Development of the nervous system
| This article is part of a series on the |
| Development of organ systems |
|---|
|
The development of the nervous system, or neural development (neurodevelopment), refers to the processes that generate, shape, and reshape the
Defects in neural development can lead to malformations such as holoprosencephaly, and a wide variety of neurological disorders including limb paresis and paralysis, balance and vision disorders, and seizures,[1] and in humans other disorders such as Rett syndrome, Down syndrome and intellectual disability.[2]
Overview of vertebrate brain development
The
Aspects
Some landmarks of neural development include the birth and
Typically, these neurodevelopmental processes can be broadly divided into two classes: activity-independent mechanisms and
Developmental neuroscience uses a variety of
Neural induction
During early
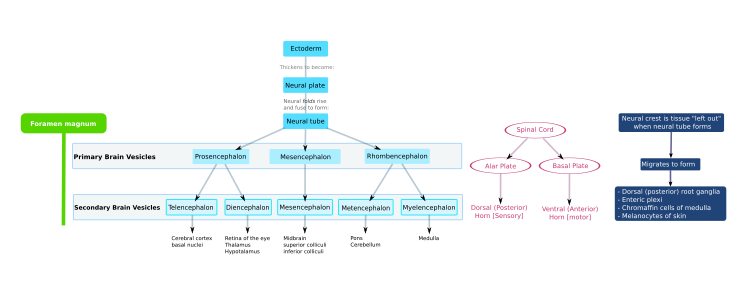
In the early embryo, the neural plate folds outwards to form the
A transplanted blastopore lip can convert ectoderm into neural tissue and is said to have an inductive effect. Neural inducers are molecules that can induce the expression of neural genes in ectoderm explants without inducing mesodermal genes as well. Neural induction is often studied in Xenopus embryos since they have a simple body plan and there are good markers to distinguish between neural and non-neural tissue. Examples of neural inducers are the molecules noggin and chordin.
When embryonic ectodermal cells are cultured at low density in the absence of mesodermal cells they undergo neural differentiation (express neural genes), suggesting that neural differentiation is the default fate of ectodermal cells. In
Regionalization
In a later stage of development the superior part of the neural tube flexes at the level of the future midbrain—the
The alar plate of the prosencephalon expands to form the
Patterning of the nervous system
In
Dorsoventral axis
The ventral half of the neural plate is controlled by the notochord, which acts as the 'organiser'. The dorsal half is controlled by the ectoderm plate, which flanks either side of the neural plate.[17]
Ectoderm follows a default pathway to become neural tissue. Evidence for this comes from single, cultured cells of ectoderm, which go on to form neural tissue. This is postulated to be because of a lack of BMPs, which are blocked by the organiser. The organiser may produce molecules such as follistatin, noggin and chordin that inhibit BMPs.
The ventral neural tube is patterned by
In this context Shh acts as a morphogen - it induces cell differentiation dependent on its concentration. At low concentrations it forms ventral interneurons, at higher concentrations it induces motor neuron development, and at highest concentrations it induces floor plate differentiation. Failure of Shh-modulated differentiation causes holoprosencephaly.
The dorsal neural tube is patterned by BMPs from the epidermal ectoderm flanking the neural plate. These induce sensory interneurons by activating
Rostrocaudal (Anteroposterior) axis
Signals that control anteroposterior neural development include
Neurogenesis
Neuronal migration
Neuronal

Radial migration
Neuronal precursor cells proliferate in the
Subsequent waves of neurons split the preplate by migrating along
Tangential migration
Most interneurons migrate tangentially through multiple modes of migration to reach their appropriate location in the cortex. An example of tangential migration is the movement of interneurons from the ganglionic eminence to the cerebral cortex. One example of ongoing tangential migration in a mature organism, observed in some animals, is the rostral migratory stream connecting subventricular zone and olfactory bulb.
Axophilic migration
Many neurons migrating along the anterior-posterior axis of the body use existing
Multipolar migration
There is also a method of neuronal migration called multipolar migration.
