Neighbouring group participation
In
NGP by heteroatom lone pairs
In this type of substitution reaction, one group of the substrate participates initially in the reaction and thereby affects the reaction. Due to NGP, the reaction rate gets increased by many folds. [clarification needed] A classic example of NGP is the reaction of a
]
Ph−S−CH2−CH2−Cl reacts with water 650 times faster than CH3−CH2−CH2−Cl. [citation needed]
NGP by an alkene
The π orbitals of an

The carbocationic intermediate will be stabilized by
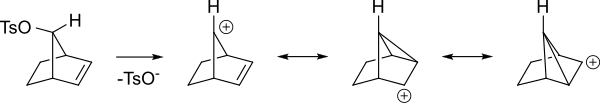
Here is a different view of the same intermediates.

Even if the alkene is more remote from the reacting center the alkene can still act in this way. For instance in the following
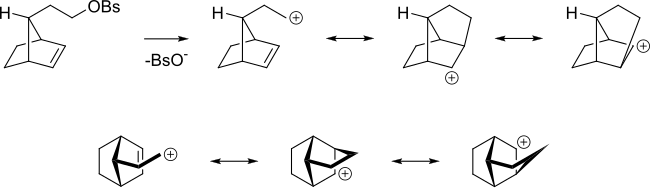
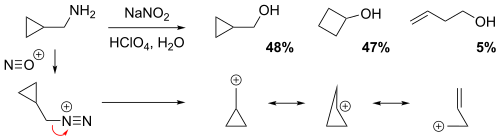
NGP by a cyclopropane, cyclobutane or a homoallyl group
The reaction of cyclopropylmethamine with sodium nitrite in dilute aqueous perchloric acid solution yielded a mixture of 48% cyclopropylmethyl alcohol, 47% cyclobutanol, and 5% homoallylic alcohol (but-3-en-1-ol).[4] In the non-classical perspective, the positive charge is delocalized throughout the carbocation intermediate structure via resonance, resulting in partial (electron-deficient) bonds. Evidently, the relatively low yield of the homoallylic alcohol implies that the homoallylic structure is the weakest resonance contributor.
NGP by an aromatic ring
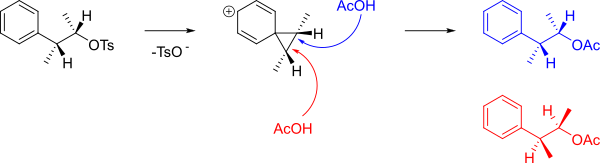
An

When the following
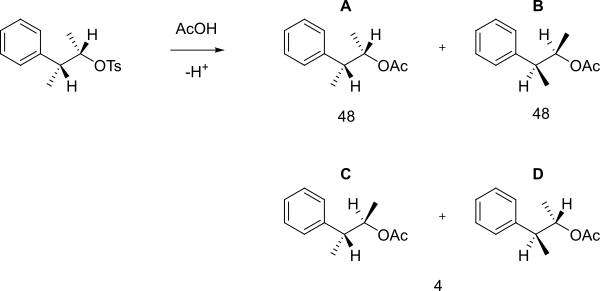
The mechanism which forms A and B is shown below.
NGP by aliphatic C-C or C-H bonds
Aliphatic C-C or C-H bonds can lead to charge delocalization if these bonds are close and antiperiplanar to the leaving group. Corresponding intermediates are referred to a nonclassical ions, with the 2-norbornyl system as the most well known case.
External links
References
- PMID 461188.
- PMID 19907773.
- ISBN 9780120335282. Retrieved 2019-12-17.
- .
