Neural plate
| Neural plate | |
|---|---|
neural folds | |
| System | Central nervous system |
| Identifiers | |
| Latin | lamina neuralis |
| MeSH | D054258 |
| TE | plate_by_E5.13.1.0.1.0.1 E5.13.1.0.1.0.1 |
| Anatomical terminology] | |
The neural plate is a key developmental structure that serves as the basis for the nervous system. Cranial to the
Signaling proteins are also important in neural plate development, and aid in differentiating the tissue destined to become the neural plate. Examples of such proteins include bone morphogenetic proteins and cadherins. Expression of these proteins is essential to neural plate folding and subsequent neural tube formation.
Involvement in primary neurulation
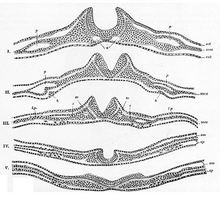
Generally divided into four, the process of
In primary neurulation, the layer of
Development
During the stage of neural plate formation the embryo consists of three
Stretched over the notochord, the ectodermal cells on the dorsal portion of the embryo are ultimately the ones that form the neural plate. Approximately half of those cells will be induced to remain ectoderm, while the other half will form the neural plate.[2][3]
There are four stages of neural plate and neural tube formation: formation, bending, convergence, and closure. The formation of the neural plate starts when dorsal mesoderm signals ectodermal cells above it to lengthen into columnar neural plate cells.[4] This different shape distinguishes the cells of the presumptive neural plate from other pre-epidermal cells. If the neural plate is separated by itself, it will still develop to make a thinner plate but will not form a neural tube. If the region containing presumptive epidermis and neural plate tissue is isolated, small neural folds will form. Elongation that occurs throughout the formation of the neural plate and closure of the neural tube is vital; the closing areas of the neural tube are seen to have very increased elongation activity in the midline compared to already closed areas when the plate was beginning to shape itself into a tube.[5]

The bending of the neural plate involves the formation of hinges, where the neural plate is connected to surrounding tissues. The midline of the neural plate is referred to the median hinge point (MHP). Cells in this area, known as medial hinge point cells because of their involvement with this structure, are stabilized and connected to the notochord. They are derived from the area of the neural plate anterior to primitive knot. The notochord will begin the shape changes in MHP cells. These cells will decrease in height and become wedge-shaped. Another type of hinge point occurs dorsal-laterally, referred to as dorsal-lateral hinge point (DLHP). These regions furrow and change shape in the same way as MHP cells do before connecting together to form the neural tube. It was seen in an experiment that without the notochord, the MHP characteristics did not develop correctly, so the neural plate and neural tube formation did not happen properly.[6] The communication between the neural plate and the notochord is important for the future induction and formation of the neural tube.
Closure of the neural tube is completed when the neural folds are brought together, adhering to each other. While the cells that remain as the neural tube form the brain and spinal cord, the other cells that were part of the neural plate migrate away from the tube as neural crest cells. After an epithelial–mesenchymal transition, these cells form the autonomic nervous system and certain cells of the peripheral nervous system.[7]
Cell signaling and essential proteins
Critical to the proper folding and function of the neural plate is N-cadherin, a type of cadherin protein associated with the nervous system. N-cadherin is critical to holding neural plate cells together. Additionally, cells destined to become neural plate cells express nerve cell adhesion molecule (NCAM) to further neural plate cohesion. Another cadherin, E-cadherin, is expressed by ectodermal cells in the process of neural plate development.[1]

Bone morphogenetic protein 4, or BMP4, is a transforming growth factor that causes the cells of the ectoderm to differentiate into skin cells. Without BMP4 the ectoderm cells would develop into neural cells. Axial mesoderm cells under the ectoderm secrete inhibitory signals called chordin, noggin and follistatin. These inhibitory signals prevent the action of BMP4, which would normally make the cells ectoderm; as a result, the overlying cells take their normal course and develop into neural cells. The cells in the ectoderm that circumscribe these neural cells do not receive the BMP4 inhibitor signals and as a result BMP4 induces these cells to develop into skin cells.[8]
Neural plate border specifiers are induced as a set of transcription factors. Distalless-5,
In a newly formed neural plate, PAX3 mRNA,
Low pSMAD 1, 5, 8 levels allow a greater mobility at the median hinge point than in lateral neural plate cells.[10] This flexibility allows for the pivoting and hinging that allows the buckling and lifting of the neural plate when formatting the neural tube. The neural plate has to be rigid enough for morphogenic movements to occur while being flexible enough to undergo shape and position changes for the transformation to the neural tube.
