legal status | ||
| Legal status | ||
JSmol) | ||
| ||
| ||
| | ||

Nebivolol is a beta blocker used to treat high blood pressure and heart failure.[5] As with other β-blockers, it is generally a less preferred treatment for high blood pressure.[6] It may be used by itself or with other blood pressure medication.[6] It is taken by mouth.[6]
Common side effects include dizziness, feeling tired, nausea, and headaches.
Nebivolol was patented in 1983 and came into medical use in 1997.[9] It is available as a generic medication in the United Kingdom.[5] In 2021, it was the 244th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[10][11]
Medical uses
It is used to treat high blood pressure and heart failure.[5] Nebivolol is used in the treatment of angina, to decrease the heart rate and contractile force. This is relevant in patients who need to decrease the oxygen demand of the heart so that the blood supplied from stenosed or constricted arteries is adequate.
Pharmacology and biochemistry
β1-selectivity
Beta blockers help patients with cardiovascular disease by blocking β1 receptors, while many of the side-effects of these medications are caused by their blockade of β2 receptors.[16] For this reason, beta blockers that selectively block β1 adrenergic receptors (termed cardioselective or β1-selective beta blockers) produce fewer adverse effects (for instance, bronchoconstriction) than those drugs that non-selectively block both β1 and β2 receptors.
In a laboratory experiment conducted on biopsied heart tissue, nebivolol proved to be the most β1-selective of the
Nebivolol
Due to enzymatic inhibition, fluvoxamine increases the exposure to nebivolol and its active hydroxylated metabolite (4-OH-nebivolol) in healthy volunteers.[24]
Vasodilator action
Nebivolol is unique as a beta-blocker.[25] Unlike carvedilol, it has a nitric oxide (NO)-potentiating, vasodilatory effect via stimulation of β3 receptors.[26][27][28]
Nebivolol induces vasodilation by stimulating the production of nitric oxide, a natural blood vessel relaxant. This effect is achieved by activating the endothelial isoform of
Along with labetalol, celiprolol and carvedilol, it is one of four beta blockers to cause dilation of blood vessels in addition to effects on the heart.[28]
Antihypertensive effect
Nebivolol lowers blood pressure (BP) by reducing
Pharmacokinetics
Nebivolol plasma protein binding is approximately 98%, mostly to albumin and its half-life of low doses is 12 hours in extensive CYP2D6 metabolizers and 19 hours in poor metabolizers.[33]
Contraindications
- Severe bradycardia
- Heart block greater than first degree
- Patients with cardiogenic shock
- Decompensated cardiac failure
- Sick sinus syndrome (unless a permanent pacemaker is in place)
- Patients with severe hepatic impairment (Child-Pugh class B)
- Patients who are hypersensitive to any component of this product.
Side effects
Side effects might include headache, tiredness, dizziness, lightheadedness, reduced blood flow to extremities, bradycardia.[34]
Controversies
Pharmacology of side-effects
Several studies have suggested that nebivolol has reduced typical beta-blocker-related side effects, such as
Bystolic is associated with a number of serious risks. Bystolic is contraindicated in patients with severe bradycardia, heart block greater than first degree, cardiogenic shock, decompensated cardiac failure, sick sinus syndrome (unless a permanent pacemaker is in place), severe hepatic impairment (Child-Pugh > B) and in patients who are hypersensitive to any component of the product. Bystolic therapy is also associated with warnings regarding abrupt cessation of therapy, cardiac failure, angina and acute myocardial infarction, bronchospastic diseases, anesthesia and major surgery, diabetes and hypoglycemia, thyrotoxicosis, peripheral vascular disease, non-dihydropyridine calcium channel blockers use, as well as precautions regarding use with CYP2D6 inhibitors, impaired renal and hepatic function, and anaphylactic reactions. Finally, Bystolic is associated with other risks as described in the Adverse Reactions section of its PI. For example, a number of treatment-emergent adverse events with an incidence greater than or equal to 1 percent in Bystolic-treated patients and at a higher frequency than placebo-treated patients were identified in clinical studies, including headache, fatigue, and dizziness.
FDA warning letter about advertising claims
In August 2008, the FDA issued a
History
Mylan Laboratories licensed the US and Canadian rights to nebivolol from
In India, nebivolol is available as Nebula (Zydus Healthcare Ltd), Nebizok (Eris life-sciences), Nebicip (Cipla ltd), Nebilong (Micro Labs), Nebistar (Lupin ltd), Nebicard (Torrent), Nubeta (
In Greece and Italy, nebivolol is marketed by Menarini as Lobivon.
In Germany it is marketed as Nebilet by Berlin Chemie.
In the Middle East, Russia and Australia, it is marketed under the name Nebilet and in Pakistan it is marketed by The Searle Company Limited as Byscard.
References
- ^ "Nebivolol". go.drugbank.com. Retrieved 17 August 2022.
- ^ "Drugs@FDA: FDA-Approved Drugs Nebivolol - ALLERGAN" (PDF). accessdata.fda.gov. Retrieved 17 August 2022.
- PMID 28509722.
- ^ "DrugBank Nebivolol". go.drugbank.com. Retrieved 17 August 2022.
- ^ ISBN 9780857113382.
- ^ a b c d e f "Nebivolol Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 3 March 2019.
- ^ "Nebivolol Pregnancy and Breastfeeding Warnings". Drugs.com. Retrieved 3 March 2019.
- S2CID 24186687.
- ISBN 9783527607495.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Nebivolol - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- S2CID 34462045.
- PMID 22480336.
- S2CID 220732879.
- )
- S2CID 32202416.
- PMID 12535855.
- ^ a b c d "Prescribing information for Bystolic". Forest Laboratories, Inc. Archived from the original (PDF) on 25 March 2016. Retrieved 11 June 2009.
- S2CID 58760796.
- PMID 19389564.
- PMID 16457637.
- PMID 15935402.
- ISBN 0-443-07145-4.
- PMID 28459657.
- S2CID 25807936.
- PMID 26896479.
- PMID 17326335.
- ^ S2CID 205598744.
- PMID 19443516.
- PMID 12888152.
- PMID 16961165.
- S2CID 12785427.
- ^ "RxAbbVie - BYSTOLIC® (nebivolol) tablets, for oral use Full Prescribing Information" (PDF). www.rxabbvie.com. January 2019. Retrieved 17 August 2022.
- ^ "Nebivolol Oral: Uses, Side Effects, Interactions, Pictures, Warnings & Dosing - WebMD". www.webmd.com. Retrieved 17 August 2022.
- S2CID 20786564.
- PMID 16373195.
- S2CID 35105142.
- ^ a b Thomas Abrams (28 August 2008). "Warning Letter" (PDF). Food and Drug Administration. Archived from the original (PDF) on 28 April 2017. Retrieved 16 December 2019.
FDA is not aware of any substantial evidence or substantial clinical experience that demonstrates that Bystolic represents a 'novel' or 'next generation' beta blocker for the treatment of hypertension. Indeed, we are not aware of any well-designed trials comparing Bystolic to other β-blockers. Furthermore, FDA is not aware of any data that would render Bystolic's mechanism of action 'unique.'