Neurotrophic factors
The survival of neurons is regulated by survival factors, called trophic factors. The neurotrophic hypothesis was formulated by Victor Hamburger and
- Nerve Growth Factor(NGF): Rita Levi Montalcini and Stanley Cohen purified the first trophic factor, Nerve Growth Factor (NGF), for which they received the Nobel Prize. There are three NGF-related trophic factors: BDNF, NT3, and NT4, which regulate survival of various neuronal populations. The Trk proteins act as receptors for NGF and related factors. Trk is a receptor tyrosine kinase. Trk dimerization and phosphorylation leads to activation of various intracellular signaling pathways including the MAP kinase, Akt, and PKC pathways.
- CNTF: Ciliary neurotrophic factor is another protein that acts as a survival factor for motor neurons. CNTF acts via a receptor complex that includes CNTFRα, GP130, and LIFRβ. Activation of the receptor leads to phosphorylation and recruitment of the JAK kinase, which in turn phosphorylates LIFRβ. LIFRβ acts as a docking site for the STAT transcription factors. JAK kinase phosphorylates STAT proteins, which dissociate from the receptor and translocate to the nucleus to regulate gene expression.
- GDNF: Glial derived neurotrophic factor is a member of the TGFbfamily of proteins, and is a potent trophic factor for striatal neurons. The functional receptor is a heterodimer, composed of type 1 and type 2 receptors. Activation of the type 1 receptor leads to phosphorylation of Smad proteins, which translocate to the nucleus to activate gene expression.
Synapse formation
Neuromuscular junction
Much of our understanding of synapse formation comes from studies at the neuromuscular junction. The transmitter at this synapse is acetylcholine. The acetylcholine receptor (AchR) is present at the surface of muscle cells before synapse formation. The arrival of the nerve induces clustering of the receptors at the synapse. McMahan and Sanes showed that the synaptogenic signal is concentrated at the
In the mature synapse each muscle fiber is innervated by one motor neuron. However, during development, many of the fibers are innervated by multiple axons. Lichtman and colleagues have studied the process of synapses elimination.[38] This is an activity-dependent event. Partial blockage of the receptor leads to retraction of corresponding presynaptic terminals. Later they used a connectomic approach, i.e., tracing out all the connections between motor neurons and muscle fibers, to characterize developmental synapse elimination on the level of a full circuit. Analysis confirmed the massive rewiring, 10-fold decrease in the number of synapses, that takes place as axons prune their motor units but add more synaptic areas at the NMJs with which they remain in contact.[39]
CNS synapses
Agrin appears not to be a central mediator of CNS synapse formation and there is active interest in identifying signals that mediate CNS synaptogenesis. Neurons in culture develop synapses that are similar to those that form in vivo, suggesting that synaptogenic signals can function properly in vitro. CNS synaptogenesis studies have focused mainly on glutamatergic synapses. Imaging experiments show that dendrites are highly dynamic during development and often initiate contact with axons. This is followed by recruitment of postsynaptic proteins to the site of contact. Stephen Smith and colleagues have shown that contact initiated by
Induction of synapse formation by glial factors: Barres and colleagues made the observation that factors in glial conditioned media induce synapse formation in retinal ganglion cell cultures. Synapse formation in the CNS is correlated with astrocyte differentiation suggesting that astrocytes might provide a synaptogenic factor. The identity of the astrocytic factors is not yet known.
Neuroligins and SynCAM as synaptogenic signals: Sudhof, Serafini, Scheiffele and colleagues have shown that neuroligins and SynCAM can act as factors that induce presynaptic differentiation. Neuroligins are concentrated at the postsynaptic site and act via neurexins concentrated in the presynaptic axons. SynCAM is a cell adhesion molecule that is present in both pre- and post-synaptic membranes.
Activity dependent mechanisms in the assembly of neural circuits
The processes of
Experimental techniques such as direct electrophysiological recording, fluorescence imaging using calcium indicators and optogenetic techniques have shed light on the nature and function of these early bursts of activity.