Other animals
The neural tube closes differently in various species, the distinctions between humans and chickens being some of the most studied. In humans, the neural tube fuses together from a central region of the embryo and moves outwards. In chickens, neural tube closure begins at the future midbrain region and it closes in both directions.[1] In birds and mammals, the closure does not occur at the same time.
In newt and general amphibian embryos, cell division is not a driving role in morphogenesis. Newt embryo cells are much larger and exhibit egg pigmentation to distinguish cells from each other. The newt neural plate doubles in length, decreases in apical width, and increases in thickness.[5] The plate edges rise dorsally and fold toward the midline to form the neural tube. The apical surface area decreases.
In chicken embryos, while the neural plate increases in length and decreases in apical width, the thickness of the plate does not change drastically. As the neural plate progresses through the
Research
Research on the neural plate began in earnest by looking into the determination of the ectoderm and its commitment to the neuronal path. With the development of research and laboratory techniques there have been major advances in the study of neurulation and the development and role of the neural plate in a growing embryo. The use of such techniques vary with the stage of development and overall research goals, but include such methods as cell labeling and grafting.[11]
Cell labelling
The process of
Similar to the process of in situ hybridization, immunofluorescence (IF) also allows for the determination of particular cell element's roles in development. In contrast to in situ hybridization however, immunofluorescence uses a fluorophore attached to an antibody with biomolecule target, such as proteins, rather than DNA and RNA sequences. The allows for the visualization of biomolecule elements of the cell. In the study of embryogenesis immunofluorescence may be used for purposes similar to hybridization, for the tracking of proteins that are involved in the development of the embryo and their specific time and place of production and use.[13] Current research has expanded on the immunofluorescence technique to combined it with the methods of in situ hybridization, either fluorescent or radioactive. This combination is believed to increase specificity and take away for the limitations of each individual technique. For example, this method with enhance counterstaining in a tissue and multiple protein labeling.[12]
Cell grafting
Cell grafting in the early stages of embryo development has provided crucial information on cell fates and the processes of determination. Grafting at specific stages of neurulation has advanced research on the signaling necessary for the proper development of the neural plate and other structures. The grafting of the ectoderm and neural structures is very specialized and delicate procedure, requiring the removal and marking of a desired group of cells, followed by their transplantation, for example, into a new area of the embryo.[14]
Grafting experiments done in Xenopus and chicken embryos show the neural plate's capability to induce other regions of cells, including the pre-placodal region, a group of ectodermal cells essential to the function of sensory organs.[15]
References
![]() This article incorporates text in the public domain from the 20th edition of Gray's Anatomy (1918)
This article incorporates text in the public domain from the 20th edition of Gray's Anatomy (1918)
- ^ ISBN 978-0878933846.
- ISBN 0-03-056748-3.
- ^ Human Embryology, Module 7, Section 7.2, http://www.embryology.ch/anglais/hdisqueembry/triderm10.html Archived 2013-01-16 at the Wayback Machine.
- S2CID 39722561.
- ^ JSTOR 3883562.
- PMID 2723610.
- ISBN 0-19-850263-X.
- PMID 9272958.
- ^ S2CID 17106597.
- PMID 23277919.
- ^ de Vellis J, Carpenter E. General Development of the Nervous System. In: Siegel GJ, Agranoff BW, Albers RW, et al., editors. Basic Neurochemistry: Molecular, Cellular and Medical Aspects. 6th edition. Philadelphia: Lippincott-Raven; 1999. Available from: https://www.ncbi.nlm.nih.gov/books/NBK28253/
- ^ PMID 16899759.
- ^ Sadler, T.W. (1986). "A potential role for spectrin during neurulation". J. Embryol. 94 (1): 73–82. Retrieved 27 April 2013.
- PMID 3655649. Retrieved 27 April 2013.
- PMID 16564335.