Recent studies confirm that microglia, the resident immune cell of the brain, establish direct contacts with the cell bodies of developing neurons, and through these connections, regulate neurogenesis, migration, integration and the formation of neuronal networks in an activity-dependent manner.[60]
In the developing
Contemporary diffusion-weighted
Numerous braingraphs, computed from the Human Connectome Project can be downloaded from the http://braingraph.org site. The Consensus Connectome Dynamics (CCD) is a remarkable phenomenon that was discovered by continuously decreasing the minimum confidence-parameter at the graphical interface of the Budapest Reference Connectome Server.[66][67] The Budapest Reference Connectome Server (http://connectome.pitgroup.org
It is hypothesized in
Synapse elimination
Several motorneurons compete for each neuromuscular junction, but only one survives until adulthood.[38] Competition in vitro has been shown to involve a limited neurotrophic substance that is released, or that neural activity infers advantage to strong post-synaptic connections by giving resistance to a toxin also released upon nerve stimulation. In vivo, it is suggested that muscle fibres select the strongest neuron through a retrograde signal or that activity-dependent synapse elimination mechanisms determine the identity of the "winning" axon at a motor endplate.[39]
Mapping
Brain mapping can show how an animal's brain changes throughout its lifetime. As of 2021, scientists mapped and compared the whole brains of eight C. elegans worms across their development on the neuronal level[69][70] and the complete wiring of a single mammalian muscle from birth to adulthood.[39]
Adult neurogenesis
Neurogenesis also occurs in specific parts of the adult brain.
See also
- Axon guidance
- KCC2
- Pioneer neuron
- Neural Darwinism
- Brain development timelines
- Malleable intelligence
- Role of cell adhesions in neural development
References
- ^ "Neurological Signs & Diseases". 2 November 2016. Archived from the original on 2016-11-02. Retrieved 1 May 2020.
- ^ "Neural Tube Defects". Retrieved 6 December 2011.
- ^ ISBN 978-0-87893-250-4.
- PMID 37507490.
- OCLC 914509705.
- ^ Wolpert 2015, pp. 522–526.
- ISBN 978-0-07-337825-1.
- ISBN 978-1-59259-065-0, retrieved 2024-04-06
- ^ PMID 25165044.
- ISBN 978-1-60535-192-6.[page needed]
- PMID 37507490.
- ^ ISBN 978-0-07-139011-8.[page needed]
- PMID 26499814.

- ^ Wolpert 2015, pp. 163.
- ISBN 978-1-4160-3445-2.
- PMID 19252484.
- ISBN 978-0-8385-7701-1.
- PMID 18805086.
- ^ PMID 26950681.
- PMID 30814272.
- ^ S2CID 6208462.
- S2CID 11704754.
- S2CID 41001390.
- PMID 26933693.
- S2CID 3041502.
- S2CID 2539488.
- PMID 11567613.
- S2CID 38910547.
- S2CID 41001390.
- S2CID 4349070.
- PMID 20646175.
- PMID 17215404.
- PMID 23843509.
- PMID 25505874.
- PMID 23175850.
- ^ PMID 14602813.
- PMID 12764035.
- ^ PMID 22745601.
- ^ S2CID 237598181.
- PMID 8780651.
- PMID 7965039.
- PMID 17920131.
- PMID 24314725.
- S2CID 19418882.
- PMID 2035024.
- PMID 8126550.
- PMID 11588185.
- S2CID 37387513.
- PMID 17408581.
- PMID 19287389.
- PMID 15297578.
- PMID 10230785.
- PMID 9777737.
- PMID 19874788.
- S2CID 24563014.
- PMID 3175637.
- PMID 21787461.
- ^ PMID 24314725.
- PMID 23060192.
- S2CID 252416407.
- ^ PMID 19471270.
- PMID 20676105.
- PMID 23596392.
- PMID 22197243.
- ^ OCLC 827948474.[page needed]
- S2CID 6563189.
- PMID 28174617.
- PMID 27362431.
- ^ "Why a tiny worm's brain development could shed light on human thinking". phys.org. Douglas, Isle Of Man UK: Science X. Lunenfeld-Tanenbaum Research Institute. Archived from the original on 20 June 2022. Retrieved 21 September 2021.
- S2CID 236927815.